Abstract
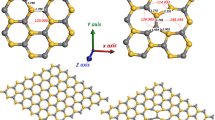
Computer-implemented cluster models have been devised for ∼2-nm anatase nanoparticles with exposed (001) and (100) faces. The Lewis acid sites occurring in these faces have been characterized by calculating the enthalpy of CO adsorption. In the Ti114O228 and Ti187O376H4 clusters, the corner oxygen atoms compensating the electric charge are bound to titanium atoms by double bonds with a length of approximately 1.7 Å, which is in agreement with experimental data. The average enthalpy of CO adsorption on the (001) and (100) faces at a zero coverage is −87.62 and −135.31 kJ/mol, respectively. The deviation from the average value is 20.2 and 8.8%, respectively. The average enthalpy of CO adsorption for the Ti114O228 cluster is −129.40 kJ/mol, and that for the Ti187O376H4 cluster is −119.79 kJ/mol.
Similar content being viewed by others
References
Sahu, N. and Parida, K.M., Kinet. Catal., 2012, vol. 53, no. 2, p. 197.
Murashkevich, A.N., Alisienok, O.A., and Zharskii, I.M., Kinet. Catal., 2011, vol. 52, no. 6, p. 809.
Vorontsov, A.V., Savinov, E.N., Barannik, G.B., Troitsky, V.N., and Parmon, V.N., Catal. Today, 1997, vol. 39, p. 207.
Zhang, F., Xie, F., Xu, H., Liu, J., and Oh, W.C., Kinet. Catal., 2013, vol. 54, no. 3, p. 297.
Yang, L., Taylor, R., Wad, J., and Hase, W.L., J. Phys. Chem. C, 2011, vol. 115, p. 12403.
Rusu, C.N. and Yates, J.T., J. Phys. Chem. B, 2000, vol. 104, p. 12292.
Lange, F., Hadjiivanov, K., Schmelz, H., and Knözinger, H., Catal. Lett., 1992, vol. 16, p. 97.
Nakajima, K., Noma, R., Kitano, M., and Hara, M., J. Mol. Catal. A: Chem. (in press). doi 10.1016/j.molcata.2013.09.012
Hadjiivanov, K., Saur, O., Lamotte, J., and Lavalley, J.C., Z. Phys. Chem., 1994, vol. 187, p. 281.
Martra, G., Appl. Catal., A, 2000, vol. 200, p. 275.
Couble, J., Gravejat, P., Gaillard, F., and Bianchi, D., Appl. Catal., A, 2009, vol. 371, p. 99.
Scaranto, J. and Giorgianni, S., Mol. Phys., 2009, vol. 107, p. 1997.
Homann, T., Bredow, T., and Jug, K., Surf. Sci., 2004, vol. 555, p. 135.
Zeng, W., Liu, T., Gou, Z., and Lin, L., Phys. E, 2012, vol. 44, p. 1567.
Scaranto, J. and Giorgianni, S., Mol. Simul., 2013, vol. 39, p. 245.
Tsipis, A. and Tsipis, C., Phys. Chem. Chem. Phys., 1999, vol. 1, p. 4453.
Tang, L., Sai, L., Zhao, J., and Qiu, R., J. Comput. Chem., 2012, vol. 33, p. 163.
Takahashi, H., Yuki, K., and Nitta, T., Fluid Phase Equilib., 2002, vol. 194, p. 153.
Onal, I., Soyer, S., and Senkan, S., Surf. Sci., 2006, vol. 600, p. 2457.
Mikheeva, E.P., Kachurovskaya, N.A., and Zhidomirov, G.M., Kinet. Catal., 2002, vol. 43, no. 2, p. 223.
Redfern, P.C., Zapol, P., Curtiss, L.A., Rajh, T., and Thurnauer, M.C., J. Phys. Chem. B, 2003, vol. 107, p. 11419.
Lee, C. and Aikens, C.M., Comput. Theor. Chem., 2013, vol. 1013, p. 32.
Qu, Z.-W. and Zhu, H., J. Comput. Chem., 2010, vol. 31, p. 2038.
Chen, M. and Dixon, D.A., J. Chem. Theory Comput., 2013, vol. 9, p. 3189.
Zavelev, D.E., Tsodikov, M.V., Zhidomirov, G.M., and Kozlovskii, R.A., Kinet. Catal., 2011, vol. 52, no. 5, p. 659.
Gurkan, Y.Y., Kasapbasi, E., and Cinar, Z., Chem. Eng. J., 2013, vol. 214, p. 34.
Wahab, H.S. and Koutselos, A.D., J. Mol. Model., 2009, vol. 15, p. 1237.
Bredow, T. and Jug, K., J. Phys. Chem., 1995, vol. 99, p. 285.
Stewart, J.J.P., J. Mol. Model., 2007, vol. 13, p. 1173.
Chen, L.X., Rajh, T., Jager, W., Nedeljkovic, J., and Thurnauer, M.C., J. Synchrotron Radiat., 1999, vol. 6, p. 445.
Chen, L.X., Rajh, T., Wang, Z., and Thurnauer, M.C., J. Phys. Chem. B, 1997, vol. 101, p. 10688.
Paukshtis, E.A., Opticheskaya spektroskopiya v adsorbtsii i katalize: Primenenie IK-spektroskopii (Optical Spectroscopy in Adsorption and Catalysis: Use of IR Spectroscopy), Novosibirsk: Inst. Kataliza, 2010.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Vorontsov, D.E. Tsydenov, 2014, published in Kinetika i Kataliz, 2014, Vol. 55, No. 4, pp. 430–437.
Rights and permissions
About this article
Cite this article
Vorontsov, A.V., Tsydenov, D.E. Arrangement of acid sites on the surfaces of anatase titanium dioxide nanoparticles according to cluster models. Kinet Catal 55, 409–415 (2014). https://doi.org/10.1134/S0023158414040156
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158414040156