Abstract
Introduction
Calcifications can be indicative of malignancy, but calcifications also can be a byproduct of necrotic tissue as cancer cells die. Current treatment regimens require excision of calcifications. The objective of this study was to examine the correlation between the extent of calcification on mammography and actual tumor size after neoadjuvant chemotherapy (NAC) as well as magnetic resonance imaging (MRI) for comparison.
Methods
We retrospectively reviewed all patients at the University of California, San Diego, who underwent NAC for breast cancer between 2007 and 2013. Pearson correlation coefficients were computed between breast imaging and pathological measurements.
Results
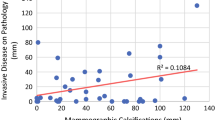
There were 136 patients total. Average age was 51 years. Fifty-three patients had calcifications on imaging (calc+); 83 did not (calc−). In the calc− group, extent of disease measured by mammogram (MMG) and MRI correlated moderately well with pathological tumor size (0.46 and 0.48, p = not significant). In the calc+ group, MRI was more likely to correlate with pathology than MMG (0.55 vs. −0.12, p = 0.01). Twenty-five calc+ patients had increased calcification after NAC; six of these had complete pathologic response. MRI correlated better with tumor size on pathology in patients with anti-HER2neu-based regimens than in patients with cytotoxic chemotherapy-alone regimens (0.88 vs. 0.4, p = 0.0001). MRI also is more accurate at predicting pathological tumor size in patients with triple negative disease (p = 0.002).
Conclusions
Magnetic resonance imaging correlated well while MMG calcification measurements correlated poorly with tumor size on final pathology. Extent of calcifications on diagnostic mammography may not be accurate in preoperative evaluation of breast cancers after NAC.
Similar content being viewed by others
References
Graham LJ, Shupe MP, Schneble EJ, et al. Current approaches and challengers in monitoring treatment responses in breast cancer. J Cancer. 2014;5:58–68.
Kaufmann M, von Minckwitz G, Bear HD, et al. Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: new perspectives 2006. Ann Oncol. 2007;18:1927–34.
Yeh E, Slanetz P, Kopans DB, et al. Prospective comparison of mammography, sonography, and MRI in patients undergoing neoadjuvant chemotherapy for palpable breast cancer. AJR Am J Roentgenol. 2005;184:868–77.
Prati R, Minami CA, Gornbein JA, Debruhi N, Chung D, Chang HR. Accuracy of clinical evaluation of locally advanced breast cancer in patients receiving neoadjuvant chemotherapy. Cancer. 2009;115:1194–202.
Rosen EL, Blackwell KL, Baker JA, Soo MS, Bentley RC, Yu D, Samulski TV, Dewhirst MW. Accuracy of MRI in the detection of residual breast cancer after neoadjuvant chemotherapy. AJR Am J Roentgenol. 2003;181:1275–82.
Fisher B, Bryant J, Wolmark N, et al. Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J Clin Oncol. 1998;16(8):2672–85.
Makris A, Powles TJ, Chang AJ, Hickish T, Tidy VA, Nash AG, Ford HT. A reduction in the requirements for mastectomy in a randomized trial of neoadjuvant chemoendocrine therapy in primary breast cancer. Ann Oncol. 1998;9:1179–84.
Wolff AC, Davidson NE. Primary systemic therapy in operable breast cancer. J Clin Oncol. 2000;18(7):1558–69.
Gralow JR, Burstein HJ, Wood W, et al. Preoperative therapy in invasive breast cancer: pathologic assessment and systemic therapy issues in operable disease. J Clin Oncol. 2008;26(5):814–9.
Van der Hage JH, van de Velde CCJH, Mieog SJSD. Preoperative chemotherapy for women with operable breast cancer (Review). Cochrane Libr. 2007;2:1–50.
Carey LA, Metzger R, Dees EC, et al. American joint committee on cancer tumor-node-metastasis stage after neoadjuvant chemotherapy and breast cancer outcome. JNCI. 2005;97(15):1137–42.
Liedtke C, Mazouni C, Hess KR. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26(8):1275–81.
Keurer H, Newman L, Buzdar A, et al. Residual metastatic axillary lymph nodes following neoadjuvant chemotherapy predict disease-free survival in patients with locally advanced breast cancer. Am J Surg. 1998;176:502–9.
Keune JD, Jeffe DB, Schootman M, Hoffman A, Gillanders WE, Aft RL. Accuracy of ultrasonography and mammography in predicting pathologic response after neoadjuvant chemotherapy for breast cancer. Am J Surg. 2010;199(4):477–84.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45(2):228.
Peintinger F, Keurer HM, Anderson K, et al. Accuracy of the combination of mammography and sonography in predicting tumor response in breast cancer patients after neoadjuvant chemotherapy. Ann Surg Oncol. 2006;13(11):1443–9.
Shin HJ, Kim HH, Ahn JH, Kim S-B, Jung KH, Gong G, Son BH, Ahn, SH. Comparison of mammography, sonography, MRI and clinical examination in patients with locally advanced or inflammatory breast cancer who underwent neoadjuvant chemotherapy. Br J Radiol. 2011;84:612–20.
Kwong MS, Chung GG, Horvath LJ, et al. Postchemotherapy MRI estimates residual disease compared with histopathology in responders to neoadjuvant therapy for locally advanced breast cancer. Cancer J. 2006;12(3):212–21.
Yuan Y, Chen X-S, Liu S-Y, Shen K-W. Accuracy of MRI inprediction of patjologic complete remission in breast cancer after preoperative therapy: a meta-analysis. AJR Am J Roentgenol. 2010;195:260–8.
Segal MC, Paulus DD, Hortobagyi GN. Advanced primary breast cancer: assessment at mammography of response to induction chemotherapy. Radiology. 1988;169:49–54.
Moskovic EC, Mansi JL, King DM, Murch CR, Smith IE. Mammography in the assessment of response to medical treatment of large primary breast cancer. Clin Radiol. 1993;47:339–44.
Esserman LE, d’Almeido M, Da Costa D, et al. Mammographic appearance of microcalcifications: can they change after neoadjuvant chemotherapy? Breast J. 2006;12(1):86–7.
Croshaw R, Shapiro-Wright H, Svensson E, Erb K, Julian T. Accuracy of clinical examination, digital mammogram, ultrasound, and MRI in determining postneoadjuvant pathological tumor response in operable breast cancer patients. Ann Surg Oncol. 2011;18:3160–3.
Chagpar AB, Middleton LP, Sahin AA, et al. Accuracy of physical examination, ultrasonography, and mammography in predicting residual pathological tumor size in patients treated with neoadjuvant chemotherapy. Ann Surg. 2006;243(2):257–64.
Weatherall PT, Evans GF, Metzger GJ, Saborrian MH, Leitch AM. MRI vs. histologic measurement of breast cancer following chemotherapy: comparison with X-ray mammography and palpation. J MRI. 2001;13:868–75.
Vinnicombe SJ, MacVicar AD, Guy RL, Sloane JP, Powles TJ, Knee G, Husband JE. Primary breast cancer: mammographic changes after neoadjuvant chemotherapy with pathologic correlation. Radiology. 1996;198:333–40.
Atkins JJ, Appleton CM, Fisher C, Gao F, Margenthaler JA. Which imaging modality is superior for prediction of response to neoadjuvant chemotherapy in patients with triple negative breast cancer? J Oncol. 2013;964863. doi:10.1155/2013/964863.
Herrada J, Iyer RB, Atkinson EN, et al. Relative value of physical examination, mammography, and breast sonography in evaluating the size of the primary tumor and regional lymph node metastases in women receiving neoadjuvant chemotherapy for locally advanced breast cancers. Clin Cancer Res. 1997;3:1565–9.
Marinovich ML, Houssami N, Macaskill, et al. Meta-analysis of magnetic resonance imaging in detecting residual breast cancer after neoadjuvant therapy. JNCI. 2013;105:321–33.
Marinovich ML, Macaskill P, Irwig L, et al. Meta-analysis of agreement between MRI and pathologic breast tumour size after neoadjuvant chemotherapy. Br J Cancer. 2013;109:1528–36.
Londero V, Bazzocchi M, Del Frate C, Puglisi F, Loreto C, Francescutti G, Zuiani C. Locally advanced breast cancer: comparison of mammography, sonography and MR imaging in evaluation of residual disease in women receiving neoadjuvant chemotherapy. Eur Radiol. 2004;14:1371–9.
Bosch AM, Kessels AGH, Beets GL, Rupa JD, Koster D, van Engelshoven JMA, von Meyenfeldt MF. Preoperative estimation of the pathological breast tumour size by physical examination, mammography, and ultrasound: a prospective study on 105 invasive tumors. EJR. 2003;48:285–92.
Li JJ, Chen C, Gu Y, Di G, Wu J, Liu G, Shao Z. The role of mammographic calcification in the neoadjuvant therapy of breast cancer imaging evaluation. PLOS. 2014;233(3):830–49.
McGuire KP, Toro-Burguete J, Dang H, et al. MRI staging after neoadjuvant chemotherapy for breast cancer: does tumor biology affect accuracy? Ann Surg Oncol. 2011;18:3149–54.
Ko ES, Han B-K, Kim R-B, et al. Analysis of factors that influence the accuracy of magnetic resonance imaging for predicting response after neoadjuvant chemotherapy in locally advanced breast cancer. Ann Surg Oncol. 2013;20:2562–8.
Cortazar P, Zhang L, Untch M, et al. Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis. Lancet. 2014 (in press).
Warner E, Messersmith H, Causer P, et al. Systematic review: using magnetic resonance imaging to screen women at high risk for breast cancer. Ann Intern Med. 2008;384(9938):164–72.
Berg WA, Zhang Z, Lehrer D, et al. Detection of breast cancer with addition of annual screening ultrasound or a single screening MRI to mammography in women with elevated breast cancer risk. JAMA. 2012;307:1394–404.
Berg WA, Gutierrez L, Nessalver MS, Carter WB, Bhargavan M, Lews RS, Ioffe OB. Diagnostic accuracy of mammography, clinical examination, US, and MR imaging in the preoperative assessment of breast cancer. Radiology. 2004;233:830–49.
Disclosure
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Accepted for presentation at ASBS, Las Vegas NV, May 2, 2014.
Rights and permissions
About this article
Cite this article
Weiss, A., Lee, K.C., Romero, Y. et al. Calcifications on Mammogram Do Not Correlate with Tumor Size After Neoadjuvant Chemotherapy. Ann Surg Oncol 21, 3310–3316 (2014). https://doi.org/10.1245/s10434-014-3914-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3914-0