Abstract
Background
Malignant phyllodes tumors of the breast are unusual neoplasms, with an incidence of approximately 500 cases annually in the United States. Published local recurrence rates after margin-negative breast-conserving resections of borderline malignant and malignant phyllodes tumors are unacceptably high, at 24 and 20%, respectively. It is uncertain whether radiotherapy after resection of phyllodes tumors is beneficial.
Methods
We prospectively enrolled patients who were treated with a margin-negative breast-conserving resection of borderline malignant or malignant phyllodes tumors to adjuvant radiotherapy. The primary endpoint was local recurrence.
Results
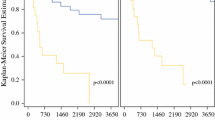
Forty-six women were treated at 30 different institutions. The mean patient age was 49 years (range, 18–76 years). Thirty patients (65%) had malignant phyllodes tumors; the rest were borderline malignant. The mean tumor diameter was 3.7 cm (range, .8–11 cm). Eighteen patients had a negative margin on the first excision. The median size of the negative margin was .35 cm (range, <.1–2 cm). Twenty-eight patients underwent a re-excision because of positive margins in the initial resection. Two patients died of metastatic phyllodes tumor. During a median follow-up of 56 months (range, 12–129 months), none of the 46 patients developed a local recurrence (local recurrence rate, 0%; 95% confidence interval, 0–8).
Conclusions
Margin-negative resection combined with adjuvant radiotherapy is very effective therapy for local control of borderline and malignant phyllodes tumors. The local recurrence rate with adjuvant radiotherapy was significantly less than that observed in reported patients treated with margin-negative resection alone.

Similar content being viewed by others
References
SEER 17 data on phyllodes tumors. Available at: http://seer.cancer.gov/data. Accessed December 6, 2007
Reinfuss M, Mitus J, Duda K, et al. The treatment and prognosis of patients with phyllodes tumor of the breast. Cancer. 1996;77:910–6.
Asoglu O, Ugurlu M, Blanchard K, et al. Risk factors for recurrence and death after primary surgical treatment of malignant phyllodes tumors. Ann Surg Oncol. 2004;11:1011–7.
Belkacemi Y, Bousquet G, Marsiglia H, et al. Phyllodes tumors of the breast. Int J Radiat Oncol Biol Phys. 2008;70:492–500.
Macdonald O, Lee C, Tward J, Chappel C, Gaffney D. Malignant phyllodes tumor of the female breast. Cancer. 2006;107:2127–33.
Barth R. Histologic features predict local recurrence after breast conserving therapy of phyllodes tumors. Breast Cancer Res Treat. 1999;57:291–5.
World Health Organization. Histologic typing of breast tumors. Tumori. 1982;68:181–98.
de Roos W, Kaye P, Dent D. Factors leading to local recurrence or death after surgical resection of phyllodes tumors of the breast. Br J Surg. 1999;86:396–9.
Zissis C, Apostolikas N, Konstantinidou A, Griniatsos J, Vassilopoulos P. The extent of surgery and prognosis of patients with phyllodes tumor of the breast. Breast Cancer Res Treat. 1998;48:205–10.
Holthouse D, Smith P, Naunton-Morgan R, Minchin D. Cystosarcoma phyllodes: the western Australian experience. Aust N Z J Surg. 1999;69:635–8.
Chaney A, Pollack A, Mcneese M, et al. Primary treatment of cystosarcoma phyllodes of the breast. Cancer. 2000;89:1502–11.
Kapiris I, Nasir N, A’hern R, Healy V, Gui G. Outcome and predictive factors of local recurrence and distant metastases following primary surgical treatment of high-grade malignant phyllodes tumors of the breast. Eur J Surg Oncol. 2001;27:723–30.
Kok K, Telesinghe P, Yapp S. Treatment and outcome of cystosarcoma phyllodes in Brunei: a 13 year experience. J R Coll Surg Edinb. 2001;46:198–201.
Fou A, Schnabel F, Hamele-Bena D, et al. Long term outcomes of malignant phyllodes tumors patients: an institutional experience. Am J Surg. 2006;192:492–5.
Abdalla H, Sakr M. Predictive factors of local recurrence and survival following primary surgical treatment of phyllodes tumors of the breast. J Egypt Natl Canc Inst. 2006;18:125–33.
Taira N, Takabatake D, Aogi K, et al. Phyllodes tumor of the breast: stromal overgrowth and histological classification are useful prognosis-predictive factors for local recurrence in patients with a positive surgical margin. Jpn J Clin Oncol. 2007;37:730–6.
Barrio A, Clark B, Goldberg J, et al. Clinicopathologic features and long term outcomes of 293 phyllodes tumors of the breast. Ann Surg Oncol. 2007;14:2961–70.
Lenhard M, Kahlert S, Himsl I, et al. Phyllodes tumor of the breast: clinical follow-up of 33 cases of this rare disease. Eur J Obstet Gynecol Reprod Biol. 2007;138:217–21.
Pietruszka M, Barnes L. Cystosarcoma phyllodes. Cancer. 1978;41:1974–83.
Moffat C, Pinder S, Dixon A, et al. Phyllodes tumors of the breast: a clinicopathological review of 32 cases. Histopathology. 1995;27:205–18.
Pezner R, Schultheiss T, Paz I. Malignant phyllodes tumor of the breast: local control rates with surgery alone. Int J Radiat Oncol Biol Phys. 2008;71:710–13.
Chen W, Cheng S, Tzen C, et al. Surgical treatment of phyllodes tumors of the breast: retrospective review of 172 cases. J Surg Oncol. 2005;91:185–94.
Pandey M, Mathew A, Kattoor J, et al. Malignant phyllodes tumor. Breast J. 2001;7:411–6.
Mangi A, Smith B, Gadd M, et al. Surgical management of phyllodes tumors. Arch Surg. 1999;134:487–93.
Clark M, Collins R, Darby S, et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15 year survival: an overview of the randomized trials. Lancet. 2005;366:2087–106.
Hoover H, Testioreanu A, Ketcham A. Metastatic cystosarcoma phyllodes in an adolescent girl. Ann Surg. 1975;181:279–82.
Burton G, Hart L, Leight G, et al. Cystosarcoma phyllodes, effective treatment with chemotherapy. Cancer. 1989;63:2088–92.
Thomas A, Ashworth B, Blake P. Regression of recurrent cystosarcoma phyllodes tumor after neutron treatment. Br J Radiol. 1984;57:926–9.
Allen R, Nixon D, York M, et al. Successful chemotherapy for cystosarcoma phyllodes. Arch Intern Med. 1985;145:1127–8.
Turalba C, El-Mahdi A, Ladga L. Fatal metastatic cystosarcoma phyllodes. J Surg Oncol. 1986;33:176–81.
Stockdale A, Leader M. Case report: phyllodes tumor of the breast: response to radiotherapy. Clin Radiol. 1987;38:287.
Lindquist K, van Heerden J, Wieland L, et al. Recurrent and metastatic cystosarcoma phyllodes. Am J Surg. 1982;144:341–3.
Reinfuss M, Mitus J, Smolak K, Stelkmach A. Malignant phyllodes tumors of the breast. Eur J Cancer. 1993;29A:1252–6.
Joshi S, Sharma D, Bahadur A, et al. Cystosarcoma phyllodes: our institutional experience. Australas Radiol. 2003;47:434–7.
Kleer C, Giordano T, Braun T, Oberman H. Pathologic, immunohistochemical and molecular features of benign and malignant phyllodes tumors of the breast. Mod Pathol. 2001;14:185–90.
Soumarova R, Seneklova Z, Horova H, et al. Retrospective analysis of 25 women with malignant cystosarcoma phyllodes—treatment results. Arch Gynecol Obstet. 2004;269:278–81.
Rissanen P, Holsti P. A retrospective study of sarcoma of the breast. Oncology. 1968;22:258–68.
Chatson G, Anderson C, Berman M, et al. Phyllodes tumors. Contemp Surg. 1992;40:36–41.
Chaney A, Pollack A, McNeese M, Zagars G. Adjuvant radiation therapy for phyllodes tumors of the breast. Radiat Oncol Investig. 1998;6:264–7.
Tan P, Jayabaskar T, Chuah K, et al. Phyllodes tumors of the breast, the role of pathologic parameters. Am J Clin Pathol. 2005;123:529.
Vicini F, Beitsch P, Quiet C, et al. Three year analysis of treatment efficacy, cosmesis and toxicity in the American Society of Breast Surgeons Mammosite Brachytherapy Registry Trial in patients treated with accelerated partial breast irradiation. Cancer. 2008;112:758–66.
Acknowledgment
We acknowledge the support of medical librarian Cindy Stewart, clinical research associates Megan Patey, Cynara Nayar and Denise Machado from the Clinical Research Office of the Norris Cotton Cancer Center, and Elizabeth Bankert and the staff of the Dartmouth Committee for the Protection of Human Subjects. The authors have no commercial interests in this work. The authors did not receive external funding for this study.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Participating institutions (and number of patients treated): Dartmouth-Hitchcock Medical Center, Lebanon, NH (13); U. of Massachusetts Medical Center, Worcester MA (4); Elliot Hospital, Manchester, NH (3). The following institutions each treated one patient: Jordan Hospital, Plymouth, MA; St. Francis Hospital, Hartford, CT; Central Maine Medical Center, Lewiston, ME; Maine Medical Center, Scarborough, ME; Lahey Clinic, Burlington, MA; Attleboro Surgical Associates, Attleboro, MA; Nashua Regional Cancer Center, Nashua, NH; Moore Regional Hospital, Pinehurst, NC; Hartford Hospital, Hartford, CT; Finger Lakes Radiation Oncology, Clifton Springs, NY; Ohio State University, Columbus, OH; 21st Century Oncology, Fort Myers, FL; Redwood Regional Oncology Center, Santa Rosa, CA; Northeast Regional Radiation Oncology Network, Manchester, CT; Holy Name Hospital, Teaneck, NJ; Exeter Hospital, Exeter, NH; Greater Baltimore Medical Center, Baltimore, MD; Carson Tahoe Regional Healthcare, Carson City, NV; Baptist Hospital East, Louisville, KY; St. Agnes Medical Center, Fresno, CA; Cancer Care of Maine, Bangor, ME; Radiant Care, Lacey, WA; Oncologics Inc., Lafayette, LA; Memorial Medical Center, Springfield, IL; McLeod Health, Florence, SC; Virginia Commonwealth University, Richmond, VA.
Rights and permissions
About this article
Cite this article
Barth, R.J., Wells, W.A., Mitchell, S.E. et al. A Prospective, Multi-Institutional Study of Adjuvant Radiotherapy After Resection of Malignant Phyllodes Tumors. Ann Surg Oncol 16, 2288–2294 (2009). https://doi.org/10.1245/s10434-009-0489-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-009-0489-2