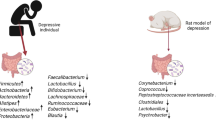
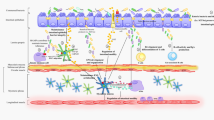
Abstract
Functional interaction of the gastrointestinal tract (GI) and the central nervous system (CNS) is due to various relationships, which includes autonomic and enteral nervous systems as well as the immune and neuroendocrine systems. The microbiota of the macroorganism plays the central role in this interaction. Microbiota produces hundreds of biologically active substances that have a neurochemical effects through neuroendocrine, immune, and metabolic pathways. The microbiota also synthesizes and releases products (neurotoxins, neurotransmitters, lipopolysaccharides, amyloids, etc.) that can negatively affect the neurochemistry of the CNS, stimulating the development of amyloidosis, synucleinopathies, and tauopathies, thereby promoting the development and/or progression of neurodegenerative diseases. Under the influence of external and internal factors, human microbiota can be changed and the symbionts/pathogens ratio is also changed. The permeability of intestinal and blood-brain barrier varies. Metabolites produced by the altered microflora are able to enter the bloodstream and possibly into the CNS, thereby disrupting its functioning. Infections can play a significant role and even act as a cofactor in the induction of neurodegenerative diseases. Disturbance of the functions of the GI can precede long before the neurodegenerative processes. Early diagnosis, detection, monitoring, and treatment of negative gastrointestinal symptoms, including normalization of the microbiota, can lead to a significant improvement in the quality of life of patients with neurodegenerative diseases.

Similar content being viewed by others
REFERENCES
Agostini, S., Clerici, M., and Mancuso, R., How plausible is a link between HSV-1 and AD?, Expert Rev. Anti. Infect. Ther., 2014, vol. 12, pp. 275–278. doi 10.1586/ 14787210.2014.887442
Albenberg, L.G. and Wu, G.D., Diet and the intestinal microbiome: associations, functions, and implications for health and disease, Gastroenterology, 2014, vol. 146, no. 6, pp. 1564–1572. doi 10.1053/j.gastro.2014.01.058
Allen, K.D., Wegrzyn, R.D., Chernova, T.A., et al., Hsp70 chaperones as modulators of prion life cycle: novel effects of Ssa and Ssb on the Saccharomyces cerevisiae prion [PSI+], Genetics, 2005, vol. 169, no. 3, pp. 1227–1242.
Alonso, R., Pisa, D., Marina, A.I., et al., Fungal infection in patients with Alzheimer’s disease, J. Alzheimers Dis., 2014, vol. 41, pp. 301–311. doi 10.3233/JAD-132681
Álvarez, G., Aldudo, J., Alonso, M., et al., Herpes simplex virus type 1 induces nuclear accumulation of hyperphosphorylated tau in neuronal cells, J. Neurosci. Res., 2012, vol. 90, no. 5, pp. 1020–1029.
Arroyo, D.S., Soria, J.A., Gaviglio, E.A., et al., Toll-like receptors are key players in neurodegeneration, Int. Immunopharmacol., 2011, vol. 11, no. 10, pp. 1415–1421. doi 10.1016/j.intimp.2011.05.006
Asti, A. and Gioglio, L., Can a bacterial endotoxin be a key factor in the kinetics of amyloid fibril formation?, J. Alzheimer’s Dis., 2014, vol. 39, no. 1, pp. 169–179. doi 10.3233/JAD-131394
Aziz, Q., Doré, J., Emmanuel, A., Guarner, F., and Quigley, E.M., Gut microbiota and gastrointestinal health: current concepts and future directions, Neurogastroenterol. Motil., 2013, vol. 25, pp. 4–15. doi 10.1111/nmo.12046
Backhed, F., Ley, R.E., Sonnenburg, J.L., et al., Host–acterial mutualism in the human intestine, Science, 2005, vol. 307, pp. 1915–1920. doi 10.1126/science.1104816
Ball, M.J., Lukiw, W.J., Kammerman, E.M., and Hill, J.M., Intracerebral propagation of Alzheimer’s disease: strengthening evidence of a herpes simplex virus etiology, Alzheimer’s Dement., 2013, vol. 9, pp. 169–175. doi 10.1016/j.jalz.2012.07.005
Barajon, I., Serrao, G., Arnaboldi, F., et al., Toll-like receptors 3, 4, and 7 are expressed in the enteric nervous system and dorsal root ganglia, J. Histochem. Cytochem., 2009, vol. 57, no. 11, pp. 1013–1023. doi 10.1369/jhc.2009.953539
Barrett, E., Ross, R.P., O’Toole, P.W., et al., γ-Aminobutyric acid production by culturable bacteria from the human intestine, J. Appl. Microbiol., 2012, vol. 113, pp. 411–417. doi 10.1111/j.1365-2672.2012.05344.x
Berer, K., Mues, M., Koutrolos, M., et al., Commensal microbiota and myelin autoantigen cooperate to trigger autoimmune demyelination, Nature, 2011, vol. 479, no. 7374, pp. 538–541. doi 10.1038/nature10554
Bhattacharjee, S. and Lukiw, W.J., Alzheimer’s disease and the microbiome, Front. Cell Neurosci., 2013, vol. 7, p. 153. doi 10.3389/fncel.2013.00153
Bojarski, L., Herms, J., and Kuznicki, J., Calcium dysregulation in Alzheimer’s disease, Neurochem. Int., 2008, vol. 52, nos. 4–5, pp. 621–633.
Borjabad, A. and Volsky, D.J., Common transcriptional signatures in brain from patients with HIV-associated neurocognitive disorders, Alzheimer’s, and multiple sclerosis, J. Neuroimmune Pharmacol., 2012, vol. 7, pp. 914–926. doi 10.1007/s11481-012-9409-5
Bowery, N.G., Bagetta, G., and Nistico, G., Intrahippocampal tetanus toxin produces generalized convulsions and neurodegeneration in rats: antagonism by NMDA receptor blockers, Epilepsy Res. Suppl., 1992, vol. 9, pp. 249–256.
Bowma, C.C., Rasley, A., Tranguch, S.L., and Marriott, I., Cultured astrocytes express toll-like receptors for bacterial products, Glia, 2003, vol. 43, pp. 281–291.
Braak, H., Rüb, U., Gai, W.P., and Del Tredici, K., Idiopathic Parkinson’s disease: possible routes by which vulnerable neuronal types may be subject to neuroinvasion by an unknown pathogen, J. Neural Transm. (Vienna), 2003, vol. 110, no. 5, pp. 517–536.
Bradley, W.G. and Mash, D.C., Beyond Guam: the cyanobacteria/BMAA hypothesis of the cause of ALS and other neurodegenerative diseases, Amyotroph. Lateral Scler., 2009, vol. 10, suppl. 2, pp. 7–20. doi 10.3109/ 17482960903286009
Braniste, V., Al-Asmakh, M., Kowal, C., et al., The gut microbiota influences blood-brain barrier permeability in mice, Sci. Transl. Med., 2014, vol. 6, p. 263ra158.
Branton, W.G., Lu, J.Q., Surette, M.G., et al., Brain microbiota disruption within inflammatory demyelinating lesions in multiple sclerosis, Sci. Rep., 2016, vol. 6, p. 37344. doi 10.1038/srep37344
Bravo, J.A., Forsythe, P., Chew, M.V., et al., Ingestion of lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve, Proc. Natl. Acad. Sci. U. S. A., 2011, vol. 108, no. 38, pp. 16050–16055. doi 10.1073/pnas.1102999108
Bravo, J.A., Julio-Pieper, M., Forsythe, P., et al., Communication between gastrointestinal bacteria and the nervous system, Curr. Opin. Pharmacol., 2012, vol. 12, no. 6, pp. 667–672. doi 10.1016/j.coph.2012.09.010
Brenner, S.R., Blue-green algae or cyanobacteria in the intestinal microflora may produce neurotoxins such as Beta-N-Methylamino-L-Alanine (BMAA) which may be related to development of amyotrophic lateral sclerosis, Alzheimer’s disease and Parkinsons-Dementia-Complex in humans and Equine Motor Neuron Disease in horses, Med. Hypotheses, 2013, vol. 80, pp. 103–108. doi 10.1016/j.mehy.2012.10.010
Brew, B.J., Robertson, K., and Wright, E.J., HIV eradication symposium: will the brain be left behind?, J. Neurovirol., 2015, vol. 21, no. 3, pp. 322–334. http://dx.doi.org/ 10.1007/s13365-015-0322-6.
Camfield, D.A., Owen, L., Scholey, A.B., et al., Dairy constituents and neurocognitive health in ageing, Br. J. Nutr., 2011, vol. 106, pp. 159–174. doi 10.1017/ S0007114511000158
Carlino, D., DeVanna, M., and Tongiorgi, E., Is altered BDNF biosynthesis a general feature in patients with cognitive dysfunction?, Neuroscientist, 2013, vol. 19, pp. 345–353. doi 10.1177/1073858412469444
Carter, C.J., Alzheimer’s disease: a pathogenetic autoimmune disorder caused by herpes simplex in a gene-dependent manner, Int. J. Alzheimers Dis., 2010, p. 140539. doi 10.4061/2010/140539
Carty, M. and Bowie, A.G., Evaluating the role of toll-like receptors in diseases of the central nervous system, Biochem. Pharmacol., 2011, vol. 81, no. 7, pp. 825–837. doi 10.1016/j.bcp.2011.01.003
Chen, B., Soto, C., and Morales, R., Peripherally administrated prions reach the brain at sub-infectious quantities, FEBS Lett., 2014, vol. 588, pp. 795–800. doi 10.1016/j.febslet.2014.01.038
Chiu, A.S., Gehringer, M.M., and Braidy, N., Gliotoxicity of the cyanotoxin, β-methyl-amino-L-alanine (BMAA), Sci. Rep., 2013, vol. 3, p. 1482. doi 10.1038/srep01482
Chiu, W.C., Tsan, Y.T., Tsai, S.L., et al., Hepatitis C viral infection and the risk of dementia, Eur. J. Neurol., 2013, vol. 21, no. 8, p. 1068-e59. doi 10.1111/ene.12317
Cho, I. and Blaser, M.J., The human microbiome: at the interface of health and disease, Nat. Rev. Genet., 2012, vol. 13, pp. 260–270. doi 10.1038/nrg3182
Churchill, M.J., Wesselingh, S.L., and Cowley, D., Extensive astrocyte infection is prominent in human immunodeficiency virus-associated dementia, Ann. Neurol., 2009, vol. 66, no. 2, pp. 253–258. http://dx.doi.org/ 10.1002/ana.21697.
Clarke, G., Stilling, R.M., Kennedy, P.J., et al., Gut microbiota: the neglected endocrine organ, Mol. Endocrinol., 2014, vol. 28, pp. 1221–1238.
Cox, P.A., Banack, S.A., and Murch, S.J., Biomagnification of cyanobacterial neurotoxins and neurodegenerative disease among the Chamorro people of Guam, Proc. Natl. Acad. Sci. U. S. A., 2003, vol. 100, no. 23, pp. 13380–13383.
Cox, P.A., Davis, D.A., Mash, D.C., et al., Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain, Proc. Biol. Sci., 2016, vol. 283, no. 1823, p. 20152397. doi 10.1098/rspb.2015.2397
Cribbs, D.H., Azizeh, B.Y., Cotman, C.W., and Laferla, F.M., Fibril formation and neurotoxicity by a herpes simplex virus glycoprotein B fragment with homology to the Alzheimer’s Aβ peptide, Biochemistry, 2000, vol. 39, pp. 5988–5994.
Dasari, M., Espargaro, A., Sabate, R., et al., Bacterial inclusion bodies of Alzheimer’s disease β-amyloid peptides can be employed to study native-like aggregation intermediate states, Chembiochem, 2011, vol. 12, pp. 407–423.
Derkinderen, P., Rouaud, T., Lebouvier, T., et al., Parkinson disease: the enteric nervous system spills its guts, Neurology, 2011, vol. 77, pp. 1761–1767. doi 10.1212/ WNL.0b013e318236ef60
Devos, D., Lebouvier, T., Lardeux, B., et al., Colonic inflammation in Parkinson’s disease, Neurobiol. Dis., 2013, vol. 50, pp. 42–48.
Diaz Heijtz, R., Wang, S., Anuar, F., et al., Normal gut microbiota modulates brain development and behavior, Proc. Natl. Acad. Sci. U. S. A., 2011, vol. 108, pp. 3047–3052. doi 10.1073/pnas.1010529108
Dickens, A.M., Yoo, S.W., Chin, A.C., et al., Chronic low-level expression of hiv-1 tat promotes a neurodegenerative phenotype with aging, Sci. Rep., 2017, vol. 7, p. 7748. doi 10.1038/s41598-017-07570-5
Dinan, T.G. and Cryan, J.F., Gut instincts: microbiota as a key regulator of brain development, ageing and neurodegeneration, J. Physiol., 2017, vol. 595, no. 2, pp. 489–503. doi 10.1113/JP273106
Douglas-Escobar, M., Elliott, E., and Neu, J., Effect of intestinal microbial ecology on the developing brain, JAMA Pediatr., 2013, vol. 167, pp. 374–379. doi 10.1001/jamapediatrics.2013.497
Dreses-Werringloer, U., Lambert, J.C., Vingtdeux, V., et al., A polymorphism in CALHM1 influences Ca2+ homeostasis, Abeta levels, and Alzheimer’s disease risk, Cell, 2008, vol. 133, no. 7, pp. 1149–1161.
Dunlop, R.A., Cox, P.A., Banack, S.A., and Rodgers, J.K., The non-protein amino acid BMAA is misincorporated into human proteins in place of L-serine causing protein misfolding and aggregation, PLoS One, 2013, vol. 8, no. 9. e75376. doi 10.1371/journal.pone.0075376
Eisenhofer, G., Aneman, A., Friberg, P., et al., Substantial production of dopamine in the human gastrointestinal tract, J. Clin. Endocrinol. Metab., 1997, vol. 82, pp. 3864–3871.
Foley, N.C., Affoo, R.H., and Martin, R.E., A systematic review and meta-analysis examining pneumonia-associated mortality in dementia, Dement. Geriatr. Cogn. Disord., 2015, vol. 39, nos. 1–2, pp. 52–67. doi 10.1159/000367783
Forsythe, P., Kunze, W.A., and Bienenstock, J., On communication between gut microbes and the brain, Curr. Opin. Gastroenterol., 2012, vol. 28, pp. 557–562. doi 10.1097/MOG.0b013e3283572ffa
Foster, J.A., Lyte, M., Meyer, E., and Cryan, J.F., Gut microbiota and brain function: an evolving field in neuroscience, Int. J. Neuropsychopharmacol., 2016, vol. 19. yv114. doi 10.1093/ijnp/pyv114
Frye, R.E., Rose, S., Slattery, J., and MacFabe, D.F., Gastrointestinal dysfunction in autism spectrum disorder: the role of the mitochondria and the enteric microbiome, Microb. Ecol. Health Dis., 2015, vol. 26, p. 27458.
Furness, J.B., Callaghan, B.P., Rivera, L.R., and Cho, H.J., The enteric nervous system and gastrointestinal innervation: integrated local and central control, Adv. Exp. Med. Biol., 2014, vol. 817, pp. 39–71. doi 10.1007/978-1-4939-0897-4_3
Galland, L., The gut microbiome and the brain, J. Med. Food, 2014, vol. 17, pp. 1261–1272.
Gao, B., Bian, X., Mahbub, R., and Lu, K., Sex-specific effects of organophosphate diazinon on the gut microbiome and its metabolic functions, Environ. Health Perspect., 2017, vol. 125, no. 2, pp. 198–206. doi 10.1289/ EHP202
von Geldern, G. and Mowry, E.M., The influence of nutritional factors on the prognosis of multiple sclerosis, Nat. Rev. Neurol., 2012, vol. 8, pp. 678–689. doi 10.1038/nrneurol.2012.194
Gibson, G.E. and Thakkar, A., Interactions of mitochondria/metabolism and calcium regulation in Alzheimer’s disease: a calcinist point of view, Neurochem. Res., 2017. doi 10.1007/s11064-017-2182-3
Glass, C.K., Saijo, K., Winner, B., et al., Mechanisms underlying inflammation in neurodegeneration, Cell, 2010, vol. 140, no. 6, pp. 918–934. doi 10.1016/ j.cell.2010.02.016
González-Scarano, F. and Martín-García, J., The neuropathogenesis of AIDS, Nat. Rev. Immunol., 2005, vol. 5, no. 1, pp. 69–81. http://dx.doi.org/10.1038/nri1527.
van de Haar, H.J., Burgmans, S., Jansen, J.F., et al., Blood-brain barrier leakage in patients with early Alzheimer disease, Radiology, 2016, vol. 281, no. 2, pp. 527–535. doi 10.1148/radiol.2016152244
Hammond, C.J., Hallock, L.R., Howanski, R.J., et al., Immunohistological detection of Chlamydia pneumoniae in Alzheimer’s disease, BMC Neurosci., 2010, vol. 11, p. 121. doi 10.1186/1471-2202-11-121
Hanisch, U.K., Microglia as a source and target of cytokines, Glia, 2002, vol. 40, no. 2, pp. 140–155.
Hansen, M.B., The enteric nervous system I: organisation and classification, Pharmacol. Toxicol., 2003, vol. 92, no. 3, pp. 105–113. PMID 12753424
Harrington, P.R., Schnell, G., Letendre, S.L., et al., Cross-sectional characterization of HIV-1 env compartmentalization in cerebrospinal fluid over the full disease course, AIDS, 2009, vol. 23, no. 8, pp. 907–915. http://dx.doi.org/10.1097/QAD.0b013e3283299129.
Hattori, M. and Taylor, T.D., The human intestinal microbiome: a new frontier of human biology, DNA Res., 2009, vol. 16, pp. 1–12.
Heaton, R.K., Clifford, D.B., Franklin, D.R., Jr., et al., HIV-associated neurocognitive disorders persist in the era of potent antiretroviral therapy: CHARTER Study, Neurology, 2010, vol. 75, no. 23, pp. 2087–2096. http://dx.doi.org/10.1212/WNL.0b013e318200d727.
Heintz, C. and Mair, W., You are what you host: microbiome modulation of the aging process, Cell, 2014, vol. 156, pp. 408–411. doi 10.1016/j.cell.2014.01.025
Hernandez-Rapp, J., Martin-Lannerée, S., Hirsch, T.Z., et al., Hijacking PrP(c)-dependent signal transduction: when prions impair Aβ clearance, Front. Aging Neurosci., 2014, vol. 6, p. 25. doi 10.3389/fnagi.2014.00025
Hill, J.M. and Lukiw, W.J., Comparing miRNAs and viroids; highly conserved molecular mechanisms for the transmission of genetic information, Front. Cell Neurosci., 2014, vol. 8, p. 45. doi 10.3389/fncel.2014.00045
Hill, J.M. and Lukiw, W.J., Microbial-generated amyloids and Alzheimer’s disease (AD), Front. Aging Neurosci., 2015, vol. 7, p. 9. doi 10.3389/fnagi.2015.00009
Hill, J.M., Zhao, Y., Clement, C., et al., HSV-1 infection of human brain cells induces miRNA-146a and Alzheimer-type inflammatory signaling, Neuroreport, 2009, vol. 20, pp. 1500–1505. doi 10.1097/WNR.0b013e3283329c05
Hill, J.M., Clement, C., and Pogue, A.I., Pathogenic microbes, the microbiome, and Alzheimer’s disease (AD), Front. Aging Neurosci., 2014, vol. 6, p. 127. doi 10.3389/fnagi.2014.00127
Holmqvist, S., Chutna, O., Bousset, L., et al., Direct evidence of Parkinson pathology spread from the gastrointestinal tract to the brain in rats, Acta Neuropathol., 2014, vol. 128, no. 6, pp. 805–820. doi 10.1007/s00401-014-1343-6
Holtcamp, W., The emerging science of BMAA: do cyanobacteria contribute to neurodegenerative disease?, Environ. Health. Perspect., 2012, vol. 120, no. 3, pp. A110–A116. doi 10.1289/ehp.120-a110
Holzer, P. and Farzi, A., Neuropeptides and the microbiota–gut–brain axis, in Microbial Endocrinology: The Microbiota-Gut-Brain Axis in Health and Disease, Lyte, M. and Cryan, J.F., Eds., Adv. Exp. Med. Biol., New York: Springer, 2014, vol. 817, pp. 195–219.
Hornig, M., The role of microbes and autoimmunity in the pathogenesis of neuropsychiatric illness, Curr. Opin. Rheumatol., 2013, vol. 25, pp. 488–795. doi 10.1097/ BOR.0b013e32836208de
Huang, W.S., Yang, T.Y., Shen, W.C., et al., Association between Helicobacter pylori infection and dementia, J. Clin. Neurosci., 2014, vol. 21, no. 8, pp. 1355–1358. doi 10.1016/j.jocn.2013.11.018
Jangi, S., Gandhi, R., Cox, L.M., et al., Alterations of the human gut microbiome in multiple sclerosis, Nat. Commun., 2016, vol. 7, p. 12015. doi 10.1038/ncomms12015
Kannarkat, G.T., Boss, J.M., and Tansey, M.G., The role of innate and adaptive immunity in Parkinson’s disease, J. Parkinsons Dis., 2013, vol. 3, pp. 493–514.
Karlsson, O., Berg, A.L., Hanrieder, J., et al., Intracellular fibril formation, calcification, and enrichment of chaperones, cytoskeletal, and intermediate filament proteins in the adult hippocampus CA1 following neonatal exposure to the nonprotein amino acid BMAA, Arch. Toxicol., 2015, vol. 89, no. 3, pp. 423–436. doi 10.1007/ s00204-014-1262-2
Kelly, J.R., Allen, A.P., Temko, A., et al., Lost in translation? The potential psychobiotic Lactobacillus rhamnosus (JB-1) fails to modulate stress or cognitive performance in healthy male subjects, Brain Behav. Immun., 2017, vol. 61, pp. 50–59. doi 10.1016/j.bbi.2016.11.018
Kielian, T., Toll-like receptors in central nervous system glial inflammation and homeostasis, J. Neurosci. Res., 2006, vol. 83, no. 5, pp. 711–730.
Kim, J.S. and Sung, H.Y., Gastrointestinal autonomic dysfunction in patients with Parkinson’s disease, J. Mov. Disord., 2015, vol. 8, no. 2, pp. 76–82. doi 10.14802/jmd.15008
Kim, H.S., Lee, J.H., Lee, J.P., et al., Amyloid beta peptide induces cytochrome c release from isolated mitochondria, Neuroreport, 2002, vol. 13, pp. 1989–1993.
Kim, B.S., Jeon, Y.S., and Chun, J., Current status and future promise of the human microbiome, Pediatr. Gastroenterol. Hepatol. Nutr., 2013, vol. 16, pp. 71–79. doi 10.5223/pghn.2013.16.2.71
König, J., Wells, J., Cani, P.D., et al., Human intestinal barrier function in health and disease. Clin. Transl. Gastroenterol., 2016, vol. 7, no. 10. e196. doi 10.1038/ctg.2016.54
Kountouras, J., Tsolaki, M., Gavalas, E., et al., Relationship between Helicobacter pylori infection and Alzheimer disease, Neurology, 2006, vol. 66, pp. 938–940.
Kountouras, J., Boziki, M., Gavalas, E., et al., Five-year survival after Helicobacter pylori eradication in Alzheimer disease patients, Cogn. Behav. Neurol., 2010, vol. 23, no. 3, pp. 199–204. doi 10.1097/WNN.0b013e3181df3034
Kramer-Hämmerle, S., Rothenaigner, I., Wolff, H., et al., Cells of the central nervous system as targets and reservoirs of the human immunodeficiency virus, Virus Res., 2005, vol. 111, no. 2, pp. 194–213. http://dx.doi.org/ 10.1016/j.virusres. 2005.04.009. PMID: 15885841.
Krogh, K.A., Lyddon, E., and Thayer, S.A., HIV-1 Tat activates a RhoA signaling pathway to reduce NMDA evoked calcium responses in hippocampal neurons via an actin-dependent mechanism, J. Neurochem., 2015, vol. 132, no. 3, pp. 354–366. http://dx.doi.org/10.1111/jnc.12936.
Laferla, F.M., Calcium dyshomeostasis and intracellular signalling in Alzheimer’s disease, Nat. Rev. Neurosci., 2002, vol. 3, pp. 862–872.
Lakhan, S.E., Caro, M., and Hadzimichalis, N., NMDA receptor activity in neuropsychiatric disorders, Front. Psychiatry, 2013, vol. 4, pp. 52–55. doi 10.3389/ fpsyt.2013.00052
Lehnardt, S., Innate immunity and neuroinflammation in the CNS: the role of microglia in Toll-like receptor-mediated neuronal injury, Glia, 2010, vol. 58, no. 3, pp. 253–263. doi 10.1002/glia.20928
Letenneur, L., Peres, K., Fleury, H., et al., Seropositivity to herpes simplex virus antibodies and risk of Alzheimer’s disease: a population-based cohort study, PLoS One, 2008, vol. 3. e3637
Licastro, F., Porcellini, E., Caruso, C., et al., Genetic risk profiles for Alzheimer’s disease: integration of APOE genotype and variants that up-regulate inflammation, Neurobiol. Aging, 2007, vol. 28, pp. 1637–1643.
Liu, M. and Bing, G., Lipopolysaccharide animal models for Parkinson’s disease, Parkinsons Dis., 2011, vol. 2011, p. 327089. doi 10.4061/2011/327089
Lobner, D., Piana, P.M., Salous, A.K., and Peoples, R.W., Beta-N-methylamino-L-alanine enhances neurotoxicity through multiple mechanisms, Neurobiol Dis., 2007, vol. 25, no. 2, pp. 360–366.
Lu, B., Nagappan, G., Guan, X., et al., BDNF-based synaptic repair as a disease-modifying strategy for neurodegenerative diseases, Nat. Rev. Neurosci., 2013, vol. 14, pp. 401–416. doi 10.1038/nrn3505
Lukiw, W.J., Bacteroides fragilis lipopolysaccharide and inflammatory signaling in Alzheimer’s disease, Front. Microbiol., 2016, vol. 7, p. 1544. eCollection 2016.
Lyte, M., Microbial endocrinology in the microbiome–gut–rain axis: how bacterial production and utilization of neurochemicals influence behavior, PLoS Pathog., 2013, vol. 9, no. 11. e1003726. doi 10.1371/journal.ppat.1003726
Lyte, M. and Cryan, J.F., Microbial Endocrinology: The Microbiota–Gut–Brain Axis in Health and Disease, Adv. Exp. Med. Biol., New York: Springer, 2014, vol. 817.
MacFabe, D.F., Cain, D.P., Rodriguez-Capote, K., et al., Neurobiological effects of intraventricular propionic acid in rats: possible role of short chain fatty acids on the pathogenesis and characteristics of autism spectrum disorders, Behav. Brain Res., 2007, vol. 176, pp. 47–54. doi 10.1016/j.bbr.2010.10.005
MacFabe, D.F., Cain, N.E., Boon, F., et al., Effects of the enteric bacterial metabolic product propionic acid on object-directed behavior, social behavior, cognition, and neuroinflammation in adolescent rats: relevance to autism spectrum disorder, Behav. Brain Res., 2011, vol. 217, no. 1, pp. 149–169. doi 10.1016/j.bbr.2006.07.025
Malaguarnera, M., Bella, R., Alagona, G., et al., Helicobacter pylori and Alzheimer’s disease: a possible link, Eur. J. Int. Med., 2004, vol. 15, pp. 381–386.
Manabe, T., Mizukami, K., Akatsu, H., et al., Influence of pneumonia complications on the prognosis of patients with autopsy-confirmed Alzheimer’s disease, dementia with Lewy bodies, and vascular dementia, Psychogeriatrics, 2016, vol. 16, no. 5, pp. 305–314. doi 10.1111/psyg.12163
Mancuso, R., Baglio, F., Cabinio, M., et al., Titers of HSV-1 antibodies correlate with grey matter volumes in AD, J. Alzheimers Dis., 2014, vol. 38, no. 4, pp. 741–745. doi 10.3233/JAD-130977
Manuelidis, L., Infectious particles, stress, and induced prion amyloids: a unifying perspective, Virulence, 2013, vol. 4, pp. 373–383. doi 10.4161/viru.24838
Marques, F., Sousa, J.C., Sousa, N., and Palha, J.A., Blood–brain-barriers in aging and in Alzheimer’s disease, Mol. Neurodegener., 2013, vol. 8, p. 38.
Maruszak, A., Pilarski, A., Murphy, T., et al., Hippocampal neurogenesis in Alzheimer’s disease: is there a role for dietary modulation?, J. Alzheimers Dis., 2014, vol. 38, pp. 11–38.
Matsumoto, M., Kibe, R. Ooga, T., et al., Cerebral low-molecular metabolites influenced by intestinal microbiota: a pilot study, Front. Syst. Neurosci., 2013, vol. 7, p. 9. doi 10.3389/fnsys.2013.00009
Mattson, M.P., Cheng, B., Davis, D., et al., Beta-amyloid peptides destabilize calcium homeostasis and render human cortical neurons vulnerable to excitotoxicity, J. Neurosci., 1992, vol. 12, no. 2, pp. 376–389.
Mattson, M.P., Infectious agents and age-related neurodegenerative disorders, Ageing Res. Rev., 2004, vol. 3, pp. 105–120.
Mayer, E.A., Knight, R., Mazmanian, S.K., et al., Gut microbes and the brain: paradigm shift in neuroscience, J. Neurosci., 2014, vol. 34, no. 46, pp. 15490–15496. doi 10.1523/JNEUROSCI.3299-14.2014
Mayer, E.A., Tillisch, K., and Gupta, A., Gut/brain axis and the microbiota, J. Clin. Invest., 2015, vol. 125, no. 3, pp. 926–938. doi 10.1172/JCI76304
McKimmie, C.S., Johnson, N., Fooks, A.R., and Fazakerley, J.K., Viruses selectively upregulate Toll-like receptors in the central nervous system, Biochem. Biophys. Res. Commun., 2005, vol. 336, pp. 925–933.
McNaught, K.S., Perl, D.P., Brownell, A.L., and Olanow, C.W., Systemic exposure to proteasome inhibitors causes a progressive model of Parkinson’s disease, Ann. Neurol., 2004, vol. 56, no. 1, pp. 149–162.
McVey, NeufeldK.A., Mao, Y.K., Bienenstock, J., et al., The microbiome is essential for normal gut intrinsic primary afferent neuron excitability in the mouse, Neurogastroenterol. Motil., 2013, vol. 25, pp. 183–188. doi 10.1111/nmo.12049
Miklossy, J., Emerging role of pathogens in Alzheimer’s disease, Expert. Rev. Mol. Med., 2011, vol. 13. e30. doi 10.1017/S1462399411002006
Minter, M.R., Taylor, J.M., and Crack, P.J., The contribution of neuroinflammation to amyloid toxicity in Alzheimer’s disease, J. Neurochem., 2016, vol. 136, no. 3, pp. 457–474. doi 10.1111/jnc.13411
Mitew, S., Kirkcaldie, M.T., Dickson, T.C., and Vickers, J.C., Altered synapses and gliotransmission in Alzheimer’s disease and ad model mice, Neurobiol. Aging, 2013, vol. 34, pp. 2341–2351. doi 10.1016/j.neurobiolaging.2013.04.010
Montagne, A., Barnes, S.R., Sweeney, M.D., et al., Blood–brain barrier breakdown in the aging human hippocampus, Neuron, 2015, vol. 85, no. 2, pp. 296–302. doi 10.1016/j.neuron.2014.12.032
Moulignier, A., Gueguen, A., Lescure, F.X., et al., Does HIV infection alter Parkinson disease?, J. Acquir. Immune Defic. Syndr., 2015, vol. 70, no. 2, pp. 129–136. http://dx.doi.org/10.1097/QAI.0000000000000677.
Mulligan, V.K. and Chakrabartty, A., Protein misfolding in the late-onset neurodegenerative diseases: common themes and the unique case of amyotrophic lateral sclerosis, Proteins, 2013, vol. 81, pp. 1285–1303. doi 10.1002/prot.24285
Obata, Y. and Pachnis, V., The effect of microbiota and the immune system on the development and organization of the enteric nervous system, Gastroenterology, 2016, vol. 151, no. 5, pp. 836–844. doi 10.10153/j.gastro.2016.07.044
Oh, S.H., Kim, H.N., Park, H.J., et al., Mesenchymal stem cells increase hippocampal neurogenesis and neuronal differentiation by enhancing the wnt signaling pathway in an Alzheimer’s disease model, Cell Transplant., 2015, vol. 24, pp. 1097–1109.
Oleskin, A.V. and Shenderov, B.A., Neuromodulatory effects and targets of the SCFAs and gasotransmitters produced by the human symbiotic microbiota, Microb. Ecol. Health Dis., 2016, vol. 27, p. 30971.
Oleskin, A.V., El’-Registan, G.I., and Shenderov, B.A., Role of neuromediators in the functioning of the human microbiota: “business talks” among microorganisms and the microbiota–host dialogue, Microbiology (Moscow), 2016, vol. 85, no. 1, pp. 1–22.
Pablo, J., Banack, S.A., Cox, P.A., et al., Cyanobacterial neurotoxin BMAA in ALS and Alzheimer’s disease, Acta Neurologica Scandinavica, 2009, vol. 120, no. 4, pp. 216–225. doi 10.1111/j.1600-0404.2008.01150.x
Parashar, A. and Udayabanu, M., Gut microbiota: implications in Parkinson’s disease, Parkinsonism Relat. Disord., 2017, vol. 38, pp. 1–7. doi 10.1016/j.parkreldis.2017.02.002
Prandota, J., Possible link between Toxoplasma gondii and the anosmia associated with neurodegenerative diseases, Am. J. Alzheimers Dis. Other Demen., 2014, vol. 29, pp. 205–214. doi 10.1177/1533317513517049
Prusiner, S.B., Biology and genetics of prions causing neurodegeneration, Ann. Rev. Genet., 2013, vol. 47, pp. 601–623. doi 10.1146/annurev-genet-110711-155524
Putney, J.W., Jr., Presenilins, Alzheimer’s disease, and capacitative calcium entry, Neuron, 2000, vol. 27, no. 3, pp. 411–412.
Rao, S.D., Banack, S.A., Cox, P.A., and Weiss, J.H., BMAA selectively injures motor neurons via AMPA/kainate receptor activations. Exp. Neurol., 2006, vol. 201, no. 1, pp. 244–252. doi 10.1016/j.expneurol.2006.04.017
Rhee, S.H., Pothoulakis, C., and Mayer, E.A., Principles and clinical implications of the brain–gut–enteric microbiota axis, Nat. Rev. Gastroenterol. Hepatol., 2009, vol. 6, no. 5, pp. 306–314. doi 10.1038/nrgastro.2009.35
Ritz, B.R., Paul, K.C., and Bronstein, J.M., Of pesticides and men: a California story of genes and environment in Parkinson’s disease, Curr. Environ. Health Rep., 2016, vol. 3, pp. 40–52.
Rivest, S., Regulation of innate immune responses in the brain, Nat. Rev. Immunol., 2009, vol. 9, pp. 429–439.
Sabatier, J.M., Vives, E., Mabrouk, K., et al., Evidence for neurotoxic activity of tat from human immunodeficiency virus type 1, J. Virol., 1991, vol. 65, pp. 961–967.
Sampson, T.R., Debelius, J.W., Thron, T., et al., Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease, Cell, 2016, vol. 167, no. 6, pp. 1469–1480. doi 10.1016/ j.cell.2016.11.018
Sanchez-Guajardo, V., Barnum, C.J., Tansey, M.G., and Romero-Ramos, M., Neuroimmunological processes in Parkinson’s disease and their relation to a-synuclein: microglia as the referee between neuronal processes and peripheral immunity, ASN Neuro, 2013, vol. 5, pp. 113–139.
Santana, S., Recuero, M., Bullido, M.J., et al., Herpes simplex virus type I induces the accumulation of intracellular beta-amyloid in autophagic compartments and the inhibition of the non-amyloidogenic pathway in human neuroblastoma cells, Neurobiol. Aging, 2012, vol. 33, no. 2, p. 430. e19–33
Saulnier, D.M., Ringel, Y., Heyman, M.B., et al., The intestinal microbiome, probiotics and prebiotics in neurogastroenterology, Gut Microbes, 2013, vol. 4, no. 1, pp. 17–27. doi 10.4161/gmic.22973
Di Scala, C., Yahi, N., Boutemeur, S., et al., Common molecular mechanism of amyloid pore formation by Alzheimer’s β-amyloid peptide and α -synuclein, Sci. Rep., 2016, vol. 6, p. 28781. doi 10.1038/srep28781
Schwartz, K. and Boles, B.R., Microbial amyloids-functions and interactions within the host, Curr. Opin. Microbiol., 2013, vol. 16, pp. 93–99. doi 10.1016/j.mib.2012.12.001
Selkrig, J., Wong, P., Zhang, X., and Pettersson, S., Metabolic tinkering by the gut microbiome: implications for brain development and function, Gut Microbes, 2014, vol. 5, no. 3, pp. 369–380. doi 10.4161/gmic.28681
Sharon, G., Sampson, T.R., Geschwind, D.H., and Mazmanian, S.K., The central nervous system and the gut microbiome, Cell, 2016, vol. 167, no. 4, pp. 915–932. doi 10.1016/j.cell.2016.10.027
Shenderov, B.A., Probiotic (symbiotic) bacterial languages, Anaerobe, 2011, vol. 17, pp. 490–495.
Silei, V., Fabrizi, C., Venturini, G., et al., Activation of microglial cells by PrP and beta-amyloid fragments raises intracellular calcium through L-type voltage sensitive calcium channels, Brain Res., 1999, vol. 818, no. 1, pp. 168–170.
Smith, M.Z., Wightman, F., and Lewin, S.R., HIV reservoirs and strategies for eradication, Curr. HIV/AIDS. Rep., 2012, vol. 9, no. 1, pp. 5–15. http://dx.doi.org/ 10.1007/s11904-011-0108-2.
Sobol, C.V., Mikroflora i serdechno-sosudistaya sistema v norme i pri patologii (Microflora and the Cardiovascular System in Health and Disease), Lambert Academic Publishing, 2014.
Sobol., C.V., Antineoplastic and antimutagenic effects of a new probiotic product at the cellular level and in rats with transplanted fast-growing Pliss’ lymphosarcoma, Int. J. Probiot. Prebiot., 2015, vol. 10, pp. 133–144.
Sobol., C.V., A novel complementary approach using new probiotic product for the improvement of HIV therapy, in Frontiers in Clinical Drug Research-Anti Invectives, Atta-ur-Rahman, Ed., Sharjah, United Arab Emirates: Bentham Science Publishers, 2017a, vol. 3, pp. 49–121. https:// www.dropbox.com/s/4cykdoth8iuuyiu/9781681083698R. pdf?dl=0.
Sobol, C.V., A new class of pharmabiotics with unique properties, in Soft Chemistry and Food Fermentation, Grumezescu, A.M. and Holban, A.M., Eds., Elsevier, 2017b, vol. 3, pp. 79–112. https://www.sciencedirect. com/science/article/pii/B9780128114124000047.
Sobol, C.V. and Belostotskaya, G.B., Product fermented by Lactobacilli induces changes in intracellular calcium dynamics in rat brain neurons, Biochemistry (Moscow) Suppl. Series A: Membr. Cell Biol., 2016, vol. 10, no. 1, pp. 37–45.
Sobol, C.V. and Sobol, Yu.Ts., Composition and method for producing and use of a fermented hydrolyzed medium containing microorganisms and products of their metabolism, US Patent no. 6953574, 2005.
Sobol., C.V., Belostotskaya, G.B., and Kenworthy, M.W., Calcium signalling in rat brain neurons and differentiation of PC-12 cells induced by application of a probiotic product, Neurophysiology (Ukraine), 2005, vol. 37, pp. 284–293.
Sobol, C.V., Korotkov, S.M., Belostotskaya, G.B., and Nesterov, V.P., The influence of probiotics and probiotic product on respiration of mitochondria and intracellular calcium signal in cells of cardiovascular system, Biochemistry (Moscow) Suppl. Series A: Membr. Cell Biol., 2013, vol. 7, no. 4, pp. 294–301.
Soret, R., Chevalier, J., De Coppet, P., et al., Short-chain fatty acids regulate the enteric neurons and control gastrointestinal motility in rats, Gastroenterology, 2010, vol. 138, no. 5, pp. 1772–1782. doi 10.1053/j.gastro.2010.01.053
Soscia, S.J., Kirby, J.E., Washicosky, K.J., et al., The Alzheimer’s disease-associated amyloid β-protein is an antimicrobial peptide, PLoS One, 2010, vol. 5. e9505
Suh, J., Sinclair, E., Peterson, J., et al., Progressive increase in central nervous system immune activation in untreated primary HIV-1 infection, J. Neuroinflammation, 2014, vol. 11, p. 199. http://dx.doi.org/10.1186/s12974-014-0199-y.
Tkachenko, E.I. and Uspenskii, Yu.P., Pitanie, mikrobiotsenoz i intellekt cheloveka (Nutrition, Microbiocenosis, and Human Intelligence), St. Petersburg: SpetsLit, 2006.
Torbick, N., Hession, S., Stommel, E., and Caller, T., Mapping amyotrophic lateral sclerosis lake risk factors across northern New England, Int. J. Health Geogr., 2014, vol. 13, p. 1. doi 10.1186/1476-072X-13-1
Tran, L. and Greenwood-Van Meerveld, B., Age-associated remodeling of the intestinal epithelial barrier, J. Gerontol. A Biol. Sci. Med. Sci., 2013, vol. 68, pp. 1045–1056. doi 10.1093/gerona/glt106
Del Tredici, K. and Braak, H., A not entirely benign procedure: progression of Parkinson’s disease, Acta Neuropathol., 2008, vol. 115, pp. 379–384.
Udit, S. and Gautron, L., Molecular anatomy of the gut-brain axis revealed with transgenic technologies: implications in metabolic research, Front. Neurosci., 2013, vol. 7, p. 134. doi 10.3389/fnins.2013.00134
Valera, E. and Masliah, E., Combination therapies: the next logical step for the treatment of synucleinopathies?, Mov. Disord., 2016, vol. 31, pp. 225–234.
Varatharaj, A. and Galea, I., The blood-brain barrier in systemic inflammation, Brain Behav. Immun, 2017, vol. 60, pp. 1–12. doi 10.1016/j.bbi.2016.03.010
Verma, R., Verma, A.K., Ahuja, V., and Paul, J., Real-time analysis of mucosal flora in patients with inflammatory bowel disease in India, J. Clin. Microbiol., 2010, vol. 48, pp. 4279–4282.
Voloshko, L.N. and Pinevich, A.V., Diversity of cyanobacterial toxins, Astrakhan. Vestn. Ekol. Obraz., 2014, vol. 1, no. 27, pp. 68–80.
Wall, R., Cryan, J.F., Ross, R.P., et al., Bacterial neuroactive compounds produced by psychobiotics, Adv. Exp. Med. Biol., 2014, vol. 817, pp. 221–239.
Wang, X.L., Zeng, J., Feng, J., et al., Helicobacter pylori filtrate impairs spatial learning and memory in rats and increases β-amyloid by enhancing expression of presenilin-2, Front. Aging Neurosci., 2014, vol. 6, p. 66. doi 10.3389/fnagi.2014.00066
Welling, M.M., Nabuurs, R.J., and van der Weerd, L., Potential role of antimicrobial peptides in the early onset of Alzheimer’s disease, Alzheimers Dement., 2015, vol. 11, no. 1, pp. 51–57. doi 10.1016/j.jalz.2013.12.020
Widera, M., Klein, A.N., Cinar, Y., et al., The D-amino acid peptide D3 reduces amyloid fibril boosted HIV-1 infectivity, AIDS Res. Ther., 2014, vol. 11, no. 1, p. 1. doi 10.1186/1742-6405-11-1
Wozniak, M.A., Frost, A.L., Preston, C.M., and Itzhaki, R.F., Antivirals reduce the formation of key Alzheimer’s disease molecules in cell cultures acutely infected with herpes simplex virus type 1, PLoS One, vol. 6. e25152.
Xie, X., Basile, M., and Mash, D.C., Cerebral uptake and protein incorporation of cyanobacterial toxin β-N-methylamino-L-alanine, Neuroreport, 2013, vol. 24, no. 14, pp. 779–784. doi 10.1097/WNR.0b013e328363fd89
Xu, J. and Ikezu, T., The comorbidity of HIV-associated neurocognitive disorders and Alzheimer’s disease: a foreseeable medical challenge in post-HAART era, J. Neuroimmune Pharmacol., 2009, vol. 4, no. 2, pp. 200–212.
Yin, H.Z., Yu, S., Hsu, C.-I., et al., Intrathecal infusion of BMAA induces selective motor neuron damage and astrogliosis in the ventral horn of the spinal cord, Exp. Neurol., 2014, vol. 261, pp. 1–9. doi 10.1016/ j.expneurol.2014.06.003
Yu, Y. and Ye, R.D., Microglial Aβ receptors in Alzheimer’s disease, Cell. Mol. Neurobiol., 2015, vol. 35, no. 1, pp. 71–83. doi 10.1007/s10571-014-0101-6
Yu, J.T., Chang, R.C., and Tan, L., Calcium dysregulation in Alzheimer’s disease: from mechanisms to therapeutic opportunities, Prog. Neurobiol., 2009, vol. 89, no. 3, pp. 240–255.
Zambrano, A., Solis, L., Salvadores, N., et al., Neuronal cytoskeletal dynamic modification and neurodegeneration induced by infection with herpes simplex virus type 1, J. Alzheimers Dis., 2008, vol. 14, no. 3, pp. 259–269.
Zhang, H., Sun, S., Wu, L., et al., Store-operated calcium channel complex in postsynaptic spines: a new therapeutic target for Alzheimer’s disease treatment, J. Neurosci., 2016, vol. 36, pp. 11837–11850.
Zhao, Y., Dua, P., and Lukiw, W.J., Microbial sources of amyloid and relevance to amyloidogenesis and Alzheimer’s disease (AD), J. Alzheimers Dis. Parkinsonism, 2015, vol. 5, no. 1, p. 177.
Zhou, Y., Blanco, L.P., Smith, D.R., and Chapman, M.R., Bacterial amyloids, Methods Mol. Biol., 2012, vol. 849, pp. 303–320.
Zhou, L., Miranda-Saksena, M., and Saksena, N.K., Viruses and neurodegeneration, Virol. J., 2013, vol. 10, p. 172. doi 10.1186/1743-422X-10-172
ACKNOWLEDGMENTS
I am grateful to Doctor of Biology I.V. Shemarova for a number of valuable comments and additions.
The work was supported by the Federal Agency for Scientific Organizations of Russia (topic no. АААА-А18-118012290142-9).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by A. Barkhash
Rights and permissions
About this article
Cite this article
Sobol, C.V. Role of Microbiota in Neurodegenerative Diseases. Russ J Dev Biol 49, 297–313 (2018). https://doi.org/10.1134/S1062360418060061
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062360418060061