Abstract
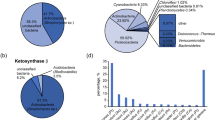
In the present study, we analyze the sequences of gene fragments of polyketide synthases (PKS) of microorganisms associated with the endemic Baikal sponge Baikalospongia fungiformis. PKSs are multienzyme complexes responsible for the synthesis of low molecular weight biologically active substances (BAS). Cloning and sequencing of the amplification products of the ketosynthase (KS) domain of PKS in the B. fungiformis microbiome reveal 18 unique sequences, most of which were highly identical (97–99%) to the genes of PKS of microorganisms from communities of other species of Baikal sponges. On phylogenetic trees, these groups of sequences form characteristic “Baikal clades.” Apparently, the basic sponge community that formed during the coevolution contains common groups of microorganisms that are potential producers of biologically active substances. BlastX analysis showed that the obtained fragments belong to bacterial polyketide synthases (phyla Cyanobacteria, Proteobacteria, Planctomycetes, Latescibacteria, and Gemmatimonadates), as well as eukaryotic algae (divisions Haptophyta and Ochrophyta). This work indicates a significant biotechnological potential of freshwater sponge communities and the prospects for research in the direction of searching for new natural metabolites in complex freshwater microbiomes.


Similar content being viewed by others
REFERENCES
Webster, N.S. and Taylor, M.W., Marine sponges and their microbial symbionts: love and other relationships, Environ. Microbiol., 2012, vol. 14, no. 2, pp. 335—346. https://doi.org/10.1111/j.1462-2920.2011.02460.x
Thomas, T.R., Kavlekar, D.P., and LokaBharathi, P.A., Marine drugs from sponge microbe association—a review, Mar. Drugs, 2010, vol. 8, pp. 1417—1468. https://doi.org/10.3390/md8041417
Khalifa, S.A.M., Elias, N., Farag, M.A., et al., Marine natural products: a source of novel anticancer drugs, Mar. Drugs, 2019, vol. 17, no. 9. https://doi.org/10.3390/md17090491
Ehrenreich, I., Waterbury, J., and Webb, E., Distribution and diversity of natural product genes in marine and freshwater cyanobacterial cultures and genomes, Appl. Environ. Microb., 2005, vol. 71, pp. 7401—7413. https://doi.org/10.1128/AEM.71.11.7401-7413.2005
Weissman, K.J., Introduction to polyketide biosynthesis, Methods Enzymol., 2009, vol. 459, pp. 3—16. https://doi.org/10.1016/S0076-6879(09)04601-1
Yu, D., Xu, F., Zeng, J., et al., Type III polyketide synthases in natural product biosynthesis, IUBMB Life, 2012, vol. 64, pp. 285—295. https://doi.org/10.1002/iub.1005
Mullis, M.M., Rambo, I.M., Baker, B.J., et al., Diversity, ecology, and prevalence of antimicrobials in nature, Front. Microbiol., 2019, vol. 10. https://doi.org/10.3389/fmicb.2019.02518
Wang, B., Guo, F., Huang, C., et al., Unraveling the iterative type I polyketide synthases hidden in Streptomyces, Proc. Natl. Acad. Sci. U.S.A., 2020, vol. 117, no. 15, pp. 8449—8454. https://doi.org/10.1073/pnas.1917664117
Kurnia, N.M., Uria, A.R., Yudi Kusnadi, Y., et al., Metagenomic survey of potential symbiotic bacteria and polyketide synthase genes in an Indonesian marine sponge, HAYATI J. Biosci., 2017, vol. 24, no. 1, pp. 6—15. https://doi.org/10.1016/j.hjb.2017.04.004
Hertweck, C., The biosynthetic logic of polyketide diversity, Angew. Chem. Int. Ed. Engl., 2009, vol. 48, no. 26, pp. 4688—4716. https://doi.org/10.1002/anie.200806121
Ansari, M.Z., Yadav, G., Gokhale, R.S., et al., NRPS-PKS: a knowledge-based resource for analysis of NRPS/PKS megasynthases, Nucleic Acids Res., 2004, vol. 32, pp. 405—413. https://doi.org/10.1093/nar/gkh359
Moffitt, M.C. and Neilan, B.A., Evolutionary affiliations within the superfamily of ketosynthases reflect complex pathway associations, J. Mol. Evol., 2003, vol. 56, pp. 446—457. https://doi.org/10.1007/s00239-002-2415-0
Kennedy, J., Codling, C.E., Jones, B.V., et al., Diversity of microbes associated with the marine sponge, Haliclona simulans, isolated from Irish waters and identification of polyketide synthase genes from the sponge metagenome, Environ. Microbiol., 2008, vol. 10, pp. 1888—1902. https://doi.org/10.1111/j.1462-2920.2008.01614.x
Hochmuth,T. and Piel, J., Polyketide synthases of bacterial symbionts in sponges-evolution-based applications in natural products research, Phytochemistry, 2009, vol. 70, nos. 15—16, pp. 1841—1849. https://doi.org/10.1016/j.phytochem.2009.04.010
Newman, D.J. and Cragg, G.M., Natural products as sources of new drugs over the 30 years from 1981 to 2010, J. Nat. Prod., 2012, vol. 75, pp. 311—335. https://doi.org/10.1021/np200906s
Jenke-Kodama, H. and Dittmann, E., Evolution of metabolic diversity: insights from microbial polyketidesynthases, Phytochemistry, 2009, vol. 70, pp. 1858—1866. https://doi.org/10.1016/j.phytochem.2009.05.021
Esteves, A.I., Hardoim, C.C., Xavier, J.R., et al., Molecular richness and biotechnological potential of bacteria cultured from Irciniidae sponges in the north-east Atlantic, FEMS Microbiol. Ecol., 2013, vol. 85, no. 3, pp. 519—536. https://doi.org/10.1111/1574-6941.12140
Della Sala, G., Hochmuth, T., Teta, R., et al., Polyketide synthases in the microbiome of the marine sponge Plakortis halichondrioides: a metagenomic update, Mar. Drugs, 2014, vol. 12, pp. 5425—5440. https://doi.org/10.3390/md12115425
Trindade-Silva, A.E., Rua, C.P.J., Andrade, B.G.N., et al., Polyketide synthase gene diversity within the microbiome of the sponge Arenosclera brasiliensis, endemic to the Southern Atlantic Ocean, Appl. Environ. Microbiol., 2013, vol. 79, no. 5, pp. 1598—1605. https://doi.org/10.1128/AEM.03354-12
Woodhouse, J.N., Fan, L., Brown, M.V., et al., Deep sequencing of non-ribosomal peptide synthases and polyketide synthases from the microbiomes of Australian marine sponges, ISME J., 2013, vol. 7, no. 9, pp. 1842—1851. https://doi.org/10.1038/ismej.2013.65
Santos, O.C., Soares, A.R., Machado, F.L., et al., Investigation of biotechnological potential of sponge-associated bacteria collected in Brazilian coast, Lett. Appl. Microbiol., 2015, vol. 60, no. 2, pp. 140—147. https://doi.org/10.1111/lam.12347
Borchert, E., Jackson, S.A., O’Gara, F., et al., Diversity of natural product biosynthetic genes in the microbiome of the deep sea sponges Inflatella pellicula, Poecillastra compressa, and Stelletta normani, Front. Microbiol., 2016, vol. 7. https://doi.org/10.3389/fmicb.2016.01027
Talevska, A., Pejin, B., Beric, T., et al., Further insight into the bioactivity of the freshwater sponge Ochridaspongia rotunda, Pharm. Biol., 2017, vol. 55, no. 1, pp. 1313—1316. https://doi.org/10.1080/13880209.2017.1297468
Talevska, A., Pejin, B., Kojic, V., et al., A contribution to pharmaceutical biology of freshwater sponges, Nat. Prod. Res., 2018, vol. 32, no. 5, pp. 568—571. https://doi.org/10.1080/14786419.2017.1315719
Keller-Costa, T., Jousset, A., van Overbeek, L., et al., The freshwater sponge Ephydatia fluviatilis harbours diverse Pseudomonas species (Gammaproteobacteria, Pseudomonadales) with broad-spectrum antimicrobial activity, PLoS One, 2014, vol. 9, no. 2. https://doi.org/10.1371/journal.pone.0088429
Laport, M.S., Pinheiro, U., and Rachid, C.T.C.D.C., Freshwater sponge Tubella variabilis presents richer microbiota than marine sponge species, Front. Microbiol., 2019, vol. 10. https://doi.org/10.3389/fmicb.2019.02799
Gernert, C., Glockner, F.O., Krohne, G., et al., Microbial diversity of the freshwater sponge Spongilla lacustris, Microb. Ecol., 2005, vol. 50, pp. 206—212. https://doi.org/10.1007/s00248-004-0172-x
Costa, R., Keller-Costa, T., Gomes, N.C.M., et al., Evidence for selective bacterial community structuring in the freshwater sponge Ephydatia fluviatilis, Microb. Ecol., 2013, vol. 65, pp. 232—244. https://doi.org/10.1007/s00248-012-0102-2
Gaikwad, S., Shouche, Y.S., and Gade, W.N., Microbial community structure of two freshwater sponges using Illumina MiSeq sequencing revealed high microbial diversity, AMB Express, 2016, vol. 6, p. 40. https://doi.org/10.1186/s13568-016-0211-2
Kaluzhnaya, O.V., Kulakova, N.V., and Itskovich, V.B., Diversity of polyketide synthase (PKS) genes in metagenomic community of freshwater sponge Lubomirskia baicalensis, Mol. Biol. (Moscow), 2012, vol. 46, no. 6, pp. 790—795.
Kaluzhnaya, O.V. and Itskovich, V.B., Distinctive features of the microbial diversity and the polyketide synthase genes spectrum in the community of the endemic Baikal sponge Swartschewskia papyracea, Russ. J. Genet., 2016, vol. 52, no. 1, pp. 38—48. https://doi.org/10.1134/S1022795416010099
Kaluzhnaya, O.V., Lipko, I.A., Itskovich, V.B., et al., PCR-screening of bacteria isolated from freshwater sponge Lubomirskia baicalensis for detection of genes of secondary metabolites, Voda: Khim. Ekol., 2013, no. 7, pp. 70—74.
Lipko, I.A., Kalyuzhnaya, O.V., Kravchenko, O.S., et al., Identification of polyketide synthase genes in genome of Pseudomonas fluorescens strain 28Bb-06 from freshwater sponge Baikalospongia bacillifera, Mol. Biol. (Moscow), 2012, vol. 46, no. 4, pp. 609—611. https://doi.org/10.1134/S0026893312040073
Efremova, S.M., Sponges, in Annotirovannyi spisok fauny ozera Baikal i ego vodosbornogo basseina (An Annotated List of the Fauna of Lake Baikal and Its Catchment Area), Timoshkin, O.A., Ed., Novosibirsk: Nauka, 2001, vol. 1, pp. 177—190.
Kaluzhnaya, O.V., Krivich, A.A., and Itskovich, V.B., Diversity of 16S rRNA genes in metagenomic community of the freshwater sponge Lubomirskia baicalensis, Russ. J. Genet., 2012, vol. 48, no. 8, pp. 855—858. https://doi.org/10.1134/S1022795412070058
Kaluzhnaya, O.V., Itskovich, V.B., and McCormack, G.P., Phylogenetic diversity of bacteria associated with the endemic freshwater sponge Lubomirskia baicalensis, World J. Microb. Biotech., 2011, vol. 27, no. 8, pp. 1955—1959. https://doi.org/10.1007/s11274-011-0654
Kaluzhnaya, O.V. and Itskovich, V.B., Phylogenetic diversity of microorganisms associated with the deepwater sponge Baikalospongia intermedia, Russ. J. Genet., 2014, vol. 50, no. 7, pp. 667—676. https://doi.org/10.1134/S1022795414060052
Gladkikh, A.S., Kaluzhnaya, O.V., Belykh, O.I., et al., Analysis of bacterial communities of two Lake Baikal endemic sponge species, Microbiology (Moscow), 2014, vol. 83, no. 6, pp. 787—797.
Seo, E.-Y., Jung, D., Belykh, O.I., et al., Comparison of bacterial diversity and species composition in three endemic Baikalian sponges, Ann. Limnol., 2016, vol. 52, pp. 27—32. https://doi.org/10.1051/limn/2015035
Kulakova, N., Sakirko, M., Adelshin, R., et al., Brown rot syndrome and changes in the bacterial community of the Baikal sponge Lubomirskia baicalensis, Microb. Ecol., 2018, vol. 75, no. 4, pp. 1024—1034. https://doi.org/10.1007/s00248-017-1097-5
Kozhov, M.M., Biologiya ozera Baikal (Biology of Lake Baikal), Moscow: Akad. Nauk SSSR, 1962.
Barrios-Llerena, M.E., Burja, A.M., and Wright, P.C., Genetic analysis of polyketide synthase and peptide synthetase genes in cyanobacteria as a mining tool for secondary metabolites, J. Ind. Microbiol. Biotechnol., 2007, vol. 34, pp. 443—456. https://doi.org/10.1007/s10295-007-0216-6
Chang, A.Y., Chau, V.W.Y., Landas, J.A., et al., Preparation of calcium competent Escherichia coli and heat-shock transformation, JEMI-Methods, 2017, vol. 1, pp. 22—25. https://jemi.microbiology.ubc.ca/node/127.
Maniatis, T., Fritsch, E.F., and Sambrook, J., Molecular Cloning: A Laboratory Manual, Cold Spring Harbor: Cold Spring Harbor Lab., 1982.
Hall, T.A., BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT, Nucleic Acids Symp., 1999, vol. 41, pp. 95—98.
Altschul, S.F., Warren, G., Miller, W., et al., Basic local alignment search tool, J. Mol. Biol., 1990, vol. 215, pp. 403—410. https://doi.org/10.1016/S0022-2836(05)80360-2
Tamura, K., Dudley, J., Nei, M., and Kumar, S., MEGA4: molecular genetics analysis (MEGA) software version 4.0, Mol. Biol. Evol., 2007, vol. 24, pp. 1596—1599. https://doi.org/10.1093/molbev/msm092
Kaluzhnaya, O. and Itskovich, V., Diversity of potential producers of bioactive metabolites having polyketide nature in the Baikal sponge community of Rezinkovia echinata, Limnol. Freshwater Biol., 2020, no. 3, pp. 423—428. https://doi.org/10.31951/2658-3518-2020-A-3-423
Fawley, K.P. and Fawley, M.W., Observations on the diversity and ecology of freshwater Nannochloropsis (Eustigmatophyceae), with descriptions of new taxa, Protist, 2007, vol. 158, no. 3, pp. 325—336. https://doi.org/10.1016/j.protis.2007.03.003
Fietz, S., Bleis, W., Hepperle, D., et al., First record of Nannochloropsis limnetica (Eustigmatophyceae) in the autotrophic phytoplankton from Lake Baikal, J. Phycol., 2005, vol. 41, no. 4, pp. 780—790. https://doi.org/10.1111/j.0022-3646.2005.04198.x
Tamburic, B., Szaby, M., Tran, N.-A.T., et al., Action spectra of oxygen production and chlorophyll a fluorescence in the green microalga Nannochloropsis oculata, Bioresour. Technol., 2014, vol. 169, pp. 320—327. https://doi.org/10.1016/j.biortech.2014.07.008
Zhang, R, Parniakov, O., Grimi, N., et al, Emerging techniques for cell disruption and extraction of valuable bio-molecules of microalgae Nannochloropsis sp., Bioprocess Biosyst. Eng., 2019, vol. 42, no. 2, pp. 173—186. https://doi.org/10.1007/s00449-018-2038-5
Hovde, B.T., Deodato, C.R., Hunsperger, H.M., et al., Genome sequence and transcriptome analyses of Chrysochromulina tobinii: metabolic tools for enhanced algal fitness in the prominent order Prymnesiales (Haptophyceae), PLoS Genet., 2015, vol. 11, no. 9. https://doi.org/10.1371/journal.pgen.1005469
Deodatoa, C., Barlowb, S.B., Hovdec, B.T., et al., Naked Chrysochromulina (Haptophyta) isolates from lake and river ecosystems: an electron microscopic comparison including new observations on the type species of this taxon, PLoS Genet., 2019, vol. 40. https://doi.org/10.1016/j.algal.2019.101492
Eikrem, W., Medlin, L. K., Henderiks, J., et al., Haptophyta, Handbook of the Protists, Archibald, J.M., Simpson, A.G.B., Slamovits, C.H., Eds., Cham: Springer, 2016. pp. 1—61. https://doi.org/10.1007/978-3-319-32669-6_38-1
Izmest’eva, L.R., Silow, E.A., and Litchman, E., Long-term dynamics of Lake Baikal pelagic phytoplankton under climate change, Inland Water Biol., 2011, vol. 4, pp. 301—307. https://doi.org/10.1134/S1995082911030102
Kaluzhnaya, O.V. and Itskovich, V.B., Phototrophic microorganisms in the symbiotic communities of Baikal sponges: diversity of psbA gene (encoding D1 protein of photosystem II) sequences, Mol. Biol. (Moscow), 2017, vol. 51, no. 3, pp. 372—378. https://doi.org/10.1134/S0026893317030086
Yi, Z., Berney, C., Hartikainen, H., et al., High-throughput sequencing of microbial eukaryotes in Lake Baikal reveals ecologically differentiated communities and novel evolutionary radiations, FEMS Microbiol. Ecol., 2017, vol. 93, no. 8. https://doi.org/10.1093/femsec/fix073
Cabello-Yeves, P.J., Zemskaya, T.I., Rosselli, R., et al., Genomes of novel microbial lineages assembled from the sub-ice waters of Lake Baikal, Appl. Environ. Microbiol., 2018, vol. 84, no. 1. https://doi.org/10.1128/AEM.02132-17
Zhang, H., Sekiguchi, Y., Hanada, S., et al., Gemmatimonas aurantiaca gen. nov., sp. nov., a Gram-negative, aerobic, polyphosphate-accumulating micro-organism, the first cultured representative of the new bacterial phylum Gemmatimonadetes phyl. nov., Int. J. Syst. Evol. Microbiol., 2003, vol. 53, pp. 1155—1163. https://doi.org/10.1099/ijs.0.02520-0
Zeng, Y., Selyanin, V., Lukes, M., et al., Characterization of the microaerophilic, bacteriochlorophyll a-containing bacterium Gemmatimonas phototrophica sp. nov., and emended descriptions of the genus Gemmatimonas and Gemmatimonas aurantiaca, Int. J. Syst. Evol. Microbiol., 2015, vol. 65, pp. 2410—2419. https://doi.org/10.1111/1758-2229.12363
Zeng, Y., Baumbach, J., Barbosa, E.G.V., et al., Metagenomic evidence for the presence of phototrophic Gemmatimonadetes bacteria in diverse environments, Environ. Microbiol. Rep., 2016, vol. 8, pp. 139–149. https://doi.org/10.1111/1758-2229.12363
Zeng, Y., Nupur, N., Wu, N., et al., Gemmatimonas groenlandica sp. nov. is an aerobic anoxygenic phototroph in the phylum Gemmatimonadetes, Front. Microbiol., 2021. https://doi.org/10.3389/fmicb.2020.606612
Parfenova, V.V., Gladkikh, A.S., and Belykh, O.I., Comparative analysis of biodiversity in the planktonic and biofilm bacterial communities in Lake Baikal, Microbiology (Moscow), 2013, vol. 82, no. 1, pp. 91—101. https://doi.org/10.7868/S0026365613010126
Hu, P., Lang, J., Wawrousek, K., et al., Draft genome sequence of Rubrivivax gelatinosus CBS, J. Bacteriol., 2012, vol. 194, no. 12, p. 3262. https://doi.org/10.1128/JB.00515-12
Nagashima, S., Kamimura, A., Shimizu, T., et al., Complete genome sequence of phototrophic betaproteobacterium Rubrivivax gelatinosus IL144, J. Bacteriol., 2012, vol. 194, no. 13, pp. 3541—3542. https://doi.org/10.1128/JB.00511-12
Kashkak, E.S., Gaisin, V.A., Dagurova, O.P., et al., Formation and functioning of the microbial mats in the Khoito-Gol mineral spring (Eastern Sayan), Izv. Samarsk. Nauchn. Tsentra Ross. Akad. Nauk, 2016, vol. 18, no. 2, pp. 397—402.
Hisada, T., Okamura, K., and Hiraishi, A., Isolation and characterization of phototrophic purple nonsulfur bacteria from Chloroflexus and cyanobacterial mats in hot springs, Microbes Environ., 2007, vol. 22, no. 4, pp. 405—411. https://doi.org/10.1264/jsme2.22.405
Hesselsoe, M., Boysen S., Iversen, N., et al., Degradation of organic pollutants by methane grown microbial consortia, Biodegradation, 2005, vol. 16, no. 5, pp. 435—448. https://doi.org/10.1007/s10532-004-4721-2
Sukhanova, E.V., Shtykova, Y.R., Suslova, M.Y., et al., Diversity and physiological and biochemical properties of heterotrophic bacteria isolated from Lake Baikal epilithic biofilms, Microbiology (Moscow), 2019, vol. 88, no. 3, pp. 324—334. https://doi.org/10.1134/S0026261719030147
Wang, Z., Li, W., Li, H., et al., Phylogenomics of Rhodocyclales and its distribution in wastewater treatment systems, Sci. Rep., 2020, vol. 10, no. 1. https://doi.org/10.1038/s41598-020-60723-x
Youssef, N.H., Farag, I.F., Rinke, C., et al., In silico analysis of the metabolic potential and niche specialization of candidate phylum “Latescibacteria” (WS3), PLoS One, 2015, vol. 10, no. 6. https://doi.org/10.1371/journal.pone.0127499
Farag, I.F., Youssef, N.H., Elshahed, M.S., Global distribution patterns and pangenomic diversity of the candidate phylum “Latescibacteria” (WS3), Appl. Environ. Microbiol., 2017, vol. 83, no. 10. https://doi.org/10.1128/AEM.00521-17
Li, Y., Naman, C.B., Alexander, K.L., et al., The chemistry, biochemistry and pharmacology of marine natural products from Leptolyngbya, a chemically endowed genus of Cyanobacteria, Mar. Drugs, 2020, vol. 18, no. 10. https://doi.org/10.3390/md18100508
Belykh, O.I., Tikhonova, I.V., Kuz’min, A.V., et al., Toxin-producing cyanobacteria in Lake Baikal and reservoirs of Baikal region, Teor. Prikl. Ekol., 2020, no. 1, pp. 21—27. https://doi.org/10.25750/1995-4301-2020-1-021-027
Kalyuzhnaya, O.V., Itskovich, V.B., and Kupchinskii, A.B., Molecular identification of cyanobacteria forming mucous fouling at the surface of endemic sponge Lubomirskia baicalensis, Voda: Khim. Ekol., 2017, no. 1, pp. 26—33.
Belykh, O.I., Tikhonova, I.V., Sorokovikova, E.G., et al., Picoplanctonic Cyanoprokaryota of the genera Synechococcus Nageli and Cyanobium Rippka et Cohen-Baz. from Lake Baikal, Russia, Al’gologiya, 2011, vol. 21, pp. 36—50.
Erwin, P.M., Lypez-Legentil, S., Gonzalez-Pech, R., et al., A specific mix of generalists: bacterial symbionts in Mediterranean Ircinia spp., FEMS Microb. Ecol., 2012, vol. 79, no. 3, pp. 619—637. https://doi.org/10.1111/j.1574-6941.2011.01243.x
Kaluzhnaya, O.V. and Itskovich, V.B., Bleaching of Baikalian sponge affects the taxonomic composition of symbiotic microorganisms, Russ. J. Genet., 2015, vol. 51, no. 11, pp. 1153—1157. https://doi.org/10.1134/S1022795415110071
Kulakova, N.V., Denikina, N.N., and Belikov, S.I., Diversity of bacterial photosymbionts in Lubomirskiidae sponges from Lake Baikal, Int. J. Biodiversity, 2014, vol. 2014. https://doi.org/10.1155/2014/152097
Kehr, J.-C., Picchi, D.G., Dittmann, E., et al., Natural product biosynthesis in cyanobacteria: a treasure trove of unique enzymes, Beilstein. J. Org. Chem., 2011, vol. 7, pp. 1622—1635. https://doi.org/10.3762/bjoc.7.191
Newton, R.J., Jones, S.E., Eiler, A., et al., A guide to the natural history of freshwater lake bacteria, Microb. Mol. Biol. Rev., 2011, vol. 75, no. 1, pp. 14—49.
Fuerst, J.A. and Sagulenko, E., Beyond the bacterium: planctomycetes challenge our concepts of microbial structure and function, Nat. Rev. Microbiol., 2011, vol. 9, no. 6, pp. 403—413. https://doi.org/10.1038/nrmicro2578
Wiegand, S., Jogler, M., and Jogler, C., On the maverick planctomycetes, FEMS Microbiol. Rev., 2018, vol. 42, no. 6, pp. 739—760. https://doi.org/10.1093/femsre/fuy029
Costa, R., Keller-Costa, T., Gomes, N.C.M., et al., Evidence for selective bacterial community structuring in the freshwater sponge Ephydatia fluviatilis, Microb. Ecol., 2013, vol. 65, pp. 232—244. https://doi.org/10.1007/s00248-012-0102-2
Gaikwad, S., Shouche, Y.S., and Gade, W.N., Microbial community structure of two freshwater sponges using Illumina MiSeq sequencing revealed high microbial diversity, AMB Express, 2016, vol. 6, no. 1. https://doi.org/10.1186/s13568-016-0211-2
Funding
This work was supported by the Russian Foundation for Basic Research and the Government of the Irkutsk Oblast as part of the project no. 20-44-380023 r_a (molecular biological research) and the state budget funding, project no. 0279-2021-0011 “Genomics of Symbiosis. Investigation of Interactions between the Host and Consortia of Microorganisms and Parasites” (provision of experimental equipment).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest. The authors declare they have no conflicts of interest.
Statement on the welfare of animals. All applicable international, national, and/or institutional guidelines for the care and use of animals have been followed.
Additional information
Translated by A. Lisenkova
Rights and permissions
About this article
Cite this article
Kaluzhnaya, O.V., Itskovich, V.B. Features of Diversity of Polyketide Synthase Genes in the Community of Freshwater Sponge Baikalospongia fungiformis. Russ J Genet 58, 336–346 (2022). https://doi.org/10.1134/S1022795422030061
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1022795422030061