Abstract
Background
Tissue banking has become a major initiative at many oncology centers. The influence of warm ex-vivo ischemia times, storage times, and biobanking protocols on RNA integrity and subsequent microarray data is not well documented.
Methods
A prospective institutional review board–approved protocol for the banking of abdominal neoplasms was initiated at Memorial Sloan-Kettering Cancer Center in 2001. Sixty-four representative pancreas cancer specimens snap-frozen at various ex-vivo procurement times (≤10 min, 11–30 min, 31–60 min, >1 h) and banked during three time periods (2001–2004, 2004–2006, 2006–2008) were processed. RNA integrity was determined by microcapillary electrophoresis using the RNA integrity number (RIN) algorithm and by results of laser-capture microdissection (LCM).
Results
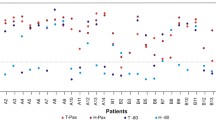
Overall, 42% of human pancreas cancer specimens banked under a dedicated protocol yielded RNA with a RIN of ≥7. Limited warm ex-vivo ischemia times did not negatively impact RNA quality (percentage of tissue with total RNA with RIN of ≥7 for ≤10 min, 42%; 11–30 min, 58%; 31–60 min, 33%; >60 min, 42%), and long-term storage of banked pancreas cancer biospecimens did not negatively influence RNA quality (total RNA with RIN of ≥7 banked 2001–2004, 44%; 2004–2006, 38%; 2006–2008, 50%). RNA retrieved from pancreatic cancer samples with RIN of ≥7 subject to LCM yielded RNA suitable for further downstream applications.
Conclusions
Fresh-frozen pancreas tissue banked within a standardized research protocol yields high-quality RNA in approximately 50% of specimens and can be used for enrichment by LCM. Quality of tissues of the biobank were not adversely impacted by limited variations of warm ischemia times or different storage periods. This study shows the challenges and investments required to initiate and maintain high-quality tissue repositories.





Similar content being viewed by others
References
Garman KS, Nevins JR, Potti A. Genomic strategies for personalized cancer therapy. Hum Mol Genet. 2007;16(Spec No. 2): R226–32.
Acharya CR, Hsu DS, Anders CK, et al. Gene expression signatures, clinicopathological features, and individualized therapy in breast cancer. JAMA. 2008;299:1574–87.
Brenton JD, Carey LA, Ahmed AA, Caldas C. Molecular classification and molecular forecasting of breast cancer: ready for clinical application? J Clin Oncol. 2005;23:7350–60.
Compton C. Getting to personalized cancer medicine: taking out the garbage. Cancer. 2007;110:1641–3.
National Cancer Institute. NCI best practices for biospecimen resources. 2007. http://www.biospecimens.cancer.gov/practices/.
Srivastava S, Gray JW, Reid BJ, et al; Translational Research Working Group. Translational Research Working Group developmental pathway for biospecimen-based assessment modalities. Clin Cancer Res. 2008;14:5672–7.
Hwang RF, Wang H, Lara A, et al. Development of an integrated biospecimen bank and multidisciplinary clinical database for pancreatic cancer. Ann Surg Oncol. 2008;15:1356–66.
Albores-Saavedra J, Heffess C, Hruban RH, Klimstra D, Longnecker D. Recommendations for the reporting of pancreatic specimens containing malignant tumors. The Association of Directors of Anatomic and Surgical Pathology. Am J Clin Pathol. 1999;111:304–7.
Bild AH, Yao G, Chang JT, et al. Oncogenic pathway signatures in human cancers as a guide to targeted therapies. Nature. 2006;439(7074):353–7.
Potti A, Mukherjee S, Petersen R, et al. A genomic strategy to refine prognosis in early-stage non-small-cell lung cancer. N Engl J Med. 2006;355:570–80.
Copois V, Bibeau F, Bascoul-Mollevi C, et al. Impact of RNA degradation on gene expression profiles: assessment of different methods to reliably determine RNA quality. J Biotechnol. 2007;127:549–59.
Strand C, Enell J, Hedenfalk I, Fernö M. RNA quality in frozen breast cancer samples and the influence on gene expression analysis—a comparison of three evaluation methods using microcapillary electrophoresis traces. BMC Mol Biol. 2007;8:38.
Weis S, Llenos IC, Dulay JR, et al. Quality control for microarray analysis of human brain samples: The impact of postmortem factors, RNA characteristics, and histopathology. J Neurosci Methods. 2007;165:198–209.
Jewell SD, Srinivasan M, McCart LM, et al. Analysis of the molecular quality of human tissues: an experience from the Cooperative Human Tissue Network. Am J Clin Pathol. 2002;118:733–41.
Dash A, Maine IP, Varambally S, et al. Changes in differential gene expression because of warm ischemia time of radical prostatectomy specimens. Am J Pathol. 2002;161:1743–8.
Spruessel A, Steimann G, Jung M, et al. Tissue ischemia time affects gene and protein expression patterns within minutes following surgical tumor excision. Biotechniques. 2004;36:1030–7.
Lin DW, Coleman IM, Hawley S, et al. Influence of surgical manipulation on prostate gene expression: implications for molecular correlates of treatment effects and disease prognosis. J Clin Oncol. 2006;24:3763–70.
Ohashi Y, Creek KE, Pirisi L, Kalus R, Young SR. RNA degradation in human breast tissue after surgical removal: a time-course study. Exp Mol Pathol. 2004;77:98–103.
Barnes RO, Parisien M, Murphy LC, Watson PH. Influence of evolution in tumor biobanking on the interpretation of translational research. Cancer Epidemiol Biomarkers Prev. 2008;17:3344–50.
Schlomm T, Näkel E, Lübke A, et al. Marked gene transcript level alterations occur early during radical prostatectomy. Eur Urol. 2008;53:333–44.
Iacobuzio-Donahue CA, Maitra A, Shen-Ong GL, et al. Discovery of novel tumor markers of pancreatic cancer using global gene expression technology. Am J Pathol. 2002;160:1239–49.
Sato N, Fukushima N, Maitra A, et al. Gene expression profiling identifies genes associated with invasive intraductal papillary mucinous neoplasms of the pancreas. Am J Pathol. 2004;164:903–14.
Buchholz M, Braun M, Heidenblut A, et al. Transcriptome analysis of microdissected pancreatic intraepithelial neoplastic lesions. Oncogene. 2005;24:6626–36.
Prasad NB, Biankin AV, Fukushima N, et al. Gene expression profiles in pancreatic intraepithelial neoplasia reflect the effects of Hedgehog signaling on pancreatic ductal epithelial cells. Cancer Res. 2005;65:1619–26.
Imbeaud S, Graudens E, Boulanger V, et al. (2005) Towards standardization of RNA quality assessment using user-independent classifiers of microcapillary electrophoresis traces. Nucleic Acids Res. 33:56.
Schroeder A, Mueller O, Stocker S, et al. (2006) The RIN: an RNA integrity number for assigning integrity values to RNA measurements. BMC Mol Biol. 7:3.
Micke P, Ohshima M, Tahmasebpoor S, et al. Biobanking of fresh frozen tissue: RNA is stable in nonfixed surgical specimens. Lab Invest. 2006;86:202–11.
Fleige S, Pfaffl MW. RNA integrity and the effect on the real-time qRT-PCR performance. Mol Aspects Med. 2006;27:126–39.
Blansfield JA, Caragacianu D, Alexander HR 3rd, et al. Combining agents that target the tumor microenvironment improves the efficacy of anticancer therapy. Clin Cancer Res. 2008;14:270–80.
Lee J, Hever A, Willhite D, Zlotnik A, Hevezi P. Effects of RNA degradation on gene expression analysis of human postmortem tissues. FASEB J. 2005;19:1356–8.
Author information
Authors and Affiliations
Corresponding author
Additional information
The first two authors contributed equally to this study.
Rights and permissions
About this article
Cite this article
Rudloff, U., Bhanot, U., Gerald, W. et al. Biobanking of Human Pancreas Cancer Tissue: Impact of Ex-Vivo Procurement Times on RNA Quality. Ann Surg Oncol 17, 2229–2236 (2010). https://doi.org/10.1245/s10434-010-0959-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-010-0959-6