ABSTRACT
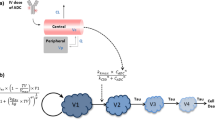
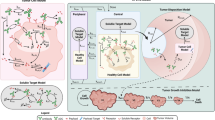
A mechanism-based pharmacokinetic/pharmacodynamic (PK/PD) model was used for preclinical to clinical translation of inotuzumab ozogamicin, a CD22-targeting antibody-drug conjugate (ADC) for B cell malignancies including non-Hodgkin’s lymphoma (NHL) and acute lymphocytic leukemia (ALL). Preclinical data was integrated in a PK/PD model which included (1) a plasma PK model characterizing disposition and clearance of inotuzumab ozogamicin and its released payload N-Ac-γ-calicheamicin DMH, (2) a tumor disposition model describing ADC diffusion into the tumor extracellular environment, (3) a cellular model describing inotuzumab ozogamicin binding to CD22, internalization, intracellular N-Ac-γ-calicheamicin DMH release, binding to DNA, or efflux from the tumor cell, and (4) tumor growth and inhibition in mouse xenograft models. The preclinical model was translated to the clinic by incorporating human PK for inotuzumab ozogamicin and clinically relevant tumor volumes, tumor growth rates, and values for CD22 expression in the relevant patient populations. The resulting stochastic models predicted progression-free survival (PFS) rates for inotuzumab ozogamicin in patients comparable to the observed clinical results. The model suggested that a fractionated dosing regimen is superior to a conventional dosing regimen for ALL but not for NHL. Simulations indicated that tumor growth is a highly sensitive parameter and predictive of successful outcome. Inotuzumab ozogamicin PK and N-Ac-γ-calicheamicin DMH efflux are also sensitive parameters and would be considered more useful predictors of outcome than CD22 receptor expression. In summary, a multiscale, mechanism-based model has been developed for inotuzumab ozogamicin, which can integrate preclinical biomeasures and PK/PD data to predict clinical response.







Similar content being viewed by others
REFERENCES
Webb S. Pharma interest surges in antibody drug conjugates. Nat Biotechnol. 2011;29(4):297–8.
Zolot RS, Basu S, Million RP. Antibody-drug conjugates. Nat Rev Drug Discov. 2013;12(4):259–60.
Sohayla Rostami IQ, Robert Sikorski. The clinical landscape of antibody-drug conjugates http://adcreview.com/articles/doi-10-14229jadc-2014-8-1-001/: InPress Media Group, LLC; 2014.
Mack F, Ritchie M, Sapra P. The next generation of antibody drug conjugates. Semin Oncol. 2014;41(5):637–52.
ImmunoGen, Inc. Announces discontinuation of IMGN901 study in small-cell lung cancer (SCLC) http://investor.immunogen.com/releasedetail.cfm?ReleaseID=804315: Immunogen, Inc.; 2013.
Williams R. Discontinued in 2013: oncology drugs. Expert Opin Investigat Drugs. 2015;24(1):95–110.
Panowksi S, Bhakta S, Raab H, Polakis P, Junutula JR. Site-specific antibody drug conjugates for cancer therapy. MAbs. 2014;6(1):34–45.
Maderna A, Doroski M, Subramanyam C, Porte A, Leverett CA, Vetelino BC, et al. Discovery of cytotoxic dolastatin 10 analogues with N-terminal modifications. J Med Chem. 2014;57(24):10527–43.
Agoram BM, Martin SW, van der Graaf PH. The role of mechanism-based pharmacokinetic-pharmacodynamic (PK-PD) modelling in translational research of biologics. Drug Discov Today. 2007;12(23–24):1018–24.
Shah DK, Haddish-Berhane N, Betts A. Bench to bedside translation of antibody drug conjugates using a multiscale mechanistic PK/PD model: a case study with brentuximab-vedotin. J Pharmacokinet Pharmacodyn. 2012;39(6):643–59.
Jumbe NL, Xin Y, Leipold DD, Crocker L, Dugger D, Mai E, et al. Modeling the efficacy of trastuzumab-DM1, an antibody drug conjugate, in mice. J Pharmacokinet Pharmacodyn. 2010;37(3):221–42.
Haddish-Berhane N, Shah DK, Ma D, Leal M, Gerber HP, Sapra P, et al. On translation of antibody drug conjugates efficacy from mouse experimental tumors to the clinic: a PK/PD approach. J Pharmacokinet Pharmacodyn. 2013;40(5):557–71.
Sadekar S, Figueroa I, Tabrizi M. Antibody drug conjugates: application of quantitative pharmacology in modality design and target selection. AAPS J. 2015;17(4):828–36.
Shah DK, King LE, Han X, Wentland JA, Zhang Y, Lucas J, et al. A priori prediction of tumor payload concentrations: preclinical case study with an auristatin-based anti-5T4 antibody-drug conjugate. AAPS J. 2014;16(3):452–63.
DiJoseph JF, Armellino DC, Boghaert ER, Khandke K, Dougher MM, Sridharan L, et al. Antibody-targeted chemotherapy with CMC-544: a CD22-targeted immunoconjugate of calicheamicin for the treatment of B-lymphoid malignancies. Blood. 2004;103(5):1807–14.
Dijoseph JF, Dougher MM, Armellino DC, Evans DY, Damle NK. Therapeutic potential of CD22-specific antibody-targeted chemotherapy using inotuzumab ozogamicin (CMC-544) for the treatment of acute lymphoblastic leukemia. Leukemia. 2007;21(11):2240–5.
Pfizer discontinues phase 3 study of inotuzumab ozogamicin in relapsed or refractory aggressive non-Hodgkin lymphoma (NHL) due to futility http://press.pfizer.com/press-release/pfizer-discontinues-phase-3-study-inotuzumab-ozogamicin-relapsed-or-refractory-aggress: Pfizer Inc.; 2013 [May 20th 2013].
Wang L, Abbasi F, Gaigalas AK, Hoffman RA, Flagler D, Marti GE. Discrepancy in measuring CD4 expression on T-lymphocytes using fluorescein conjugates in comparison with unimolar CD4-phycoerythrin conjugates. Cytometry B Clin Cytom. 2007;72(6):442–9.
Sherree Friend TG, Hall B, Venkatachalam V, Morrissey P. Intracellular localization and trafficking using the ImageStream® imaging flow cytometer https://www.amnis.com/documents/posters/07-003-1-pos.pdf: Amnis Corporation.
Poulsen TR, Jensen A, Haurum JS, Andersen PS. Limits for antibody affinity maturation and repertoire diversification in hypervaccinated humans. J Immunol. 2011;187(8):4229–35.
Tianhu Li ZZ, Virginia A, Estevez A, Baldenius KU, Nicolaou KC, Joyce GF. Carbohydrate-minor groove interactions in the binding of Calicheamicin g1 to duplex DNA. J Am Chem Soc. 1994;116:3709–15.
Polson AG, Williams M, Gray AM, Fuji RN, Poon KA, McBride J, et al. Anti-CD22-MCC-DM1: an antibody-drug conjugate with a stable linker for the treatment of non-Hodgkin’s lymphoma. Leukemia. 2010;24(9):1566–73.
Carnahan J, Wang P, Kendall R, Chen C, Hu S, Boone T, et al. Epratuzumab, a humanized monoclonal antibody targeting CD22: characterization of in vitro properties. Clin Cancer Res : Off J Am Assoc Cancer Res. 2003;9(10 Pt 2):3982S–90S.
Schmidt MM, Wittrup KD. A modeling analysis of the effects of molecular size and binding affinity on tumor targeting. Mol Cancer Ther. 2009;8(10):2861–71.
Simeoni M, Magni P, Cammia C, De Nicolao G, Croci V, Pesenti E, et al. Predictive pharmacokinetic-pharmacodynamic modeling of tumor growth kinetics in xenograft models after administration of anticancer agents. Cancer Res. 2004;64(3):1094–101.
Ogura M, Tobinai K, Hatake K, Uchida T, Kasai M, Oyama T, et al. Phase I study of inotuzumab ozogamicin (CMC-544) in Japanese patients with follicular lymphoma pretreated with rituximab-based therapy. Cancer Sci. 2010;101(8):1840–5.
Brons PP, Raemaekers JM, Bogman MJ, van Erp PE, Boezeman JB, Pennings AH, et al. Cell cycle kinetics in malignant lymphoma studied with in vivo iododeoxyuridine administration, nuclear Ki-67 staining, and flow cytometry. Blood. 1992;80(9):2336–43.
Frei 3rd E, Freireich EJ. Progress and perspectives in the chemotherapy of acute leukemia. Adv Chemotherap. 1965;2:269–98.
Holland JF. Clinical studies of unmaintained remissions in acute lymphocytic leukemia. the proliferation and spread of neoplastic cells. Baltimore: The University of Texas M. D. Anderson Hospital and Tumor Institute at Houston: The Williams& Wilkins Co; 1968. p. 453–62.
Tolo Diebkile A, Kouakou B, N’Dhatz E, Nanho CD, Meite N, Ayemou R, et al. Characteristics and results of the management of diffuse large B-cell lymphomas: the experience of Cote d’Ivoire. Adv Hematol. 2012;2012:945138.
Gregory WM, Richards MA, Slevin ML, Souhami RL. A mathematical model relating response durations to amount of subclinical resistant disease. Cancer Res. 1991;51(4):1210–6.
Armitage JO, Weisenburger DD. New approach to classifying non-Hodgkin’s lymphomas: clinical features of the major histologic subtypes. Non-Hodgkin’s lymphoma classification project. J Clin Oncol : Off J Am Soc Clin Oncol. 1998;16(8):2780–95.
Skipper HE, Perry S. Kinetics of normal and leukemic leukocyte populations and relevance to chemotherapy. Cancer Res. 1970;30(6):1883–97.
Thurber GM, Schmidt MM, Wittrup KD. Antibody tumor penetration: transport opposed by systemic and antigen-mediated clearance. Adv Drug Deliv Rev. 2008;60(12):1421–34.
Thurber GM, Schmidt MM, Wittrup KD. Factors determining antibody distribution in tumors. Trends Pharmacol Sci. 2008;29(2):57–61.
Advani A, Coiffier B, Czuczman MS, Dreyling M, Foran J, Gine E, et al. Safety, pharmacokinetics, and preliminary clinical activity of inotuzumab ozogamicin, a novel immunoconjugate for the treatment of B-cell non-Hodgkin’s lymphoma: results of a phase I study. J Clin Oncol : Off J Am Soc Clin Oncol. 2010;28(12):2085–93.
Zhao Y, Kosorok MR, Zeng D. Reinforcement learning design for cancer clinical trials. Stat Med. 2009;28(26):3294–315.
Kantarjian H, Thomas D, Jorgensen J, Kebriaei P, Jabbour E, Rytting M, et al. Results of inotuzumab ozogamicin, a CD22 monoclonal antibody, in refractory and relapsed acute lymphocytic leukemia. Cancer. 2013;119(15):2728–36.
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ, et al. Revised response criteria for malignant lymphoma. J Clin Oncol : Off J Am Soc Clin Oncol. 2007;25(5):579–86.
DeAngelo DJMS, Martinelli G, Kantarjian H, Liedtke M, Stock W, Goekbuget NKW, et al. Efficacy and safety of inotuzumab ozogamicin (INO) vs standard of care (SOC) in salvage 1 or 2 patients with acute lymphoblastic leukemia (ALL): an ongoing global phase 3 study. Vienna: European Hematology Association; 2015.
Peterson JK, Houghton PJ. Integrating pharmacology and in vivo cancer models in preclinical and clinical drug development. Eur J Cancer. 2004;40(6):837–44.
Kelland LR. Of mice and men: values and liabilities of the athymic nude mouse model in anticancer drug development. Eur J Cancer. 2004;40(6):827–36.
Wong H, Choo EF, Alicke B, Ding X, La H, McNamara E, et al. Antitumor activity of targeted and cytotoxic agents in murine subcutaneous tumor models correlates with clinical response. Clin Cancer Res : Off J Am Assoc Cancer Res. 2012;18(14):3846–55.
Kamb A. What’s wrong with our cancer models? Nat Rev Drug Discov. 2005;4(2):161–5.
Rocchetti M, Simeoni M, Pesenti E, De Nicolao G, Poggesi I. Predicting the active doses in humans from animal studies: a novel approach in oncology. Eur J Cancer. 2007;43(12):1862–8.
Ducry L, Stump B. Antibody-drug conjugates: linking cytotoxic payloads to monoclonal antibodies. Bioconjug Chem. 2010;21(1):5–13.
Agoram BM, Demin O. Integration not isolation: arguing the case for quantitative and systems pharmacology in drug discovery and development. Drug Discov Today. 2011;16(23-24):1031–6.
Shor B, Gerber HP, Sapra P. Preclinical and clinical development of inotuzumab-ozogamicin in hematological malignancies. Mol Immunol. 2015;67(2 Pt A):107–16.
Takeshita A, Shinjo K, Yamakage N, Ono T, Hirano I, Matsui H, et al. CMC-544 (inotuzumab ozogamicin) shows less effect on multidrug resistant cells: analyses in cell lines and cells from patients with B-cell chronic lymphocytic leukaemia and lymphoma. Br J Haematol. 2009;146(1):34–43.
Shah DK, Barletta F, Betts A, Hansel S. Key bioanalytical measurements for antibody-drug conjugate development: PK/PD modelers’ perspective. Bioanalysis. 2013;5(9):989–92.
Okeley NM, Miyamoto JB, Zhang X, Sanderson RJ, Benjamin DR, Sievers EL, et al. Intracellular activation of SGN-35, a potent anti-CD30 antibody-drug conjugate. Clin Cancer Res : Off J Am Assoc Cancer Res. 2010;16(3):888–97.
Thurber GM, Dane WK. A mechanistic compartmental model for total antibody uptake in tumors. J Theor Biol. 2012;314:57–68.
Minchinton AI, Tannock IF. Drug penetration in solid tumours. Nat Rev Cancer. 2006;6(8):583–92.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editor: Dhaval K. Shah
Rights and permissions
About this article
Cite this article
Betts, A.M., Haddish-Berhane, N., Tolsma, J. et al. Preclinical to Clinical Translation of Antibody-Drug Conjugates Using PK/PD Modeling: a Retrospective Analysis of Inotuzumab Ozogamicin. AAPS J 18, 1101–1116 (2016). https://doi.org/10.1208/s12248-016-9929-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12248-016-9929-7