Abstract
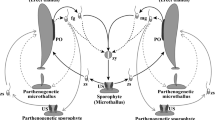
Zoospores of Laminaria at the stage of zoospore germination fixed to glass slides were irradiated by γ-rays in doses of 50, 100, or 250 Gy; or treated with colchicine at a concentration of 4 × 10−5% for 5 days. The cultivation was conducted in vessels with seawater at a temperature of 12°N and illumination of 4000 lux for one month. Once a day, from day 22 to day 30, the temperature was reduced to 0°N for 12 h. As a result, in experimental samples gametophytes appeared that did not form gametangia; these appeared by the third day of cultivation, as plaques up to 2 cm in diameter (1–2 plaques per slide). In the same culture we found structures (1–2 per slide) consisting of strictly radially arranged rows of somatic cells attached to the slides. Later, these disks transformed into cones up to 0.5 cm in diameter. We recorded the development of a single-layered sporophyte of Laminaria arising from the center of such a cone.
Similar content being viewed by others
References
Buyankina, S.K., Biotechnology of Artificial Cultivation of Kelp in Primorye, Tr. VNIRO, 1977, vol. 74, pp. 52–56.
Vasser, S.P., Kondrat’eva, N.V., Masyuk, N.P., et al., Tsikly razvitiya burykh vodoroslei. Vodorosli. Spravochnik (Developmental Cycles of Brown Algae. Algae. The Reference Book), Kiev: Naukova Dumka, 1989.
Kruglova, N.N. and Abramov, S.N., Phenomenon of the Development of Fertilization Tubes in Callus in vitro, Fiziologiya i biokhimiya kul’turnykh rastenii, 2004, vol. 36, no. 1, pp. 280–285.
Krupnova, T.N., Razmnozhenie laminarii yaponskoi Laminaria japonica Aresch.—ob’ekta marikul’tury (Reproduction of Japanese Kelp, Laminaria japonica, an Object of Mariculture), Autoref. Cand. Diss. (Biol.), Vladivostok, 1984.
Mal’tsev, V.N., Studies of the Gametophyte of Laminaria japonica in Connection with Cultivation of This Species, Tr. VNIRO, 1979, vol. 138, pp. 46–51.
Rodova, N.A. and Voskoboinikov, G.M., Cell Cultures in Macroalgae, Biol. Morya, 1993, vol. 19, no. 1, pp. 3–18.
Kawashima, S., Cultivation of the Brown Alga, Laminaria “Kombu,” Seaweed Cultivation and Marine Ranching, JICA, 1993, pp. 25–40.
Lee, T.F., Aposporous Gametophyte Formation in Stipe Explants from Laminaria saccharina (Phaeophyta), Bot. Mar., 1985, vol. 28, no. 2, pp. 179–185.
Provasoli, L., Effect of Plant Hormones on Ulva, Biol. Bull., 1958, vol. 114, pp. 375–384.
Qi, H., Rorrer, K.A., Jiang, Zh.D., and Rorrer, Gr.L., Kinetics of Non-Photosynthetic Callus Production from the Brown Alga Laminaria setchellii (Laminariales), Phycol. Res., 1995, vol. 43, no. 3, pp. 179–182.
Saga, N., Motomura, T., and Sakai, Y., Induction of Callus from the Marine Brown Alga Dictyosiphon foeniculaceus, Plant Cell Physiol., 1982, vol. 23, no. 4, pp. 727–730.
Wang, A., Shuai, L., and Duan, D., Filament Induction in Halymenia sinensis (Halymeniaceae, Rhodophyta), Bot. Mar., 2006, vol. 49, no. 4, pp. 352–354.
Yokoya, N.S., Apical Callus Formation and Plant Regeneration Controlled by Plant Growth Regulators on Axenic Culture of the Red Alga Gracilariopsis tenuifrons (Gracilariales, Rhodophyta), Phycol. Res., 2000, vol. 48, no. 3, pp. 133–142.
Zar, J.H., Biostatistical Analysis, New Jersey: Prentice Hall, 1996.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © Yu.A. Ivanovskii, V.N. Kulepanov, 2008, published in Biologiya Morya.
Rights and permissions
About this article
Cite this article
Ivanovskii, Y.A., Kulepanov, V.N. Initiation of gametophytic callus and somatic embryogenesis in Laminaria japonica (Phaeophyta) exposed to mutagenic agents. Russ J Mar Biol 34, 325–328 (2008). https://doi.org/10.1134/S106307400805009X
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106307400805009X