Abstract
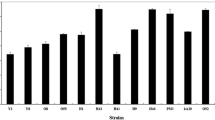
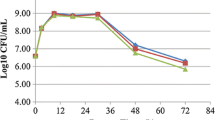
Lactic acid fermentation of seaweed is a recent topic and quite limited information is available on culture conditions. To know the suitable strains for use as a starter culture for seaweed fermentation, 14 lactic acid bacteria (LAB) strains, including 11 species, were tested in culture conditions prepared with or without salt. A commercial product of Undaria pinnatifida powder was used as a substrate for fermentation without sterilizing. Starter-suitability of the LAB strains was assessed from their predominance after culture. Among the tested strains, Lactobacillus brevis, Lactobacillus plantarum, Lactobacillus casei and Lactobacillus rhamnosus showed high (>90%) predominance in their cultures, while control cultures prepared without inoculation of LAB did not show any detectable growth of acid producing bacteria and spoiled. A total of 102 strains not showing acid producing activity were isolated and characterized from spoiled cultures, and all the isolates were observed as Bacillus strains, including 64 strains (62.7%) and 16 strains (15.7%) of a Bacillus cereus-related and B. fusiformis-related species, respectively. The Undaria substrate before fermentation contained culturable microorganisms at 1.4−3.1 × 102 CFU/g, but the Bacillus cereus-related strain was not a major composition, suggesting a concern of selective growth of the Bacillus cereus-related strain during the spoiled fermentation.
Similar content being viewed by others
References
Steinkraus KH. Comparison of fermented foods of the East and West. In: Lee C, Steinkraus KH, Reilly PJA (eds). Fish Fermentation Technology. United Nations University Press, Tokyo. 1993; 1–12.
Uchida M, Murata M. Fermentative preparation of single cell detritus from seaweed, Undaria pinnatifida, suitable as a replacement hatchery diet for unicellular algae. Aquaculture 2002; 207: 345–357.
Uchida M, Murata M. Isolation of a lactic acid bacterium and yeast consortium from a fermented material of Ulva spp. (Chlorophyta). J. Appl. Microbiol. 2004; 97: 1297–1310.
Uchida M, Numaguchi K, Murata M. Mass preparation of marine silage from Undaria pinnatifida and its dietary effect for young pearl oyster. Fish. Sci. 2004; 70: 456–462.
Uchida M, Amakasu H, Satoh Y, Murata M. Combinations of lactic acid bacteria and yeast suitable for preparation of marine silage. Fish. Sci. 2004; 70: 508–518.
Uchida M, Sato Y, Murata M, Matsushima R. Characterization of the base collection strains preserved in the Marine Microorganisms Division, a sub-bank of the Microbial Culture Collections, Fish Research Agency. Bull. Fish. Res. Agent. 2004; 11: 19–30.
Weisburg WG, Barns SM, Pelletier DA, Lane DJ. 16S ribosomal DNA amplification for phylogenic study. J. Bacteriol. 1991; 173: 697–703.
Dunnett CW. A multiple comparison procedure for comparing several treatments with a control. J. Am. Stat. Assoc. 1955; 50: 1096–1211.
Thompson JD, Higgins DG, Gibson TJ, Clustal W. Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994; 22: 4673–4680.
Kimura M. A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J. Mol. Biol. 1980; 16: 111–120.
Saitou N, Nei M. The neighbor-joining method: a new method for reconstructing phylogenic trees. Mol. Biol. Evol. 1987; 4: 406–425.
Nes IF, Diep DB, Hovarstein LS, Brurberg MB, Eijsink VGH, Holo H. Biosynthesis of bacteriocins in lactic acid bacteria. Antonie Leeuwenhoek 1996; 70: 113–128.
Japan Collection of Microorganisms. Catalog of Strains, 9th edn. Riken Bioresource Center, Tsukuba. 2005.
Rogosa M, Genus I. Lactobacillus. In: Buchnan RE, Gibbons NE (eds) Bergey’s Manual of Determinate Bacteriology, 8th edn. Williams & Wilkins, Baltimore, 1975; 576–593.
Porter KG, Feig YS. The use of DAPI for identifying and counting aquatic microflora. Limnol. Oceanogr. 1980; 5: 943–948.
Shinagawa K, Sugiyama J, Terada T, Matsusaka N, Sugii S, Improved method for purification of an enterotoxin produced by bacillus cereus. FEMS Microbiol. Lett. 1991; 64: 1–5.
Asano S, Nukumizu Y, Bando H, Iizuka T, Yamamoto T. Cloning of novel enterotoxin genes from Bacillus cereus and Bacillus thuringiensis. Appl. Environ. Microbiol. 1997; 63: 1054–1057.
Mantynen V, Lindstrom K. A rapid PCR-based DNA test for enterotoxic Bacillus cereus. Appl. Environ. Microbiol. 1998; 64: 1634–1639.
Nakamura LK. Phylogeny of Bacillus sphaericus-like organisms. Int. J. Syst. Evol. Microbiol. 2000; 50: 1715–1722.
Skerman VBD, McGowan V, Sneath PHA. Approved lists of bacterial names. Int. J. Syst. Bacteriol. 1980; 30: 225–420.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Uchida, M., Murata, M. & Ishikawa, F. Lactic acid bacteria effective for regulating the growth of contaminant bacteria during the fermentation of Undaria pinnatifida (Phaeophyta). Fish Sci 73, 694–704 (2007). https://doi.org/10.1111/j.1444-2906.2007.01383.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1444-2906.2007.01383.x