Abstract
Background
Magnetic resonance imaging (MRI) is highly accurate in local staging of rectal cancer. It can identify features known to be associated with increased risk of metastatic disease. We evaluated the incidence of synchronous metastatic disease on fludeoxyglucose–positron emission tomography/computed tomography (FDG-PET/CT) and contrast-enhanced multiple-row detector computed tomography (ceMDCT) in MRI-stratified high- and low-risk rectal cancers. The aim was to determine the incidence of synchronous metastatic disease according to MRI risk features.
Methods
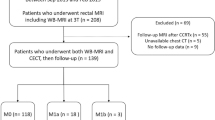
A total of 236 patients with rectal cancer were recruited to a study evaluating FDG-PET/CT. All patients underwent MRI staging and were stratified into high and low risk (high risk: extramural venous invasion, extramural spread of >5 mm or T4, involved circumferential resection margin or intersphincteric plane involved for low rectal tumors). Confirmed metastases were those identified on FDG-PET/CT and ceMDCT.
Results
Imaging data were available for 230 (97.5%) of 236 patients. Incidence of confirmed distant metastases was significantly greater in the MRI high-risk group, with 28 (20.7%) of 135 (95% confidence interval [CI] 14.8–28.3), versus the low-risk group, with 4 (4.2%) of 95 (95% CI 1.7–10.3) (odds ratio 6.0, 95% CI 2.0–17.6, P < 0.001).
Conclusions
Adverse features found on rectal MRI identify patients at increased risk of synchronous metastatic disease. This group may benefit from additional preoperative investigation for synchronous metastases such as FDG-PET/CT or liver MRI and from alternative neoadjuvant chemotherapy regimens including induction chemotherapy.




Similar content being viewed by others
References
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ. Cancer statistics, 2009. CA Cancer J Clin. 2009;59:225–49.
Scholefield JH. Guidelines for the management of colorectal cancer. 3rd ed. London: Association of Coloproctology of Great Britain and Ireland; 2007.
Valentini V, Aristei C, Glimelius B, et al. Multidisciplinary rectal cancer management: 2nd European Rectal Cancer Consensus Conference (EURECA-CC2). Radiother Oncol. 2009;92:148–63.
Smith NJ, Shihab O, Arnaout A, Swift RI, Brown G. MRI for detection of extramural vascular invasion in rectal cancer. AJR Am J Roentgenol. 2008;191:1517–22.
Brown G, Radcliffe AG, Newcombe RG, Dallimore NS, Bourne MW, Williams GT. Preoperative assessment of prognostic factors in rectal cancer using high-resolution magnetic resonance imaging. Br J Surg. 2003;90:355–64.
MERCURY Study Group. Extramural depth of tumor invasion at thin-section MR in patients with rectal cancer: results of the MERCURY study. Radiology. 2007;243:132–9.
Taylor FG, Quirke P, et al. Preoperative high-resolution magnetic resonance imaging can identify good prognosis stage I, II, and III rectal cancer best managed by surgery alone: a prospective, multicenter, European study. Ann Surg. 2011;253(4):711–9.
Chau I, Brown G, Cunningham D, et al. Neoadjuvant capecitabine and oxaliplatin followed by synchronous chemoradiation and total mesorectal excision in magnetic resonance imaging-defined poor-risk rectal cancer. J Clin Oncol. 2006;24:668–74.
Dewdney A, Cunningham D, Tabernero J, et al. EXPERT-C: a randomized phase II European multicenter trial of neoadjuvant chemotherapy (capecitabine/oxaliplatin) and chemoradiation (CRT) with or without cetuximab followed by total mesorectal excision (TME) in patients with MRI-defined high-risk rectal cancer. Presented at Gastrointestinal Cancers Symposium, San Francisco, 2011.
Newcombe RG. Improved confidence intervals for the difference between binomial proportions based on paired data. Stat Med. 1998;17:2635–50.
Aloia TA, Fahy BN. A decision analysis model predicts the optimal treatment pathway for patients with colorectal cancer and resectable synchronous liver metastases. Clin Colorectal Cancer. 2008;7:197–201.
Slupski M, Wlodarczyk Z, Jasinski M, Masztalerz M, Tujakowski J. Outcomes of simultaneous and delayed resections of synchronous colorectal liver metastases. Can J Surg. 2009;52:E241–4.
Hillingso JG, Wille-Jorgensen P. Staged or simultaneous resection of synchronous liver metastases from colorectal cancer—a systematic review. Colorectal Dis. 2009;11:3–10.
Mentha G, Majno PE, Andres A, Rubbia-Brandt L, Morel P, Roth AD. Neoadjuvant chemotherapy and resection of advanced synchronous liver metastases before treatment of the colorectal primary. Br J Surg. 2006;93:872–8.
Brouquet A, Mortenson MM, Vauthey JN, et al. Surgical strategies for synchronous colorectal liver metastases in 156 consecutive patients: classic, combined or reverse strategy? J Am Coll Surg. 2010;210:934–41.
Heriot AG, Hicks RJ, Drummond EG, et al. Does positron emission tomography change management in primary rectal cancer? A prospective assessment. Dis Colon Rectum. 2004;47:451–8.
Orlacchio A, Schillaci O, Fusco N, et al. Role of PET/CT in the detection of liver metastases from colorectal cancer. Radiol Med. 2009;114:571–85.
Kong G, Jackson C, Koh DM, et al. The use of 18F-FDG PET/CT in colorectal liver metastases—comparison with CT and liver MRI. Eur J Nucl Med Mol Imaging. 2008;35:1323–9.
Reinhardt MJ, Wiethoelter N, Matthies A, et al. PET recognition of pulmonary metastases on PET/CT imaging: impact of attenuation-corrected and non-attenuation-corrected PET images. Eur J Nucl Med Mol Imaging. 2006;33:134–9.
Dukes CE, Bussey HJ. The spread of rectal cancer and its effect on prognosis. Br J Cancer. 1958;12:309–20.
Dukes C, Bussey HJ. Venous spread in rectal cancer. Proc R Soc Med. 1941;34:571–3.
Sauer R, Becker H, Hohenberger W, et al. Preoperative versus postoperative chemoradiotherapy for rectal cancer. N Engl J Med. 2004;351:1731–40.
Grinnell RS. The grading and prognosis of carcinoma of the colon and rectum. Ann Surg. 1939;109:500–33.
Quirke P, Williams GT, Ectors N, Ensari A, Piard F, Nagtegaal I. The future of the TNM staging system in colorectal cancer: time for a debate? Lancet Oncol. 2007;8:651–7.
Galizia G, Orditura M, Ferraraccio F, et al. The lymph node ratio is a powerful prognostic factor of node-positive colon cancers undergoing potentially curative surgery. World J Surg. 2009;33:2704–13.
Kim JH, Beets GL, Kim MJ, Kessels AG, Beets-Tan RG. High-resolution MR imaging for nodal staging in rectal cancer: are there any criteria in addition to the size? Eur J Radiol. 2004;52:78–83.
Chua YJ, Barbachano Y, Cunningham D, et al. Neoadjuvant capecitabine and oxaliplatin before chemoradiotherapy and total mesorectal excision in MRI-defined poor-risk rectal cancer: a phase 2 trial. Lancet Oncol. 2010;11:241–8.
Disclosure
There are no commercial interests to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hunter, C.J., Garant, A., Vuong, T. et al. Adverse Features on Rectal MRI Identify a High-risk Group that May Benefit from More Intensive Preoperative Staging and Treatment. Ann Surg Oncol 19, 1199–1205 (2012). https://doi.org/10.1245/s10434-011-2036-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-011-2036-1