Abstract
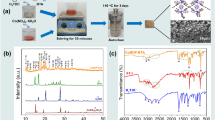
In this study, corrosion resistance was developed to ensure that AZ91 Mg alloy remains stable in the body for longer periods. For this purpose, tantalum oxide and tantalum oxide with diethanolamine coatings were made by sol–gel method on AZ91 magnesium alloy surfaces. The structural analyses of tantalum oxide and tantalum oxide with diethanolamine obtained by sol–gel method were carried out. By using the dip coating method, coatings with different concentrations of solutions and different dipping numbers were applied on AZ91 Mg alloy surfaces. Thus, the effects of dipping numbers and chemical content of the solution on the coating thicknesses, surface roughnesses, morphologies and corrosion resistances of coating layers were examined. As a result, it was determined that the coating thicknesses increased with the dipping number. The low number of dipping could not provide sufficient protective layer formation on the surface. Better results were obtained in samples with a high dipping numbers. It was observed that needle-like structures were formed in the solution using diethanolamine. As the amount of diethanolamine increased, a more homogeneous and dense coating layer was observed. Accordingly, an increase in corrosion resistance has been observed. It was determined that the corrosion rate of AZ91 Mg alloy has decreased by 86% with increasing dipping number and diethanolamine amount.











Similar content being viewed by others
REFERENCES
Hattab, M., Ben Hassen, S., Cecilia-Buenestado, J.A., Rodriguez-Castellon, E., and Ben Amor, Y., Prot. Met. Phys. Chem. Surf., 2021, vol. 57, p. 168.
Sedighi, O., Shabestari, S.G., and Yavari, F., Thermochim. Acta, 2018, vol. 667, p 165.
Zheng, T., Hu, Y., Pan F., Zhang, Y., and Tang A., J. Magnesium Alloys, 2019, vol. 7, p. 193.
Fernandez, J., Ouardi, Y.E., Bonaste, J., Molina, J.M., and Cases, F., Corros. Sci., 2019, vol. 152, p. 75.
Daroonparvar, M., Yajid, M.A., Bakhsheshi-Rad, H.R., Kumar, P., Kay, C.M., and Kalvala, P.R., Prot. Met. Phys. Chem. Surf., 2020, vol. 56, no. 5, p. 1039.
Haghighi O., Amini, K., and Gharavi, F., Prot. Met. Phys. Chem. Surf., 2020, vol. 56, p. 164.
Bakhsheshi-Rad, H.R., Ismail, A.F., Aziz, M., Hadisi, Z., Omidi, M., and Chen, X., Ceram. Int., 2019, vol. 45, p. 11883.
Jin, W., Wang, G., Lin, Z., Feng, H., Li, W., Peng, X., Qasim, A.M., and Chu, P.K., Corros. Sci., 2017, vol. 114, p. 45.
Schulze, V., Bleicher, F., Groche, P., Guo, Y.B., and Pyun, Y.S., CIRP Ann. - Manuf. Technol., 2016, vol. 65, p. 809.
Liu, Y.-H., Cheng, W.-L., Zhang, Y., Niu, X.-F., Wang, H.-X., and Wang, L.-F., J. Alloys Compd., 2020, vol. 815, p. 152414.
Mingo, B., Mohedano, M., Blawert, C., Olmo, R., Hort, N., and Arrabal, R., J. Alloys Compd., 2019, vol. 811, p. 151992.
Oleinik, S.V., Rudnev, V.S., Kuzenkov, Yu.A., Yarovaya, T.P., Trubetskaya, L.F., and Nedozorov, P.M., Prot. Met. Phys. Chem. Surf., 2019, vol. 55, p. 1324.
Ly, X.N. and Yang, S., Surf. Coat. Technol., 2019, vol. 358, p. 331.
Xiong, Y., Hu, Q., Song, R., and Hu, X., Mater. Sci. Eng., C, 2017, vol. 75, p. 1299.
Singh, S., Singh, G., and Bala, N., Mater. Chem. Phys., 2019, vol. 237, p. 121884.
Xu, G., Shen, X., Hu, Y., Ma, P., and Cai, K., Surf. Coat. Technol., 2015, vol. 272, p. 58.
Horandghadim, N., Khalil-Allafi, J., and Urgen, M., Mater. Sci. Eng., C, 2019, vol. 102, p. 683.
Rudnev, V.S., Yarovaya, T.P., Medkov, M.A., Nedozorov, P.M., Kilin, K.N., Lukiyanchuk, I.V., and Ustinov, A.Yu, Prot. Met. Phys. Chem. Surf., 2015, vol. 51, p. 968.
Hussein, A.H., Gepreel, M.A.H., Gouda, M.K., Hefnawy, A.M., and Kandil, S.H., Mater. Sci. Eng., C, 2016, vol. 61, p. 574.
Stenlund, P., Omar, O., Brohede, U., Norgren, S., Norlindh, B., Johansson, A., Lausmaa, J., Thomsen, P., and Palmquist, A., Acta Biomater., 2015, vol. 20, p. 165.
Biesiekierski, A., Lin, J., Li, Y., Ping, D., Yamabe-Mitarai, Y., and Wen, C., Acta Biomater., 2016, vol. 32, p. 336.
Liu, J., Chang, L., Liu, H., Li, Y., Yang, H., and Ruan, J., Mater. Sci. Eng., C, 2017, vol. 71, p. 512.
Sun, Y.-S., Chang, J.-H., and Huang, H.-H., Thin Solid Films, 2013, vol. 528, p 130.
Chang, Y.-Y., Huang, H.-L., Chen, H.-J., Lai, C.-H., and Wen, C.-Y., Surf. Coat. Technol., 2014, vol. 259, p. 193.
Hammami, O., Dhouibi, L., Berçot, P., Rezrazi, E.M., and Triki, E., Can. J. Chem. Eng., 2012, vol. 91, p. 19.
Zhou, J., Zhao, G., Ren, X., Song, B., and Han, G., J. sol–gel Sci. Technol., 2011, vol. 58, p. 148.
Kahraman, S., Çetinkaya, S., Çetinkara, H.A., and Güder, H.S., Mater. Res. Bull., 2014, vol. 50, p. 165.
Liu, J., Xu, X., and Lu, X.P., ACS Biomater. Sci. Eng., 2016, vol. 2, p. 579.
Pasinli, A., Yuksel, M., Celik, E., Sener, S., and Tas, A.C., Acta Biomater., 2010, vol. 6, p. 2282.
Al-Arjan, W.S., Hector, A.L., and Levason, W., J. sol–gel Sci. Technol., 2016, vol. 79, p. 550.
Brandes, E.A. and Brook, G.B., Smithells Light Metals Handbook, Oxford: Butterworth-Heinemann, 1998.
Kandil, A., J. Eng. Sci., 2012, vol. 40, p. 255.
ASM Handbook, vol. 2: Properties and Selection: Nonferrous Alloys and Special-Purpose Materials, New York: ASM Int., 1990.
Basu, B.J., Hariprakash, V., Aruna, S.T., Lakshmi, R.V., Manasa, J., and Shruthi, B.S., J. Sol–Gel Sci. Technol., 2010, vol. 56, p. 278.
Oshida, Y., Bioscience and Bioengineering of Titanium Materials, Indianapolis: Elsevier, 2017.
Wolf, M.J., Roitsch, S., Mayer, J., Nijmeijer, A., and Bouwmeester, H.J.M., Thin Solid Films, 2013, vol. 527, p. 354.
Devan, R.S., Ho, W.-D., Wu, S.Y., and Ma, Y.R., J. Appl. Crystallogr., 2010, vol. 43, p. 498.
Fujisawa, S. and Yonezu, A., Proc. Int. Congress “Recent Advances in Structural Integrity Analysis” (APCF/SIF-2014), Sydney, 2014, p. 422.
Tan, Q., Atrens, A., Mo, N., and Zhang, M.-X., Corros. Sci., 2016, vol. 112, p. 734.
Rojasi, P.N., Rodil, S.E., Muhl, S., Ramirez, G., and Arzate, H., Mater. Res. Soc. Symp. Proc., 2009, vol. 1244, p. 1.
Frankel, G.S., J. Electrochem. Soc., 1998, vol. 145, p. 2186.
Tahmasebifar, A., Kayhan, S.M., Evis, Z., Tezcaner, A., Çinici, H., and Koç, M., J. Alloys Compd., 2016, vol. 687, p. 906.
Callister, W.D. and Rethwisch, D.G., Materials Science and Engineering, New York: Wiley, 2020.
ACKNOWLEDGMENTS
This research has been done as a part of the TÜBİTAK 1001 project titled “Production of AZ91 Magnesium Alloy Coated with Layered and Hybrid Ta2O5/Hydroxyapatite for Use in Permanent Orthopedic Applications and Determination of in-vitro and in-vivo Performance (Grant number: 118M364)”. The authors would like to thank The Scientific and Technical Research Council of Turkiye (TUBITAK) for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Gül, C., Albayrak, S., Çinici, H. et al. Improvement in Corrosion Resistance of Tantalum Oxide and Tantalum Oxide with Diethanolamine Sol–Gel Coated Magnesium Alloys. Prot Met Phys Chem Surf 58, 603–614 (2022). https://doi.org/10.1134/S2070205122030108
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205122030108