Abstract
Background
Program death 1 (PD-1) and its ligand (PD-L1) have been identified as potential therapeutic targets for solid and hematologic malignancies. The current study aimed to assess PD-L1 expression in intrahepatic cholangiocarcinoma (ICC) and relate clinical outcomes to its expression.
Methods
Formalin-fixed, paraffin-embedded tumor specimens were obtained for patients undergoing surgery at Johns Hopkins Hospital between 1991 and 2011. Immunohistochemistry was used to assess PD-L1 expression in tumor-associated macrophages (TAMs) and within the tumor front (TF).
Results
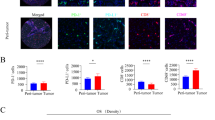
Of 54 tumor samples analyzed, 34 stained positive for PD-L1 expression on TAMs (TAMs+), and 39 stained positive for PD-L1 expression on cells within the tumor front (TF+). The TF+ patients were less likely to present with metastatic lymph nodes (N1 patients: 26.7 vs 7.7 %; p = 0.011), whereas all tumors with intrahepatic metastasis failed to demonstrate staining for PD-L1 around the tumor front (p = 0.020). Patients with tumors shown to be TAMs+ were less likely to present with multiple lesions (35.0 vs 8.8 %; p = 0.017). Patients with tumors exhibiting PD-L1 expression around the tumor front demonstrated a worse overall survival than TF patients (p = 0.008). Multivariable analysis showed that patients with tumors staining for PD-L1 in the tumor front had a 59.5 % reduced survival (TF− vs TF+: time ratio, 0.405; 95 % confidence interval, 0.215–0.761; p = 0.005).
Conclusion
Expression of PD-L1 was noted among a majority of patients, and PD-L1 expression within the tumor front was associated with a 60 % decreased survival. Future clinical trials are necessary to assess the safety and efficacy of anti-PD-L1 therapies among patients with ICC.



Similar content being viewed by others
References
Bridgewater J, Galle PR, Khan SA, et al. Guidelines for the diagnosis and management of intrahepatic cholangiocarcinoma. J Hepatol. 2014;60:1268–89.
Aljiffry M, Abdulelah A, Walsh M, et al. Evidence-based approach to cholangiocarcinoma: a systematic review of the current literature. J Am Coll Surg. 2009;208:134–47.
Shaib Y, El-Serag HB. The epidemiology of cholangiocarcinoma. Semin Liver Dis. 2004;24:115–25.
YH, Davila JA, McGlynn K, et al. Rising incidence of intrahepatic cholangiocarcinoma in the United States: a true increase? J Hepatol. 2004;40:472–7.
Topalian SL, Hodi FS, Brahmer JR, et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N Engl J Med. 2012;366:2443–54.
Brahmer JR, Tykodi SS, Chow LQM, et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N Engl J Med. 2012;366:2455–65.
Taube JM, Klein A, Brahmer JR, et al. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin Cancer Res. 2014;20:5064–74.
Schultheis AM, Scheel AH, Ozretić L, et al. PD-L1 expression in small cell neuroendocrine carcinomas. Eur J Cancer. 2015;51:421–6.
Llosa NJ, Cruise M, Tam A, et al. The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discov. 2015;5:43–51.
Le DT, Uram JN, Wang H, et al. PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med. 2015;372:2509–20.
Ye Y, Zhou L, Xie X, et al. Interaction of B7-H1 on intrahepatic cholangiocarcinoma cells with PD-1 on tumor-infiltrating T cells as a mechanism of immune evasion. J Surg Oncol. 2009;100:500–4.
Soares KC, Rucki AA, Wu AA, et al. PD-1/PD-L1 blockade together with vaccine therapy facilitates effector T-cell infiltration into pancreatic tumors. J Immunother. 2015;38:1–11.
Robert C, Ribas A, Wolchok JD, et al. Anti-programmed-death-receptor-1 treatment with pembrolizumab in ipilimumab-refractory advanced melanoma: a randomised dose-comparison cohort of a phase 1 trial. Lancet. 2014;384:1109–17.
Hamid O, Robert C, Daud A, et al. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134–44.
Taube JM, Anders RA, Young GD, et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci Transl Med. 2012; 4:127ra37.
Sabbatino F, Villani V, Yearley JH, et al. PD-L1 and HLA class I antigen expression and clinical course of the disease in intrahepatic cholangiocarcinoma. Clin Cancer Res. 2016;22(2), 470–478.
Leyva-Illades D, McMillin M, Quinn M, et al. Cholangiocarcinoma pathogenesis: role of the tumor microenvironment. Transl Gastrointest Cancer. 2012;1:71–80. Retrieved 7 Sep 2015 http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=3448449&tool=pmcentrez&rendertype=abstract.
Hasita H, Komohara Y, Okabe H, et al. Significance of alternatively activated macrophages in patients with intrahepatic cholangiocarcinoma. Cancer Sci. 2010;101:1913–9.
Tyson GL, El-Serag HB. Risk factors for cholangiocarcinoma. Hepatology. 2011;54:173–84.
Ramanathan S, Jagannathan N. Tumor-associated macrophage: a review on the phenotypes, traits, and functions. Iran J Cancer Prev. 2014;7:1–8. Retrieved 2 Sep 2015 http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=4142950&tool=pmcentrez&rendertype=abstract.
Liengswangwong U, Nitta T, Kashiwagi H, et al. Infrequent microsatellite instability in liver fluke infection-associated intrahepatic cholangiocarcinomas from Thailand. Int J Cancer. 2003;107:375–80.
Conflict of interest
Dr. Robert A. Anders receives research funding from Bristol-Myers Squibb and Five Prime Diagnostics. The remaining authors have no conflicts of interests.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gani, F., Nagarajan, N., Kim, Y. et al. Program Death 1 Immune Checkpoint and Tumor Microenvironment: Implications for Patients With Intrahepatic Cholangiocarcinoma. Ann Surg Oncol 23, 2610–2617 (2016). https://doi.org/10.1245/s10434-016-5101-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-016-5101-y