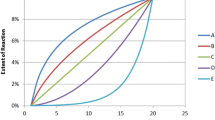
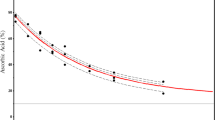
Abstract
An isoconversion paradigm, where times in different temperature and humidity-controlled stability chambers are set to provide a fixed degradant level, is shown to compensate for the complex, non-single order kinetics of solid drug products. A humidity-corrected Arrhenius equation provides reliable estimates for temperature and relative humidity effects on degradation rates. A statistical protocol is employed to determine best fits for chemical stability data, which in turn allows for accurate estimations of shelf life (with appropriate confidence intervals) at any storage condition including inside packaging (based on the moisture vapor transmission rate of the packaging and moisture sorption isotherms of the internal components). These methodologies provide both faster results and far better predictions of chemical stability limited shelf life (expiry) than previously possible. Precise shelf-life estimations are generally determined using a 2-week, product-specific protocol. Once the model for a product is developed, it can play a critical role in providing the product understanding necessary for a quality by design (QbD) filing for product approval and enable rational control strategies to assure product stability. Moreover, this Accelerated Stability Assessment Program (ASAP) enables the coupling of product attributes (e.g., moisture content, packaging options) to allow for flexibility in how control strategies are implemented to provide a balance of cost, speed, and other factors while maintaining adequate stability.





Similar content being viewed by others
REFERENCES
Waterman KC, Adami RC. Accelerated aging: prediction of chemical stability of pharmaceuticals. Int J Pharm. 2005;293:101–25.
Baertschi SW, Jansen PJ. Stress testing: a predictive tool. Drugs and the Pharm Sci. 2005;153:13–49 (Pharmaceutical Stress Testing).
Waterman KC, Carella AJ, Gumkowski MJ, Lukulay P, MacDonald BC, Roy MC, et al. Improved protocol and data analysis for accelerated shelf-life estimation of solid dosage forms. Pharm Res. 2007;24(4):780–90.
Waterman KC. Understanding and predicting pharmaceutical product shelf-life. In: Huynhba K, editor. Handbook of stability testing in pharmaceutical development: regulations, methodologies and best practices. New York: Springer; 2008. p. 115–35. Chapter 6.
Waterman KC. Unpublished results presented at American Association of Pharmaceutical Sciences National Meeting, New Orleans, 2010.
Waterman KC, MacDonald BC. Package selection for moisture protection for solid, oral drug products. J Pharm Sci. 2010;99:4437–52.
Naveršnik K, Bohanec S. Predicting drug hydrolysis based on moisture uptake in various packaging designs. Eur J Pharm Sci. 2008;35:447–56.
Allinson JG, Dansereau RJ, Sakr A. The effects of packaging on the stability of a moisture sensitive compound. Int J Pharm. 2001;221:49–56.
Badawy SIF, Gawronski AJ, Alvarez FJ. Application of sorption-desorption moisture transfer modeling to the study of chemical stability of a moisture sensitive drug product in different packaging configurations. Intern J Pharm. 2001;223(1–2):1–13.
Roskar R, Kmetec V. Evaluation of the moisture sorption behavior of several excipients by BET, GAB and microcalorimetric approaches. Chem Pharm Bull. 2005;53(6):662–5.
Dalton CR, Hancock BC. Processing and storage effects on water vapor sorption by some model pharmaceutical solid dosage formulations. Intern J Pharm. 1997;156:143–51.
Yeshwant YL, Sanzgiri D, Chen Y. A study on moisture isotherms of formulations: the use of polynomial equations to predict the moisture isotherms of tablet products. AAPS PharmSciTech. 2003;4:1–8. Article 59.
Yu LX. Pharmaceutical quality by design: product and process development, understanding, and control. Pharm Res. 2008;25:781–91.
Govindarajan R, Zinchuk A, Hancock B, Shalaev E, Suryanarayanan R. Ionization states in the microenvironment of solid dosage forms: effect of formulation variables and processing. Pharm Res. 2006;23:2454–68.
Waterman KC, Colgan ST. A science-based approach to setting expiry dating for solid drug products. Regulatory Rapporteur. 2008;5:9–14.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Robin Bogner, James Drennen, Mansoor Khan, Cynthia Oksanen, and Gintaras Reklaitis
Rights and permissions
About this article
Cite this article
Waterman, K.C. The Application of the Accelerated Stability Assessment Program (ASAP) to Quality by Design (QbD) for Drug Product Stability. AAPS PharmSciTech 12, 932–937 (2011). https://doi.org/10.1208/s12249-011-9657-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9657-3