Abstract
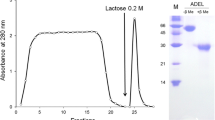
A lectin was purified from Japanese sea hare Aplysia kurodai by lactosyl-agarose affinity chromatography. The molecular mass of the lectin was determined to be 56 and 32 kDa by SDS-PAGE under non-reducing and reducing conditions, respectively. It was found to agglutinate trypsinized and glutaraldehyde-fixed rabbit and human erythrocytes in the absence of divalent cations. The lectin exhibited stable thermo-tolerance as it retained hemagglutinating activity for 1 h even at 80°C and showed stability at pH 10. By contrast, it was very sensitive at pH less than 5 and in the presence of the sulfhydryl-group preserving reagent, β-mercaptoethanol. The hemagglutinating activity by the lectin was specifically inhibited by D-galactose, galacturonic acid, methyl-α- and methyl-β-D-galactopyranoside, lactose, melibiose, and asialofetuin. The association rate constant (k ass) and dissociation rate constant (k diss) were determined for the lectin to be 4.3·105 M−1·sec−1 and 2.2·10−3 sec−1, respectively, using a surface plasmon resonance biosensor. The lectin moderately inhibited cell proliferation in the P388 cell line dose dependently. Interestingly, lectin-treated cells did not show a fragmented DNA ladder as is caused by apoptosis, suggesting that the cell proliferation inhibition was caused by another unknown mechanism.
Similar content being viewed by others
Abbreviations
- AGL:
-
Aplysia depilans gonad lectin
- AKL:
-
Aplysia kurodai egg lectin
- BCA:
-
bicinchoninic acid
- BSA:
-
bovine serum albumin
- Con A:
-
concanavalin A
- EDC:
-
1-ethyl-3-(3-dimethylamino-propyl)carbodiimide
- HOL:
-
Halichondria okadai lectin
- NHS:
-
N-hydroxysuccinimide
- RCA:
-
Ricin communis agglutinin
- SBL:
-
sialic acid binding lectin
- SPR:
-
surface plasmon resonance
- TBS:
-
Tris-buffered saline containing 150 mM NaCl, pH 7.4
- WGA:
-
wheat germ agglutinin
References
Ozeki, Y., Matsui, T., Nitta, K., Kawauchi, H., Takayanagi, Y., and Titani, K. (1991) Biochem. Biophys. Res. Commun., 178, 407–413.
Ozeki, Y., Matsui, T., Suzuki, M., and Titani, K. (1991) Biochemistry, 30, 2391–2394.
Hosono, M., Ishikawa, K., Mineki, R., Murayama, K., Numata, C., Ogawa, Y., Takayanagi, Y., and Nitta, K. (1999) Biochim. Biophys. Acta, 1472, 668–675.
Titani, K., Takio, K., Kuwada, M., Nitta, K., Sakakibara, F., Kawauchi, H., Takayanagi, G., and Hakomori, S.-I. (1987) Biochemistry, 26, 2189–2194.
Melo, V. M. M., Duarte, A. B. G., Carvalho, A. F. F. U., Siebra, E. A., and Vasconcelos, I. M. (2000) Toxicon, 38, 1415–1427.
Banerjee, S., Chaki, S., Bhowal, J., and Chatterjee, B. P. (2004) Arch. Biochem. Biophys., 421, 125–134.
Kamiya, H., and Shimizu, Y. (1981) Bull. Japan Soc. Sci. Fish., 47, 255–259.
Ozeki, Y. (1998) Mol. Biol. Int., 45, 989–995.
Gilboa-Garber, N., Susswein, A. J., Mizrahi, L., and Avichezer, D. (1985) FEBS Lett., 181, 267–270.
Zipris, D., Gilboa-Garber, N., and Susswein, A. J. (1986) Microbios., 46, 193–198.
Gilboa-Garber, N., Sudakevitz, D., Levene, C., Rahimi-Levene, N., and Yahalom, V. (2006) Immunohematology, 22, 15–22.
Gilboa-Garber, N., and Sudakevitz, D. (2001) FEMS Immunol. Med. Microbiol., 30, 235–240.
Wils, M. P., Garrow, G. M., and Levitan, I. B. (1992) J. Neurobiol., 23, 739–750.
Nitta, K., Ozaki, K., Ishikawa, M., Furusawa, S., Hosono, M., Kawauchi, H., Sasaki, K., Takayanagi, Y., Tsuiki, S., and Hakomori, S.-I. (1994) Cancer Res., 54, 920–927.
Laemmli, U. K. (1970) Nature, 227, 680–685.
Smith, P. K., Krohn, R. I., Hermanson, G. T., Mallia, A. K., Gartner, F. H., Provenzano, M. D., Fujimoto, E. K., Goeke, N. M., Olson, B. J., and Klenk, D. C. (1985) Anal. Biochem., 150, 76–85.
Wiechelman, K. J., Braun, R. D., and Fitzpatrick, J. D. (1988) Anal. Biochem., 175, 231–237.
Dubois, M., Gilles, K. A., Hamilton, J. K., Rebers, P. A., and Smith, F. (1956) Anal. Chem., 28, 350–356.
Kawsar, S. M. A., Fujii, Y., Matsumoto, R., Ichikawa, T., Tateno, H., Hirabayashi, J., Yasumitusu, H., Dogasaki, C., Hosono, M., Nitta, K., Hamako, J., Matsui, T., and Ozeki, Y. (2008) Comp. Biochem. Physiol., 150B, 349–357.
Gourdine, J.-P., Cioci, G., Miguet, L., Unverzagt, C., Silva, D. V., Varrot, A., Gautier, C., Smith-Ravin, E. J., and Imberty, A. (2008) J. Biol. Chem., 283, 30112–30120.
Shinohara, Y., Kim, F., Shimizu, M., Goto, M., Tosu, M., and Hasegawa, Y. (1994) Eur. J. Biochem., 223, 189–194.
Ahmed, S. A., Gogal, R. M., Jr., and Walsh, J. E. (1994) J. Immun. Meth., 170, 211–224.
Kawagishi, H., Yamawaki, M., Isobe, S., Usui, T., Kimura, A., and Chiba, S. (1994) J. Biol. Chem., 269, 1375–1379.
Wu, A. M., Song, S. C., Chen, Y. Y., and Gilboa-Garber, N. (2000) J. Biol. Chem., 275, 14017–14024.
Pashov, A., MacLeod, S., Saha Rinku, Perry, M., van Cott, T. C., and Kieber-Emmons, T. (2005) Glycobiology, 15, 994–1001.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Biokhimiya, 2009, Vol. 74, No. 7, pp. 877–885.
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM08-299, April 12, 2009.
Rights and permissions
About this article
Cite this article
Kawsar, S.M.A., Matsumoto, R., Fujii, Y. et al. Purification and biochemical characterization of a D-galactose binding lectin from Japanese sea hare (Aplysia kurodai) eggs. Biochemistry Moscow 74, 709–716 (2009). https://doi.org/10.1134/S0006297909070025
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297909070025