Abstract
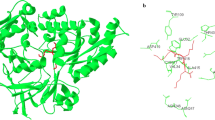
A low-molecular-weight cationic protein that can bind human and rabbit immunoglobulins G has been isolated from Yersinia pseudotuberculosis cells. This immunoglobulin binding protein (IBP) interacts with IgG Fc-fragment, the association constant of the resulting complex being 3.1 μM−1. MALDI-TOF mass spectrometry analysis of IBP revealed its molecular mass of 16.1 kDa, and capillary isoelectrofocusing analysis showed pI value of 9.2. N-Terminal sequence determination by Edman degradation revealed the sequence of the 15 terminal amino acid residues (ADKIAIVNVSSIFQ). Tryptic hydrolysate of IBP was subjected to MALDI-TOF mass spectrometry for proteolytic peptide profiling. Based on the peptide fingerprint, molecular mass, pI, and N-terminal sequence and using bioinformatic resources, IBP was identified as Y. pseudotuberculosis periplasmic chaperone Skp. Using the method of comparative modeling a spatial model of Skp has been built. This model was then used for modeling of Skp complexes with human IgG1 Fc-fragment by means of molecular docking.
Similar content being viewed by others
Abbreviations
- IBP:
-
immunoglobulin binding protein
- Skp:
-
seventeen kilodalton protein
References
Thome, B., Hoffschulte, H., Schiltz, E., and Muller, M. (1990) FEBS Lett., 269, 113–116.
Thome, B., and Muller, M. (1991) Mol. Microbiol., 5, 2815–2821.
Chen, R., and Henning, U. (1996) Mol. Microbiol., 19, 1287–1294.
De Cock, H., Schafer, U., Potgeter, M., Demel, R., Muller, M., and Tommassen, J. (1999) Eur. J. Biochem., 259, 96–103.
Schafer, U., Beck, K., and Muller, M. (1999) J. Biol. Chem., 274, 24567–24574.
Qu, J., Mayer, C., Behrens, S., Holst, O., and Kleinschmidt, J. (2007) J. Mol. Biol., 374, 91–105.
Schlapschy, M., Dommel, M., Hadian, K., Fogarasi, M., Korndorfer, I., and Skerra, A. (2004) Biol. Chem., 385, 137–143.
Bulieris, P., Behrens, S., Holst, O., and Kleinschmidt, J. H. (2003) J. Biol. Chem., 278, 9092–9099.
Geyer, R., Galanos, C., Westphal, O., and Golecki, J. (1979) Eur. J. Biochem., 98, 27–38.
Holck, A., and Kleppe, K. (1988) Gene, 67, 117–124.
Holck, A., Lossius, I., Aasland, R., and Kleppe, K. (1987) Biochim. Biophys. Acta, 914, 49–54.
Holck, A., Lossius, I., Aasland, R., Haarr, L., and Kleppe, K. (1987) Biochim. Biophys. Acta, 908, 188–199.
Koski, P., Rhen, M., Kantele, J., and Vaara, M. (1989) J. Biol. Chem., 264, 18973–18980.
Koski, P., Hirvas, L., and Vaara, M. (1990) Gene, 88, 117–120.
Chain, P. S. G., Carniel, E., Larimer, F. W., Lamerdin, J., Stoutland, P. O., Regala, W. M., Georgescu, A. M., Vergez, L. M., Land, M. L., Motin, V. L., Brubaker, R. R., Fowler, J., Hinnebusch, J., Marceau, M., Medigue, C., Simonet, M., Chenal-Francisque, V., Souza, B., Dacheux, D., Elliott, J. M., Derbise, A., Hauser, L. J., and Garcia, E. (2004) Proc. Natl. Acad. Sci. USA, 101, 13826–13831.
Vuorio, R., Hirvas, L., Raybourne, R. B., Yu, D. T. Y., and Vaara, M. (1991) Biochim. Biophys. Acta, 1129, 124–126.
Shrestha, A., Shi, L., Tanase, S., Tsukamoto, M., Nishino, N., Tokita, K., and Yamamoto, T. (2004) Am. J. Pathol., 164, 763–772.
Ezepchuk, Yu. V. (1977) in Biomolecular Bases of Bacterial Pathogenicity [in Russian], Nauka, Moscow, pp. 56–63.
Widders, P. R. (1991) in Bacterial Immunoglobulin-Binding Proteins: Microbiology, Chemistry, and Biology (Boyle, M. D. P., ed.) Academic Press Inc., San Diego, pp. 375–396.
Sidorin, E. V., Kim, N. Yu., Leichenko, E. V., Anastyuk, S. D., Dmitrenok, P. S., Nabereznykh, G. A., and Solov’eva, T. F. (2006) Biochemistry (Moscow), 71, 1278–1283.
Nabereznykh, G. A., Sidorin, E. V., Lapshina, L. A., Reunov, A. V., and Solov’eva, T. F. (2006) Biochemistry (Moscow), 71, 1284–1288.
Ovodov, Yu. S., Gorshkova, R. P., and Tomshich, S. V. (1971) Imunochemistry, 8, 1071–1079.
Laemmli, U. K. (1970) Nature, 2, 680–685.
Gasparov, V. C., and Degtyar’, V. G. (1994) Biochemistry (Moscow), 59, 563–572.
Pinchuk, L. M. (1971) Zh. Mikrobiol. Epidemiol. Immunobiol., 7, 26.
Kilar, F., Vegvari, A., and Mod, A. (1998) J. Chromatogr., 813, 349–360.
Rappsilber, J., Ishihama, Y., and Mann, M. (2003) Anal. Chem., 75, 663–670.
Mikhailov, A. T., and Simirskii, V. N. (1991) in Immunochemical Analytical Methods in Developmental Biology [in Russian], Nauka, Moscow, pp. 155–192.
Labbe, S., and Grenier, D. (1995) Infect. Immun., 63, 2785–2789.
Catty, D., and Raykundalia, C. (1991) in Antibodies. Methods (Catty, D., ed.) [Russian translation], Mir, Moscow, pp. 180–181.
Egorov, A. M., Osipov, A. P., Dzantiev, B. B., and Gavrilova, E. M. (1991) in Theory and Practice of Immunoenzyme Analysis [in Russian], Vysshaya Shkola, Moscow, pp. 182–183.
Bobrovnik, S. A. (2004) Ukr. Biokhim. Zh., 76, 5–28.
Guex, N., and Peitsch, M. C. (1997) Electrophoresis, 18, 2714–2723.
Arnold, K., Bordoli, L., Kopp, J., and Schwede, T. (2006) Bioinformatics, 22, 195–201.
Schwede, T., Kopp, J., Guex, N., and Peitsch, M. C. (2003) Nucleic Acids Res., 31, 3381–3385.
Kopp, J., and Schwede, T. (2004) Nucleic Acids Res., 32, D230–D234.
Walton, T. A., and Sousa, M. C. (2004) Mol. Cell, 15, 367–374.
Berman, H. M., Westbrook, J., Feng, Z., Gilliland, G., Bhat, T. N., Weissig, H., Shindyalov, I. N., and Bourne, P. E. (2000) Nucleic Acids Res., 28, 235–242.
Lindahl, E., Hess, B., and Spoel, D. (2001) J. Mol. Mod., 7, 306–317.
Katchalski-Katzir, E., Shariv, I., Eisenstein, M., Friesem, A. A., Aflalo, C., and Vakser, I. A. (1992) Proc. Natl. Acad. Sci. USA, 89, 2195–2199.
Padlan, E. A. (1994) Mol. Immunol., 31, 169–217.
Langone, J. (1982) Adv. Immunol., 32, 157–241.
Lewis, M. J., Meehan, M., Owen, P., and Woof, J. M. (2008) J. Biol. Chem., 283, 17615–17623.
Meininger, D. P., Rance, M., Starovasnik, A., Fairbrother, W. J., and Skelton, N. J. (2000) Biochemistry, 39, 26–36.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E. V. Sidorin, R. H. Ziganshin, G. A. Naberezhnykh, G. N. Likhatskaya, E. V. Trifonov, S. D. Anastiuk, O. V. Chernikov, T. F. Solov’eva, 2009, published in Biokhimiya, 2009, Vol. 74, No. 4, pp. 501–514.
Originally published in Biochemistry (Moscow) On-Line Papers in Press, as Manuscript BM08-262, February 8, 2009.
Rights and permissions
About this article
Cite this article
Sidorin, E.V., Ziganshin, R.H., Naberezhnykh, G.A. et al. Chaperone Skp from Yersinia pseudotuberculosis exhibits immunoglobulin G binding ability. Biochemistry Moscow 74, 406–415 (2009). https://doi.org/10.1134/S0006297909040087
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297909040087