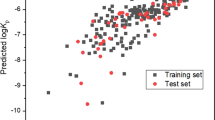
Abstract
The permeability of five benzimidazole derivates with potential cannabinoid activity was determined in two models of membranes, parallel artificial membrane permeability assay (PAMPA) and skin, in order to study the relationship of the physicochemical properties of the molecules and characteristics of the membranes with the permeability defined by the Biopharmaceutics Classification System. It was established that the PAMPA intestinal absorption method is a good predictor for classifying these molecules as very permeable, independent of their thermodynamic solubility, if and only if these have a Log P oct value <3.0. In contrast, transdermal permeability is conditioned on the solubility of the molecule so that it can only serve as a model for classifying the permeability of molecules that possess high solubility (class I: high solubility, high permeability; class III: high solubility, low permeability).



Similar content being viewed by others
References
Amidon GL, Lennernas H, Shah VP, John RC. A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo biovailability. Pharm Res. 1995;12(3):413–20.
Dressmann JB, Amidon GL, Reppas C, Shah VP. Dissolution testing as a prognostic tool for oral drug absorption: immediate release dosage forms. Pharm Res. 1998;15(1):11–22.
Dahan A, Jonathan M, Miller JM, Amidon GL. Prediction of solubility and permeability class membership: provisional BCS classification of the world’s top oral drugs. AAPS J. 2009;11(4):740–6.
Kasim NA, Whitehouse M, Ramachandran C, Bermejo M, Lennernais H, Hussain AS, et al. Molecular properties of WHO essential drugs and provisional biopharmaceutical classification. Mol Pharm. 2004;1(1):85–96.
Avendaño C. Introducción a la Química Farmacéutica. 2ath ed. Spain: McGraw Hill Interamericana; 2001.
Graham LP. An introduction to medicinal chemistry. 3ath ed. Oxford: Oxford University Press; 2005.
Hansch C, Leo A, Mekapati SC, Kurup A. QSAR and ADME. Bioorg Med Chem. 2004;12:391–40.
Costas G, Tsantili-Kakoulidou A. Alternative measures of lipophilicity: from octanol–water partitioning to IAM retention. J Pharm Sci. 2008;97(8):2984–3004.
Poole SK, Poole CF. Separation methods for estimating octanol–water partition coefficients. J Chromatogr B. 2003;797:3–19.
Liu R, Zhou D. Using molecular fingerprint as descriptors in the QSPR study of lipophilicity. J Chem Inf. 2008;48:542–9.
Fujikawa M, Ano K, Nakao K, Shimizu R, Akamatsu M. Relationships between structure and high throughput screening permeability of diverse drugs with artificial membranes: application to prediction of Caco-2 cell permeability. Bioorg Med Chem. 2005;13:4721–32.
Fujikawa M, Nakao K, Shimizu R, Akamatsu M. QSAR study on permeability of hydrophobic compounds with artificial membranes. Bioorg Med Chem. 2007;15:3756–67.
Kansy M, Senner F, Gubernator K. Physicochemical high throughput screening: parallel artificial membrane permeation assay in the description of passive absorption processes. J Med Chem. 1998;41(7):1007–10.
Sugano K, Nabuchi Y, Machida M, Aso Y. Prediction of human intestinal permeability using artificial membrane permeability. Int J Pharm. 2003;257:245–51.
Wohnsland F, Faller B. Throughput permeability ph profile and high throughput alkane/water log P with artificial membranes. J Med Chem. 2001;44:923–30.
Avdeef A, Bendels S, Di L, Faller B, Kansy M, Sugano K, et al. PAMPA—critical factors for better predictions of absorption. J Pharm Sci. 2007;96(11):2893–909.
Balimane PV, Pace E, Chong S, Zhu M, Jemal M, Pelt CK. A novel high-throughput automated chip-based nanoelectrospray tandem mass spectrometric method for PAMPA sample analysis. J Pharm Biomed Anal. 2005;39(1–2):8–16.
Bermejo M, Avdeef A, Ruiz A, Nalda R, Ruell JA, Tsinman O, et al. PAMPA—a drug absorption in vitro model 7. Comparing rat in situ, Caco-2, and PAMPA permeability of fluoroquinolones. Eur J Pharm Sci. 2004;21:429–41.
Corti G, Maestrelli F, Cirri M, Zerrouk N, Mura P. Development and evaluation of an in vitro method for prediction of human drug absorption II. Demonstration of the method suitability. Eur J Pharm Sci. 2006;27:354–62.
Kansy M, Avdeef A, Fischer H. Advances in screening for membrane permeability: high-resolution PAMPA for medicinal chemists. Drug Discov Today Technol. 2004;1(4):349–55.
Kerns EH, Di L, Petusky S, Farris M, Ley R, Jupp P. Combined application of parallel artificial membrane permeability assay and Caco-2 permeability assays in drug discovery. J Pharm Sci. 2004;93(6):1440–53.
Yamagami C, Kawase K, Iwaki K. Hydrophobicity parameters determined by reverse-phase liquid chromatography. XV: optimal conditions for prediction of log P oct by using RP-HPLC procedures. Chem Pharm Bull. 2002;50(12):1578–83.
Dias NC, Nawas MI, Poole CF. Evaluation of a reversed-phase column (Supelcosil LC-ABZ) under isocratic and gradient elution conditions for estimating octanol–water partition coefficients. Analyst. 2003;128:427–33.
Alvarez-Figueroa MJ, Pessoa-Mahana CD, González-Bustamante DA. Influence of lipophilia and of the vehicle used in the transdermal absorption of novel benzimidazole compounds with possible anti-HIV activity. Pharm Dev Technol. 2008;13:127–33.
Alvarez-Figueroa MJ, Araya-Silva I, Díaz-Tobar C. Iontophoretic transdermal delivery of haloperidol. Pharm Dev Technol. 2006;11:371–5.
Alvarez-Figueroa MJ, Blanco-Méndez J. Transdermal delivery of methotrexate: iontophoretic delivery from hydrogels and passive delivery from microemulsions. Int J Pharmacogn. 2001;215:57–65.
Bronaugh RL, Stewart RF. Methods for in vitro percutaneous absorption studies III: hydrophobic compounds. J Pharm Sci. 1984;73(9):1255–8.
Kerns EH, S Petusky, Farris M, Ley R, Jupp P. Combined application of parallel artificial membrane permeability assay and Caco-2 permeability assays in drug discovery. J Pharm Sci. 2004;93(6):1440–53.
Ruell JA, Tsinman KL, Avdeef A. PAMPA—a drug absorption in vitro model 5. Unstirred water layer in iso-pH mapping assays and pKaflux-optimized design (pOD-PAMPA). Eur J Pharm Sci. 2003;20:393–402.
Fischer H, Kansy M, Avdeef A, Senner F. Permeation of permanently positive charged molecules through artificial membranes—influence of physico-chemical properties. Eur J Pharm Sci. 2007;31:32–42.
Brain-Isasi S, Quezada C, Pessoa H, Morello A, Kogan MJ, Álvarez-Lueje A. Determination and characterization of new benzimidazoles with activity against Trypanosoma cruzi by UV spectroscopy and HPLC. Bioorg Med Chem. 2008;16:7622–30.
Zheng XS, Duan CZ, Xiao ZD, Yao BA. Transdermal delivery of praziquantel: effects of solvents on permeation across rabbit skin. Biol Pharm Bull. 2008;31(5):1045–8.
Subedi RK, Oh SY, Chun M-K, Choi H-K. Recent advances in transdermal drug delivery. Arch Pharm Res. 2010;33(3):339–51.
Barry B. Farmacia, la ciencia del diseño de las formas farmacéuticas. Chapter 33: Administración de fármacos por vía transdérmica. M.E. Aulton (ed). Elsevier España S.A., 2004 second edition. p. 499–533.
Guy RH. Transdermal drug delivery. Hand Exp Pharmacol. 2010;197:399–410.
Barry BW. Is the transdermal drug delivery research still important today? DDT. 2001;6(19):967–71.
Acknowledgments
This work was supported by the Faculty of Chemistry of the Pontificia Universidad Católica de Chile and FONDECYT (grant 1100493).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alvarez-Figueroa, M.J., Pessoa-Mahana, C.D., Palavecino-González, M.E. et al. Evaluation of the Membrane Permeability (PAMPA and Skin) of Benzimidazoles with Potential Cannabinoid Activity and their Relation with the Biopharmaceutics Classification System (BCS). AAPS PharmSciTech 12, 573–578 (2011). https://doi.org/10.1208/s12249-011-9622-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-011-9622-1