Abstract
Background
This study investigated the prognostic significance of time to the prostate-specific antigen nadir (TTPN) and its relationship to survival beyond TTPN in metastatic prostate cancer after primary androgen-deprivation therapy (ADT).
Methods
All metastatic prostate cancer patients treated with primary ADT from 2000 to 2009 were reviewed. The prognostic significance of TTPN in predicting progression-free survival (PFS) beyond TTPN and overall survival (OS) beyond TTPN was analyzed using the Cox regression model. The median PFS and OS were plotted against TTPN on a monthly interval. The PFS beyond TTPN and the OS beyond TTPN with reference to TTPN were calculated and presented.
Results
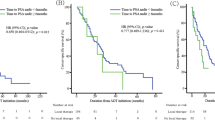
The study enrolled 419 patients with a median follow-up period of 38 months. The findings showed that TTPN was a significant prognostic indicator for both PFS beyond TTPN (hazard ratio [HR] 0.72, 95 % confidence interval [CI] 0.52–0.99, p = 0.04) and OS beyond TTPN (HR 0.65, 95 % CI 0.47–0.90, p = 0.01) according to Cox regression analyses. The relationship between TTPN and survival beyond TTPN consisted of three phases. In the first phase (<3 months for PFS and <6 months for OS), the survival beyond TTPN increased with TTPN. In the second phase (3–17 months for PFS and 6–20 months for OS), the survival beyond TTPN remained relatively static. In the third phase (>17 months for PFS and >20 months for OS), the survival beyond TTPN increased exponentially with TTPN.
Conclusions
In this study, TTPN was a good prognostic indicator for PFS beyond TTPN and OS beyond TTPN in metastatic prostate cancer cases after primary ADT. Different TTPNs had different implications for predicting survival beyond TTPN.


Similar content being viewed by others
References
Huggins C, Hodges CV. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. J Urol. 2002; 168:9–12.
Shahinian VB, Kuo YF, Freeman JL, Orihuela E, Goodwin JS. Increasing use of gonadotropin-releasing hormone agonists for the treatment of localized prostate carcinoma. Cancer. 2005;103:1615–24.
Ablin RJ, Pfeiffer L, Gonder MJ, Soanes WA. Precipitating antibody in the sera of patients treated cryosurgically for carcinoma of the prostate. Exp Med Surg. 1969;27:406–10.
Nadji M, Tabei SZ, Castro A, Chu TM, Murphy GP, Wang MC, Morales AR. Prostatic-specific antigen: an immunohistologic marker for prostatic neoplasms. Cancer. 1981;48:1229–32.
Crawford ED, Bennett CL, Andriole GL, Garnick MB, Petrylak DP. The utility of prostate-specific antigen in the management of advanced prostate cancer. BJU Int. 2013;112:548–60.
Hori S, Jabbar T, Kachroo N, Vasconcelos JC, Robson CN, Gnanapragasam VJ. Outcomes and predictive factors for biochemical relapse following primary androgen deprivation therapy in men with bone scan negative prostate cancer. J Cancer Res Clin Oncol. 2011;137:235–41.
Sasaki T, Onishi T, Hoshina A. Nadir PSA level and time to PSA nadir following primary androgen deprivation therapy are the early survival predictors for prostate cancer patients with bone metastasis. Prostate Cancer Prostatic Dis. 2011;14:248–52.
Huang SP, Bao BY, Wu MT, et al. Impact of prostate-specific antigen (PSA) nadir and time to PSA nadir on disease progression in prostate cancer treated with androgen-deprivation therapy. Prostate. 2011;71:1189–97.
Huang SP, Bao BY, Wu MT, et al. Significant associations of prostate-specific antigen nadir and time to prostate-specific antigen nadir with survival in prostate cancer patients treated with androgen-deprivation therapy. Aging Male. 2012;15:34–41.
Choueiri TK, Xie W, D’Amico AV, et al. Time to prostate-specific antigen nadir independently predicts overall survival in patients who have metastatic hormone-sensitive prostate cancer treated with androgen-deprivation therapy. Cancer. 2009;115:981–7.
Morote J, Trilla E, Esquena S, Abascal JM, Reventos J. Nadir prostate-specific antigen best predicts the progression to androgen-independent prostate cancer. Int J Cancer. 2004;108:877–81.
Benaim EA, Pace CM, Lam PM, Roehrborn CG. Nadir prostate-specific antigen as a predictor of progression to androgen-independent prostate cancer. Urology. 2002;59:73–8.
D’Amico AV, McLeod DG, Carroll PR, Cullen J, Chen MH. Time to an undetectable prostate-specific antigen (PSA) after androgen suppression therapy for postoperative or postradiation PSA recurrence and prostate cancer-specific mortality. Cancer. 2007;109:1290–5.
Chung CS, Chen MH, Cullen J, McLeod D, Carroll P, D’Amico AV. Time to prostate-specific antigen nadir after androgen suppression therapy for postoperative or postradiation PSA failure and risk of prostate cancer-specific mortality. Urology. 2008;71:136–40.
Kitagawa Y, Ueno S, Izumi K, Mizokami A, Hinotsu S, Akaza H, Namiki M. Nadir prostate-specific antigen (PSA) level and time to PSA nadir following primary androgen deprivation therapy as independent prognostic factors in a Japanese large-scale prospective cohort study (J-CaP). J Cancer Res Clin Oncol. 2014;140:673–9.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Teoh, J.Y.C., Tsu, J.H.L., Yuen, S.K.K. et al. Prognostic Significance of Time to Prostate-Specific Antigen (PSA) Nadir and Its Relationship to Survival Beyond Time to PSA Nadir for Prostate Cancer Patients With Bone Metastases After Primary Androgen Deprivation Therapy. Ann Surg Oncol 22, 1385–1391 (2015). https://doi.org/10.1245/s10434-014-4105-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-014-4105-8