Abstract
Purpose
To clarify the prognostic impact of body mass index (BMI) in patients with esophageal squamous cell carcinoma (ESCC).
Methods
Two hundred forty-three patients who underwent esophagectomy for ESCC from April 2005 through December 2010 were eligible. Prognoses of the patients were compared between groups stratified according to BMI. We also analyzed the survival difference using propensity score matching to adjust differences in staging and treatment.
Results
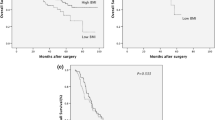
Low, normal, and high BMI groups had 35, 177, and 31 patients, respectively. The low BMI group included more advanced cases than did the normal BMI group, while tumor stage was equivalent in the normal and high BMI groups. Disease-free survival of the low and high BMI groups was significantly worse than that of the normal BMI group (P < 0.0001 between the low and normal BMI groups; P = 0.0076 between the normal and high BMI groups). Disease-free survival of the high BMI group was significantly worse than that of the normal BMI group in the propensity score-matched cohort (P = 0.0020). Multivariate analysis in this cohort demonstrated that high BMI was an independent prognostic factor (hazard ratio 2.949, 95 % confidence interval, 1.132–7.683).
Conclusions
High BMI was an independent prognostic factor after curative esophagectomy for ESCC. Although further analysis is required to clarify the influence of overweight on the biological features of ESCC, glucose metabolism may be a therapeutic target for ESCC.


Similar content being viewed by others
References
Calle EE, Rodriguez C, Walker-Thurmond K, et al. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of US adults. N Engl J Med. 2003;348:1625–38.
Protani M, Coory M, Martin JH. Effects of obesity on survival of women with breast cancer: systematic review and meta-analysis. Breast Cancer Res Treat. 2010;123:627–35.
Sinicrope FA, Foster NR, Sargent DJ, et al. Obesity is an independent prognostic variable in colon cancer survivors. Clin Cancer Res. 2010;16:1884–93.
Tokunaga M, Hiki N, Fukunaga T, et al. Better 5-year survival rate following curative gastrectomy in overweight patients. Ann Surg Oncol. 2009;16:3245–51.
Kamat AM, Shock RP, Rosser CJ, et al. Prognostic value of body mass index in patients undergoing nephrectomy for localized renal tumors. Urology. 2004;63:46–50.
Healy LA, Ryan AM, Gopinath B, et al. Impact of obesity on outcomes in the management of localized adenocarcinoma of the esophagus and esophagogastric junction. J Thorac Cardiovasc Surg. 2007;134:1284–91.
Morgan MA, Lewis WG, Hopper AN, et al. Prognostic significance of body mass indices for patients undergoing esophagectomy for cancer. Dis Esophagus. 2007;20:29–35.
Hayashi Y, Correa AM, Hofstetter WL, et al. The influence of high body mass index on the prognosis of patients with esophageal cancer after surgery as primary therapy. Cancer. 2010;116:5619–27.
Yoon HH, Lewis MA, Shi Q, et al. Prognostic impact of body mass index stratified by smoking status in patients with esophageal adenocarcinoma. J Clin Oncol. 2011;29:4561–7.
Sobin LH, GospodarowiczMK, Wittekind C. TNM classification of malignant tumors. 7th ed. Oxford: Wiley-Blackwell; 2010.
World Health Organization. WHO global database on body mass index. http://apps.who.int/bmi/index.jsp?introPage_intro_3.html. Accessed 21 Apr 2013.
Melis M, Weber JM, McLoughlin JM, et al. An elevated body mass index does not reduce survival after esophagectomy for cancer. Ann Surg Oncol. 2011;18:824–31.
Scarpa M, Cagol M, Bettini S, et al. Overweight patients operated on for cancer of the esophagus survive longer than normal-weight patients. J Gastrointest Surg. 2013;17:218–27.
Han-Geurts IJ, Hop WC, Tran TC, et al. Nutritional status as a risk factor in esophageal surgery. Dig Surg. 2006;23:159–63.
Swisher SG, Wynn P, Putnam JB, et al. Salvage esophagectomy for recurrent tumors after definitive chemoradiotherapy and radiotherapy. J Thorac Cardiovasc Surg. 2002;123:175–83.
Srokowski TP, Fang S, Hortobagyi GN, et al. Impact of diabetes mellitus on complications and outcomes of adjuvant chemotherapy in older patients with breast cancer. J Clin Oncol. 2009;27:2170–6.
Barone BB, Yeh HC, Snyder CF, et al. Long-term all-cause mortality in cancer patients with preexisting diabetes mellitus: a systematic review and meta-analysis. JAMA. 2008;300:2754–64.
Giovannucci E, Harlan DM, Archer MC, et al. Diabetes and cancer: a consensus report. Diabetes Care. 2010;60:207–21.
Considine RV, Sinha MK, Heiman ML, et al. Serum immunoreactive–leptin concentrations in normal-weight and obese humans. N Engl J Med. 1996;334:292–5.
Ogunwobi O, Mutungi G, Beales IL. Leptin stimulates proliferation and inhibits apoptosis in Barrett’s esophageal adenocarcinoma cells by cyclooxygenase-2-dependent, prostaglandin-E2-mediated transactivation of the epidermal growth factor receptor and c-Jun NH2-terminal kinase activation. Endocrinology. 2006;147:4505–16.
Chen SC, Chou CK, Wong FH, et al. Overexpression of epidermal growth factor and insulin-like growth factor receptors and autocrine stimulation in human esophageal carcinoma cells. Cancer Res. 1991;51:1898–903.
Sohda M, Kato H, Miyazaki T, et al. The role of insulin-like growth factor 1 and insulin-like growth factor binding protein 3 in human esophageal cancer. Anticancer Res. 2004;24:3029–34.
Juan HC, Tsai HT, Chang PH, et al. Insulin-like growth factor 1 mediates 5-fluorouracil chemoresistance in esophageal carcinoma cells through increasing survivin stability. Apoptosis. 2011;16:174–83.
Fidler MJ, Shersher DD, Borgia JA, et al. Targeting the insulin-like growth factor receptor pathway in lung cancer: problems and pitfalls. Ther Adv Med Oncol. 2012;4:51–60.
Brown RS, Goodman TM, Zasadny KR, et al. Expression of hexokinase II and Glut I in untreated human breast cancer. Nucl Med Biol. 2002;29:43–53.
Meyers BF, Downey RJ, Decker PA, et al. The utility of positron emission tomography in staging of potentially operable carcinoma of the thoracic esophagus. J Thorac Cardiovasc Surg. 2007;133:738–45.
Hiyoshi Y, Watanabe M, Imamura Y, et al. The relationship between the glucose transporter type 1 expression and 18F-fluorodeoxyglucose uptake in esophageal squamous cell carcinoma. Oncology. 2008;76:286–92.
Toh Y, Oki E, Minami K, et al. Follow-up and recurrence after a curative esophagectomy for patients with esophageal cancer: the first indicators for recurrence and their prognostic values. Esophagus. 2010;7:37–43.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. 1
Changes in BMIs during preoperative treatment. Significant decrease in BMI was observed in patients who underwent definitive chemoradiotherapy (P=0.0098, paired t-test). NAC, Neoadjuvant chemotherapy; NACRT, Neoadjuvant chemoradiotherapy; dCRT, Definitive chemoradiotherapy. (PDF 86 kb)
Rights and permissions
About this article
Cite this article
Watanabe, M., Ishimoto, T., Baba, Y. et al. Prognostic Impact of Body Mass Index in Patients with Squamous Cell Carcinoma of the Esophagus. Ann Surg Oncol 20, 3984–3991 (2013). https://doi.org/10.1245/s10434-013-3073-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-013-3073-8