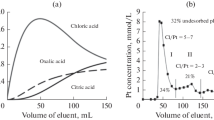
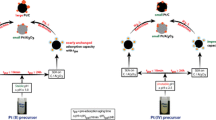
Abstract
Gradient elution experiments have revealed the difference between platinum complexes in terms of the strength of their interaction with the alumina surface. A considerable part (30–40%) of the supported platinum is nondesorbable via competitive ion exchange reactions or upon changes in the charge state of the support surface. The surface platinum complexes can be divided into ion-exchangeable and coordinatively fixed species according to the nature of their bonding with aluminum oxide. Combining desorption and spectroscopic methods (EXAFS and diffuse reflectance spectroscopy) has made it possible to characterize the surface complexes. The strongest metal-support interaction takes place in the fixation of hydrolyzed platinum species.
Similar content being viewed by others
References
Brunelle, J.P., Pure Appl. Chem., 1978, vol. 50, p. 1211.
Heise, F.J. and Schwarz, J.A., J. Colloid Interface Sci., 1986, vol. 113, p. 55.
Shah, A. and Regalbuto, J.R., Langmuir, 1994, vol. 10, p. 500.
Regalbuto, J.R., Navada, A., Shadid, S., Bricker, M.L., and Chen, Q., J. Catal., 1999, vol. 184, p. 335.
Spieker, W.A. and Regalbuto, J.R., Chem. Eng. Sci., 2000, vol. 56, p. 2365.
Agashe, K. and Regalbuto, J.R., J. Colloid Interface Sci., 1997, vol. 185, p. 174.
Spieker, W.A., Liu, J., Miller, J.T., Kropf, A.J., and Regalbuto, J.R., Appl. Catal., A, 2002, vol. 232, p. 219.
Spieker, W.A., Liu, J., Hao, X., Miller, J.T., Kropf, A.J., and Regalbuto, J.R., Appl. Catal., A, 2003, vol. 243, p. 53.
Mang, T., Breitscheidel, B., Polanek, P., and Knozinger, H., Appl. Catal., A, 1993, vol. 106, p. 239.
Boitiaux, J.P., Deves, J.M., Didillon, B., and Marcilly, C.R., in Catalytic Naphtha Reforming: Science and Technology, New York: Marcel Dekker, 1995, p. 79.
Shelimov, B., Lambert, J.-F., Che, M., and Didillon, B., J. Am. Chem. Soc., 1999, vol. 121, p. 545.
Shelimov, B., Lambert, J.-F., and Didillon, B., J. Mol. Catal., 2000, vol. 158, p. 91.
Santacesaria, E., Carra, S., and Adami, J., Ind. Eng. Chem. Prod. Res. Dev., 1977, vol. 16, no. 1, p. 41.
Maatman, R.W., Manaffy, P., Hoekstra, P., and Addink, C., J. Catal., 1971, vol. 23, p. 105.
Tsymbal, T.V., Doronin, V.P., Al’t, L.Ya., and Duplyakin, V.K., V Mezhdunar. Simp. po svyazi mezhdu gomogennym i geterogennym katalizom (V Int. Symp. on the Relation between Homogeneous and Heterogeneous Catalyses), Novosibirsk, 1986, vol. 2, part 2, p. 108.
Kireeva, T.V., Doronin, V.P., Alt, L.Ya., and Duplyakin, V.K., React. Kinet. Catal. Lett., 1987, vol. 34, no. 2, p. 261.
Morterra, C. and Magnacca, G., Catal. Today, 1996, vol. 27, p. 497.
Knozinger, H. and Ratnasamy, P., Catal. Rev. Sci. Eng., 1978, vol. 17, p. 31.
Tsyganenko, A.A. and Mardilovich, P.P., J. Chem. Soc., Faraday Trans., 1996, vol. 92, p. 4843.
Ginzburg, S.I., Gladyshevskaya, K.A., Ezerskaya, N.A., et al., Rukovodstvo po khimicheskomu analizu platinovykh metallov i zolota (Guide to the Chemical Analysis of the Platinum Metals and Gold), Moscow: Nauka, 1965.
Rieman, W. and Walton, H., Ion Exchange in Analytical Chemistry, Oxford: Pergamon, 1970.
Samuelson, O., Ion Exchange Separations in Analytical Chemistry, New York: Wiley, 1963.
Duplyakin, V.K., Fenelonov, V.B., Rikhter, K., Rodionov, A.V., Seflut, Kh., Kheifets, L.I., Neimark, A.V., and Moskovtsev, V.V., in Nauchnye osnovy tekhnologii katalizatorov (Scientific Foundations of Catalyst Technology), Novosibirsk, 1981, issue 13, p. 137.
Charlot, G., Les méthodes de la chimie analytique, Paris: Masson, 1961.
Cox, L.E., Peters, D.G., and Wehry, E.L., J. Inorg. Nucl. Chem., 1972, vol. 34, p. 297.
Zolotov, Yu.A., Varshal, G.M., and Ivanov, V.M., Analiticheskaya khimiya metallov platinovoi gruppy (Analytical Chemistry of the Platinum Metals), Moscow: KomKnig, 2005.
Lever, A.B.P., Inorganic Electronic Spectroscopy, Amsterdam: Elsevier, 1984.
Buslaeva, T.M., Umreiko, D.S., Novitskii, G.G., et al., Khimiya i spektroskopiya galogenidov platinovykh metallov (Chemistry and Spectroscopy of Platinum Metal Halides), Minsk: Universitetskoe, 1990.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © O.B. Bel’skaya, R.Kh. Karymova, D.I. Kochubey, V.K. Duplyakin, 2008, published in Kinetika i Kataliz, 2008, Vol. 49, No. 5, pp. 754–763.
Rights and permissions
About this article
Cite this article
Bel’skaya, O.B., Karymova, R.K., Kochubey, D.I. et al. Genesis of the active-component precursor in the synthesis of Pt/Al2O3 catalysts: I. Transformation of the [PtCl6]2− complex in the interaction between chloroplatinic acid and the γ-Al2O3 surface. Kinet Catal 49, 720–728 (2008). https://doi.org/10.1134/S0023158408050170
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158408050170