Abstract
Introduction
TNM status is questioned as an exact predictor of survival in different tumour entities. Recently, lymph node ratio (LNR) has been described as a predictor of survival in patients with HNSCC. The purpose of this study was to evaluate to which degree LNR could be used as a more accurate predictor than TNM staging?
Methods
A total of 291 patients, with a follow-up of at least 3 years, were analyzed using log-rank statistic, univariate and multivariate data analyzes, and p values, for prediction of lymph node ratio on overall and recurrence-free survival.
Results
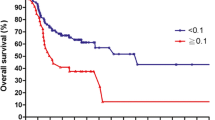
Survival differed significantly if patients were stratified for LNR. Impact of LNR on survival was significantly different even in patients with extracapsular spread. Patients with pN0 had no survival benefit compared with patients with pN1 or higher with a LNR lower than 6 %.
Conclusions
LNR is a prognostic tool in patients with a lymph node status pN0–pN2b. LNR remained significant even in patients with extracapsular spread, contrary to TNM status. With LNR, stratification for high-risk patients (higher than 6 % LNR) can be evaluated easily. We would suggest using LNR in the clinical routine.




Similar content being viewed by others
References
de Jong RJB, Hermans J, Molenaar J, Briaire JJ, le Cessie S. Prediction of survival in patients with head and neck cancer. Head Neck. 2001;23(9):718–24.
Manikantan K, Sayed SI, Syrigos KN, et al. Challenges for the future modifications of the TNM staging system for head and neck cancer: case for a new computational model? Cancer Treatment Rev. 2009;35(7):639–44.
Ebrahimi A, Clark JR, Zhang WJ, et al. Lymph node ratio as an independent prognostic factor in oral squamous cell carcinoma. Head Neck. 2011;33(9):1245–51.
Gil Z, Carlson DL, Boyle JO, et al. Lymph node density is a significant predictor of outcome in patients with oral cancer. Cancer. 2009;115(24):5700–10.
Shrime MG, Bachar G, Lea J, et al. Nodal ratio as an independent predictor of survival in squamous cell carcinoma of the oral cavity. Head Neck. 2009;31(11):1482–88.
Lanzer M, Kruse A, Lübbers HT, Zemann W, Reinisch S. Lymph node ratio and capsule penetration as independent risk factors in head and neck squamous cell carcinoma. Head Neck Oncol. 2012;5(4):89–97.
Urban D, Gluck I, Pfeffer MR, Symon Z, Lawrence YR. Lymph node ratio predicts the benefit of post-operative radiotherapy in oral cavity cancer. Radiother Oncol. 2013;106(1):74–9.
Robbins KT, Clayman G, Levine PA, et al. Neck dissection classification update—revisions proposed by the American Head and Neck Society and the American Academy of Otolaryngology-Head and Neck Surgery. Arch Otolaryngol Head Neck Surg. 2002;128(7):751–8.
Lausen B, Schumacher M. Maximally selected rank statistics. Biometrics 1992;48(1):73–85.
Goldstein DP, Bachar GY, Lea J, et al. Outcomes of squamous cell cancer of the oral tongue managed at the Princess Margaret Hospital. Head Neck. 2013;35(5):632–41.
Shingaki S, Takada M, Sasai K, et al. Impact of lymph node metastasis on the pattern of failure and survival in oral carcinomas. Am J Surg. 2003;185(3):278–84.
Svatek R, Shariat SF. Re: lymph node density is superior to TNM nodal status in predicting disease-specific survival after radical cystectomy for bladder cancer: analysis of pooled data from MDACC and MSKCC. Eur Urol. 2008;54(3):690–1.
Kassouf W, Agarwal PK, Herr HW, et al. Lymph node density is superior to TNM nodal status in predicting disease-specific survival after radical cystectomy for bladder cancer: analysis of pooled data from MDACC and MSKCC. J Clin Oncol. 2008;26(1):121–6.
Ooki A, Yamashita K, Kobayashi N, et al. Lymph node metastasis density and growth pattern as independent prognostic factors in advanced esophageal squamous cell carcinoma. World J Surg. 2007;31(11):2184–91.
Clayman GL, Frank DK. Selective neck dissection of anatomically appropriate levels is as efficacious as modified radical neck dissection for elective treatment of the clinically negative neck in patients with squamous cell carcinoma of the upper respiratory and digestive tracts. Arch Otolaryngol Head Neck Surg. 1998;124(3):348–52.
Patel RS, Clark JR, Gao K, O’Brien CJ. Effectiveness of selective neck dissection in the treatment of the clinically positive neck. Head Neck. 2008;30(9):1231–6.
Shepard PM, Olson J, Harari PM, Leverson G, Hartig GK. Therapeutic selective neck dissection outcomes. Otolaryngol Head Neck Surg. 2010;142(5):741–6.
Friedman M, Lim JW, Dickey W et al. Quantification of lymph nodes in selective neck dissection. Laryngoscope. 1999;109(3):368–70.
Beasley NJP, Prevo R, Banerji S, et al. Intratumoral lymphangiogenesis and lymph node metastasis in head and neck cancer. Cancer Res. 2002;62(5):1315–20.
Stacker SA, Achen MG, Jussila L, Baldwin ME, Alitalo K. Lymphangiogenesis and cancer metastasis. Nat Rev Cancer. 2002;2(8):573–83.
Dias FL, Lima RA, Kligerman J, et al. Relevance of skip metastases for squamous cell carcinoma of the oral tongue and the floor of the mouth. Otolaryngol Head Neck Surg. 2006;134(3):460–5.
Myers JN, Greenberg JS, Mo V, Roberts D. Extracapsular spread: a significant predictor of treatment failure in patients with squamous cell carcinoma of the tongue. Cancer. 2001;92(12):3030–6.
Acknowledgement
There is no financial disclosure.
Conflict of interest
Authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conduction of Study
Patient data derived from patients treated at the University Hospital of Graz (1). The study was approved by the Institutional Ethics Committee (EK-Nr. 21-020 ex 09/10).
Rights and permissions
About this article
Cite this article
Reinisch, S., Kruse, A., Bredell, M. et al. Is Lymph-node Ratio a Superior Predictor than Lymph Node Status for Recurrence-free and Overall Survival in Patients with Head and Neck Squamous Cell Carcinoma?. Ann Surg Oncol 21, 1912–1918 (2014). https://doi.org/10.1245/s10434-014-3634-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-3634-5