Abstract
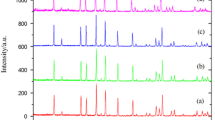
Crystalline LiNi1/3Co1/3Mn1/3O2 powders have been synthesized by two different procedures, using carbonate coprecipitation from sulfate and nitrate solutions, followed by two-step heat treatment of a mixture of the resultant Ni1/3Co1/3Mn1/3CO3 precursor and Li2CO3 at 500 and 900°C. The powders have been characterized by X-ray diffraction, scanning electron microscopy, and dynamic light scattering. The results demonstrate that the synthesized compounds have a hexagonally ordered, layered structure of the α-NaFeO2 type. The primary-particle (crystallite) size in the powders is 50 nm and the aggregate size is 150–250 nm. The average size of larger structures (agglomerates) is 11 and 18 μm in the powders prepared via the sulfate and nitrate routes, respectively. The chemical stability of the synthesized powders is shown to depend on the ambient medium. Prolonged storage in air leads to the formation of new, lithium-deficient phases, especially in the case of the powders prepared from nitrate solutions.
Similar content being viewed by others
References
He, P., Wang, H., Qi Lu, and Osaka, T., Electrochemical Characteristics of Layered LiNi1/3Co1/3Mn1/3O2 with Different Synthesis Conditions, Power Sources, 2006, vol. 160, pp. 627–632.
Perasamy, P., Kalaiselvi, N., and Kim, H.S., High Voltage and High Capacity Characteristics of LiNi1/3Co1/3Mn1/3O2 Cathode for Lithium Battery Applications, Int. J. Electrochem. Sci., 2007, vol. 2, pp. 689–699.
Ngala, J.K., Chernova, N.A., Ma, M., et al., The Synthesis, Characterization and Electrochemical Behavior of the Layered LiNi0.4Mn0.4Co0.2O2, J. Mater. Chem., 2004, vol. 14, pp. 214–220.
Tran, N., Croguennec, L., Jordy, C., et al., Influence of the Synthesis Route on the Electrochemical Properties of LiNi0.425Mn0.425Co0.15O2, Solid State Ionics, 2005, vol. 176, pp. 1539–1574.
Kosova, N.V., Devyatkina, E.T., and Kaichev, V.V., LiNi1 − x − y CoxMnyO2 (x = y = 0.1, 0.2, 0.33) Cathode Materials Prepared Using Mechanical Activation: Structure, State of Ions, and Electrochemical Performance, Inorg. Mater., 2007, vol. 43, no. 2, pp. 185–193.
Sclar, H., Kovacheva, D., Zhecheva, E., et al., On the Performance of Lithium-Ion Batteries, J. Electrochem. Soc., 2009, vol. 156, no. 11, pp. 938–948.
Zhuang, G., Chen, G., Shim, J., et al., Li2CO3 in LiNi0.8Co0.15Al0.05O2 Cathodes and Its Effects on Capacity and Power, J. Power Sources, 2004, vol. 134, pp. 293–297.
Shizuka, K., Kiyohara, C., Shima, K., and Takeda, Y., Effect of CO2 on Layered Li1 − z Ni1 − x − y MyO2 (M = Al, Mn) Cathode Materials for Lithium Ion Batteries, J. Power Sources, 2007, vol. 166, pp. 233–238.
Antolini, E., LiCoO2: Formation, Structure, Lithium and Oxygen Nonstoichiometry, Electrochemical Behavior and Transport Properties, Solid State Ionics, 2004, vol. 170, nos. 3–4, pp. 159–178.
Rossen, E., Reimers, J.N., and Dahn, J.R., Synthesis and Electrochemistry of Spinel LT-LiCoO2, Solid State Ionics, 1993, vol. 62, pp. 53–60.
Ohzuku, T. and Makimura, Y., Layered Lithium Insertion Material of LiNi1/3Co1/3Mn1/3O2 for Lithium-Ion Batteries, Chem. Lett., 2001, vol. 30, no. 7, pp. 642–643.
Reimers, J.N., Rossen, E., Jones, C.D., and Dahn, J.R., Structure and Electrochemistry of LixFeyNi1 − y O2, Solid State Ionics, 1993, vol. 61, p. 335.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Titov, Z.V. Eremenko, E.G. Goryacheva, N.P. Sokolova, N.L. Opolchenova, N.N. Stepareva, G.P. Korobko, 2013, published in Neorganicheskie Materialy, 2013, Vol. 49, No. 2, pp. 198–205.
Rights and permissions
About this article
Cite this article
Titov, A.A., Eremenko, Z.V., Goryacheva, E.G. et al. Synthesis, structure, and some properties of LiNi1/3Co1/3Mn1/3O2 . Inorg Mater 49, 202–208 (2013). https://doi.org/10.1134/S0020168513020222
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0020168513020222