-
PDF
- Split View
-
Views
-
Cite
Cite
Lauren Hennelly, Bilal Habib, Holly Root-Gutteridge, Vicente Palacios, Daniela Passilongo, Howl variation across Himalayan, North African, Indian, and Holarctic wolf clades: tracing divergence in the world’s oldest wolf lineages using acoustics, Current Zoology, Volume 63, Issue 3, June 2017, Pages 341–348, https://doi.org/10.1093/cz/zox001
Close - Share Icon Share
Abstract
Vocal divergence within species often corresponds to morphological, environmental, and genetic differences between populations. Wolf howls are long-range signals that encode individual, group, and subspecies differences, yet the factors that may drive this variation are poorly understood. Furthermore, the taxonomic division within the Canis genus remains contended and additional data are required to clarify the position of the Himalayan, North African, and Indian wolves within Canis lupus. We recorded 451 howls from the 3 most basal wolf lineages—Himalayan C. lupus chanco—Himalayan haplotype, North African C. lupus lupaster, and Indian C. lupus pallipes wolves—and present a howl acoustic description within each clade. With an additional 619 howls from 7 Holarctic subspecies, we used a random forest classifier and principal component analysis on 9 acoustic parameters to assess whether Himalayan, North African, and Indian wolf howls exhibit acoustic differences compared to each other and Holarctic wolf howls. Generally, both the North African and Indian wolf howls exhibited high mean fundamental frequency (F0) and short duration compared to the Holarctic clade. In contrast, the Himalayan wolf howls typically had lower mean F0, unmodulated frequencies, and short howls compared to Holarctic wolf howls. The Himalayan and North African wolves had the most acoustically distinct howls and differed significantly from each other and to the Holarctic wolves. Along with the influence of body size and environmental differences, these results suggest that genetic divergence and/or geographic distance may play an important role in understanding howl variation across subspecies.
Variation in vocal communication is influenced by many factors, often leading to acoustic divergence across populations and species (Wilkins et al. 2013). Multiple drivers such as environmental conditions (Morton 1975; Slabbekoorn and Smith 2002; Slabbekoorn and Peet 2003; Russo et al. 2007), body size (Badyaev and Leaf 1997; Fitch 1997), population differences in mate preferences (Gerhardt 1991), and stochastic forces (Percy et al. 2006; Irwin et al. 2008) have been shown to shape patterns in acoustic variation across a wide range of taxa and further clarify the processes governing acoustic divergence. Understanding these acoustic differences contributes to a comprehensive view of the genetic, behavioral, and ecological attributes of a taxon, and often aids in the taxonomic recognition of cryptic species (Narins 1983; Henry 1994; Kingston et al. 2001).
Population-level variation in vocalizations is found across a wide range of mammalian species (seals—Terhune 1994; Risch et al. 2007; whales—Ford 1991; Rossi-Santos and Podos 2006; rodents—Conner 1982; Roux et al. 2002; Campbell et al. 2010; primates—Delgado 2007; bats—O’Farrell et al. 2000; Law et al. 2002), yet remains relatively unexplored for most mammal groups compared to other taxa, such as birds. Additional variation in vocalizations of some taxa have been demonstrated at the subspecies, group, and individual levels, including the howl acoustic structure of wolves Canislupus (individual—Theberge and Falls 1967; Tooze et al. 1990; Palacios et al. 2007; Root-Gutteridge et al. 2014a, 2014b; group—Zaccaroni et al. 2012; subspecies—Kershenbaum et al. 2016). The howl is the main long-distance vocalization and is used for social spacing, reassembly between dispersed individuals, and territorial defense (Harrington and Mech 1979; Nowak et al. 2007). Previous studies have suggested geographic distance may play a role in howl differences across groups, such that distance among wolf packs increases the differences in howl acoustic qualities, producing a regional accent (Zaccaroni et al. 2012; Root-Gutteridge et al. 2014b). On a subspecies level, smaller canid species show a greater diversity of howl-type usages that may reflect differences in vocal production in different sized species, distinct acoustic properties of habitats, or varying emphases on the social role of howling (Kershenbaum et al. 2016). Taken together, this variation in wolf howl acoustic structure may reflect the evolutionary history, morphology, and/or ecology of a population or subspecies.
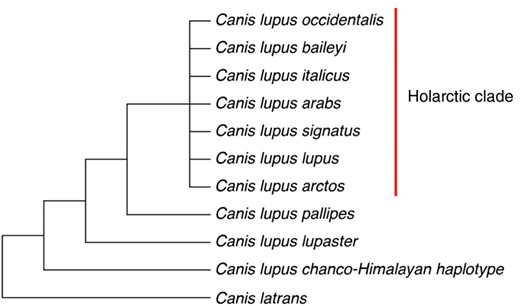
Phylogenetic tree displaying the major relationships within C. lupus clade based on 726 bp of the Cyt b gene from Rueness et al. (2011).
Although once considered to fall within the golden jackal clade, the morphology and behavior of the North African wolf actually show more similarity toward wolves, such as the lack of spinal curvature in their aggressive posture and usage of howl types (Ferguson 1981; Gaubert et al. 2012; Kershenbaum et al. 2016). This third basal wolf lineage likely colonized Africa prior to the northern hemisphere radiation of C.lupus (Gaubert et al. 2012). Based on mitochondrial DNA, the wolf is considered to be C.lupuslupaster, yet a robust description of the wolf’s morphological and behavioral attributes is currently deficient due to recent taxonomic identification (Gaubert et al. 2012). While genetically verified North African wolves are larger than golden jackals, there are some individuals or populations that fall into an intermediate phenotype, making their identity unclear due to lack of knowledge on C.lupuslupaster morphological variation and extent of hybridization in the region (Gaubert et al. 2012). While recent genetic analyses have provided insights into wolf phylogenetics (vonHoldt et al. 2011), there is a lack of understanding in the ecology, morphology, and behavior of these basal wolf lineages, especially in light of possible behavioral and ecological barriers in the maintenance of these evolutionary distinct wolf populations.
Here, we quantify the magnitude and pattern of howl acoustic structure variation to evaluate whether these long-range vocalizations show acoustic differences across wolf subspecies. Using a set of 1,070 howls from 10 wolf subspecies, we measured 9 acoustic parameters to examine how certain wolf subspecies howls may differ across specific acoustic characteristics. To test whether the basal wolf lineages show the most divergent howls, we assess how Himalayan, North African, and Indian wolf howl acoustic structure compare to the Holarctic wolf clade and each other using principal component analysis (PCA) and random forest classifier. Lastly, we discuss the relative influences of evolutionary history, morphology, and environment on understanding the variation in wolf howl acoustic structure.
Materials and Methods
Data collection and sound analysis
Howl surveys for the Indian and Himalayan wolf were conducted in the grasslands of Maharashtra as well as the Trans-Himalayas of Spiti Valley in Himachal Pradesh and the Changthang plateau of Ladakh in Jammu and Kashmir, India. Although the geographic range of the Himalayan wolf has not been properly determined, the free-ranging wolves of Spiti Valley are considered Himalayan wolves based on a genetic sample in Sharma et al.’s (2004) study that originated from captive wolves captured near Kibber in Spiti Valley. The free-ranging wolves at the study site in Ladakh have yet to be genetically determined; however, mitochondrial DNA studies on museum specimens from Ladakh and Tibet have been found to fall within the Himalayan wolf clade (Sharma et al. 2004). Therefore, in this study we consider the Ladakh wolves to be C.lupuschanco-Himalayan haplotype.
Wild and captive wolf howling from all subspecies were either elicited by playbacks using previously recorded howls or were recorded as spontaneous howls (i.e., not elicited). The Himalayan and Indian wolf howls were recorded by playback using a JBL Charge speaker (HARMAN, Stamford, CT) between 6 PM and 9 PM and 05 AM and 08 AM, corresponding to the peak daily howling time in wolves (Gazzola et al. 2002). Playback consisted of 3 trials with a 2-min wait time between trials for a howl response. Vocalizations were recorded using a Blue Yeti Pro USB Condenser Microphone (Blue Microphones, Westlake Village, CA) and a digital recorder [Zoom H4N Handheld Audio Recorder {Zoom Corporation, Tokyo, Japan}] with a sampling rate of 44.1 kHz and 16-bit accuracy. Analysis of recorded howling was performed with Raven Pro 1.3 software (Cornell Lab of Ornithology, Ithaca, NY) using the default Hanning window, 2,048 Hz time resolution, and spectrogram view (Cornell Lab of Ornithology). Recordings of other wolf subspecies were from captive and wild wolves from a variety of different sources given in Table 1 and Supplementary Table S2. Fundamental frequency, was measured every 0.05 s for Himalayan, Indian, Iberian, and Italian wolves (Palacios et al. 2007; Passilongo et al. 2009). The howls for the other wolf subspecies were used from Kershenbaum et al. (2016), in which 50 measurements of fundamental frequency was made for each howl, ranging from every 0.02 s to 0.31 s. Howl duration was from 0.52 s to 15.5 s. From these measurements, 9 acoustic parameters were obtained that were based from previous studies demonstrating them to be useful in individual and group-specific identification (Supplementary Table S3; Tooze et al. 1990; Palacios et al. 2007; Zaccaroni et al. 2012; Root-Gutteridge et al. 2014b).
Wolf subspecies included in the study with associated howl amount, habitat characteristics, and body size (kg)
| Clade . | Wolf subspecies . | Scientific classification . | Number of individuals (number of packs) . | Habitat (country recorded from) . | Body size . | Number of howls . |
|---|---|---|---|---|---|---|
| Himalayan | Himalayan wolf | C. lupus chanco— Himalayan haplotype | ∼15 (4) | High-altitude, arid mountains and valleysa | ∼35 kga | 301 |
| North African | North African wolf | C. lupus lupaster | 6 packs | Arid scrubland, forests, savannahb | “the African wolf is larger than the golden jackal, but their size may overlap”b | 33 |
| Indian | Indian wolf | C. lupus pallipes | ∼10 (4) | Semi-arid grasslands and scrublandsc | 19–25 kg (♂): 17–22 kg (♀)c | 117 |
| Holarctic | Arctic wolf | C. lupus arctos | ∼12 (7) | Tundra and taigad | ∼36.6 kg (♂); ∼29.6 kg (♀)e | 26 |
| European wolf | C. lupus lupus | ∼20 packs | Mountainous forested areas and scrublandsf | 25–55 kg (♂), 23–42 kg (♀)g | 65 | |
| Iberian wolf | C. lupus signatus | 11 (1) | Mountainous forested areash | ∼40 kg (♂); ∼27 kg (♀)h | 176 | |
| Italian wolf | C. lupus italicus | 7 packs | Deciduous foresti | 25–35 kg (♂)j | 164 | |
| Israeli wolf | C. lupus arabs | 5 (1) | Arid scrubland and desertk | 19–27 kg (♂); 17.4–22.5 kg (♀)k | 30 | |
| Mackenzie Valley wolf | C. lupus occidentalis | 9 packs | Mountainous forested areas, montane grasslandl | ∼32–64 kg (♂); ∼30–50 kg (♀)m | 127 | |
| Mexican wolf | C. lupus baileyi | 2 packs | Semi-forested areasn and semi-arid scrublando | 41–23 kgp; 40.2 kg (♂); 35.1 kg (♀)q | 31 |
| Clade . | Wolf subspecies . | Scientific classification . | Number of individuals (number of packs) . | Habitat (country recorded from) . | Body size . | Number of howls . |
|---|---|---|---|---|---|---|
| Himalayan | Himalayan wolf | C. lupus chanco— Himalayan haplotype | ∼15 (4) | High-altitude, arid mountains and valleysa | ∼35 kga | 301 |
| North African | North African wolf | C. lupus lupaster | 6 packs | Arid scrubland, forests, savannahb | “the African wolf is larger than the golden jackal, but their size may overlap”b | 33 |
| Indian | Indian wolf | C. lupus pallipes | ∼10 (4) | Semi-arid grasslands and scrublandsc | 19–25 kg (♂): 17–22 kg (♀)c | 117 |
| Holarctic | Arctic wolf | C. lupus arctos | ∼12 (7) | Tundra and taigad | ∼36.6 kg (♂); ∼29.6 kg (♀)e | 26 |
| European wolf | C. lupus lupus | ∼20 packs | Mountainous forested areas and scrublandsf | 25–55 kg (♂), 23–42 kg (♀)g | 65 | |
| Iberian wolf | C. lupus signatus | 11 (1) | Mountainous forested areash | ∼40 kg (♂); ∼27 kg (♀)h | 176 | |
| Italian wolf | C. lupus italicus | 7 packs | Deciduous foresti | 25–35 kg (♂)j | 164 | |
| Israeli wolf | C. lupus arabs | 5 (1) | Arid scrubland and desertk | 19–27 kg (♂); 17.4–22.5 kg (♀)k | 30 | |
| Mackenzie Valley wolf | C. lupus occidentalis | 9 packs | Mountainous forested areas, montane grasslandl | ∼32–64 kg (♂); ∼30–50 kg (♀)m | 127 | |
| Mexican wolf | C. lupus baileyi | 2 packs | Semi-forested areasn and semi-arid scrublando | 41–23 kgp; 40.2 kg (♂); 35.1 kg (♀)q | 31 |
Note: A total of 1,070 howls were included in the analysis
Wolf subspecies included in the study with associated howl amount, habitat characteristics, and body size (kg)
| Clade . | Wolf subspecies . | Scientific classification . | Number of individuals (number of packs) . | Habitat (country recorded from) . | Body size . | Number of howls . |
|---|---|---|---|---|---|---|
| Himalayan | Himalayan wolf | C. lupus chanco— Himalayan haplotype | ∼15 (4) | High-altitude, arid mountains and valleysa | ∼35 kga | 301 |
| North African | North African wolf | C. lupus lupaster | 6 packs | Arid scrubland, forests, savannahb | “the African wolf is larger than the golden jackal, but their size may overlap”b | 33 |
| Indian | Indian wolf | C. lupus pallipes | ∼10 (4) | Semi-arid grasslands and scrublandsc | 19–25 kg (♂): 17–22 kg (♀)c | 117 |
| Holarctic | Arctic wolf | C. lupus arctos | ∼12 (7) | Tundra and taigad | ∼36.6 kg (♂); ∼29.6 kg (♀)e | 26 |
| European wolf | C. lupus lupus | ∼20 packs | Mountainous forested areas and scrublandsf | 25–55 kg (♂), 23–42 kg (♀)g | 65 | |
| Iberian wolf | C. lupus signatus | 11 (1) | Mountainous forested areash | ∼40 kg (♂); ∼27 kg (♀)h | 176 | |
| Italian wolf | C. lupus italicus | 7 packs | Deciduous foresti | 25–35 kg (♂)j | 164 | |
| Israeli wolf | C. lupus arabs | 5 (1) | Arid scrubland and desertk | 19–27 kg (♂); 17.4–22.5 kg (♀)k | 30 | |
| Mackenzie Valley wolf | C. lupus occidentalis | 9 packs | Mountainous forested areas, montane grasslandl | ∼32–64 kg (♂); ∼30–50 kg (♀)m | 127 | |
| Mexican wolf | C. lupus baileyi | 2 packs | Semi-forested areasn and semi-arid scrublando | 41–23 kgp; 40.2 kg (♂); 35.1 kg (♀)q | 31 |
| Clade . | Wolf subspecies . | Scientific classification . | Number of individuals (number of packs) . | Habitat (country recorded from) . | Body size . | Number of howls . |
|---|---|---|---|---|---|---|
| Himalayan | Himalayan wolf | C. lupus chanco— Himalayan haplotype | ∼15 (4) | High-altitude, arid mountains and valleysa | ∼35 kga | 301 |
| North African | North African wolf | C. lupus lupaster | 6 packs | Arid scrubland, forests, savannahb | “the African wolf is larger than the golden jackal, but their size may overlap”b | 33 |
| Indian | Indian wolf | C. lupus pallipes | ∼10 (4) | Semi-arid grasslands and scrublandsc | 19–25 kg (♂): 17–22 kg (♀)c | 117 |
| Holarctic | Arctic wolf | C. lupus arctos | ∼12 (7) | Tundra and taigad | ∼36.6 kg (♂); ∼29.6 kg (♀)e | 26 |
| European wolf | C. lupus lupus | ∼20 packs | Mountainous forested areas and scrublandsf | 25–55 kg (♂), 23–42 kg (♀)g | 65 | |
| Iberian wolf | C. lupus signatus | 11 (1) | Mountainous forested areash | ∼40 kg (♂); ∼27 kg (♀)h | 176 | |
| Italian wolf | C. lupus italicus | 7 packs | Deciduous foresti | 25–35 kg (♂)j | 164 | |
| Israeli wolf | C. lupus arabs | 5 (1) | Arid scrubland and desertk | 19–27 kg (♂); 17.4–22.5 kg (♀)k | 30 | |
| Mackenzie Valley wolf | C. lupus occidentalis | 9 packs | Mountainous forested areas, montane grasslandl | ∼32–64 kg (♂); ∼30–50 kg (♀)m | 127 | |
| Mexican wolf | C. lupus baileyi | 2 packs | Semi-forested areasn and semi-arid scrublando | 41–23 kgp; 40.2 kg (♂); 35.1 kg (♀)q | 31 |
Note: A total of 1,070 howls were included in the analysis
Statistical analysis
To assess subspecies differences for each howl acoustic parameter, we compared each basal wolf lineage—the Himalayan, North African, and Indian wolf—howl acoustic parameters to each other and the subspecies within the Holarctic clade, which is comprised of the European, Iberian, Italian, Israeli, Arctic, Mexican, and Mackenzie Valley wolf subspecies. Due to the nonparametric nature of the data, we used a Kruskal–Wallis one-way analysis of variance under Dunn’s test with Bonferroni P value adjustments to assess acoustic differences across wolf subspecies. To distill major howl acoustic parameters into a single composite measure, we used PCA, which extracts linearly uncorrelated variables from a suite of potentially correlated variables, thereby reducing the dimensionality of the data. The first principal component (PC1) and second principle component (PC2) were used to evaluate subspecies differences using a Kruskal–Wallis one-way analysis of variance under Dunn’s test with Bonferroni P value adjustments. All possible subspecies comparisons were tested to investigate patterns in PC1 and PC2 differentiation within the C.lupus clade.
Subsequently, we applied Breiman’s random forest algorithm (Breiman 2001), a tree-based machine learning classifier for nonparametric data, to examine whether howls from each subspecies can be distinguished based on their 9 acoustic parameters and to identify the most important acoustic parameters in classifying wolf subspecies. The random forest algorithm consists of tree-structured classifiers, which each tree is trained on a bootstrapped sample with replacement from the training data and utilizes both bagging and random feature selection to construct decision trees (Breiman 2001). For building the random forest model, we used all 9 acoustic parameters and based the algorithm on 2,000 decision trees. Classical multidimensional scaling (MDS) was then applied to visualize dissimilarities in howl acoustic structure across wolf subspecies using the proximity matrix from the random forest algorithm.
Results
Compared to the Holarctic clade, both the North African and Indian wolf typically exhibited high mean frequencies, wide frequency ranges, and short duration. The Himalayan wolf howls characteristically had low mean frequency, short duration, and unmodulated frequency variation in howls compared to the Holarctic clade (Supplementary Table S4). Within the Holarctic clade, the smallest wolf subspecies—the Israeli wolf—had the highest mean frequency and shortest duration (average meanf = 619 Hz, average duration = 2.92 s), whereas lower mean frequencies were generally observed in larger wolf subspecies, such as the Mackenzie Valley wolf (average meanf = 492.5 Hz, average duration = 4.14 s) and European wolf (average meanf = 418.7 Hz, average duration = 5.15 s).
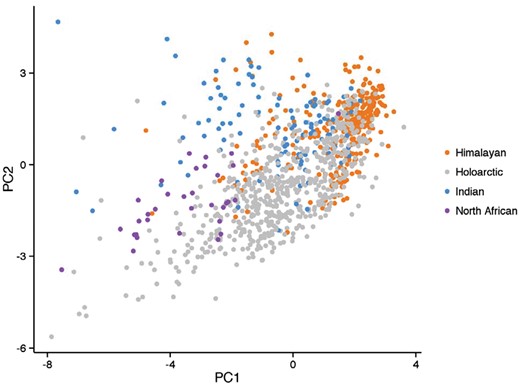
PCA plot incorporating 1,070 howls across the Holarctic, Himalayan, Indian, and North African lineages of C. lupus. The Holarctic lineage represents Iberian, Italian, Israeli, European, Mexican, Mackenzie Valley, and Arctic wolf subspecies. The basal wolf lineages form distinct separate clusters with partial overlap within the Holartic clade.
PC1 loadings, PC2 loadings, and the mean decrease in accuracy values
| Howl acoustic parameter . | Abbreviation . | PC1 loadings . | PC2 loadings . | Mean decrease in accuracy . |
|---|---|---|---|---|
| Mean frequency | Meanf | −0.52 | 0.014 | 96.15 |
| Maximum frequency | Maxf | −0.50 | −0.21 | 117.72 |
| Minimum frequency | Minf | −0.43 | 0.30 | 86.14 |
| End frequency | Endf | −0.44 | 0.30 | 96.14 |
| Duration | Duration | 0.13 | −0.31 | 131.56 |
| Range | Range | −0.27 | −0.49 | 120.36 |
| Coefficient of frequency variation [(SD/Meanf) × 100] | cofv | −0.12 | −0.54 | 109.24 |
| Position of maximum frequency | Posmax | −0.0105 | 0.29 | 75.15 |
| Position of minimum frequency | Posmin | −0.0089 | −0.25 | 62.83 |
| Howl acoustic parameter . | Abbreviation . | PC1 loadings . | PC2 loadings . | Mean decrease in accuracy . |
|---|---|---|---|---|
| Mean frequency | Meanf | −0.52 | 0.014 | 96.15 |
| Maximum frequency | Maxf | −0.50 | −0.21 | 117.72 |
| Minimum frequency | Minf | −0.43 | 0.30 | 86.14 |
| End frequency | Endf | −0.44 | 0.30 | 96.14 |
| Duration | Duration | 0.13 | −0.31 | 131.56 |
| Range | Range | −0.27 | −0.49 | 120.36 |
| Coefficient of frequency variation [(SD/Meanf) × 100] | cofv | −0.12 | −0.54 | 109.24 |
| Position of maximum frequency | Posmax | −0.0105 | 0.29 | 75.15 |
| Position of minimum frequency | Posmin | −0.0089 | −0.25 | 62.83 |
Notes: These values are based on gini impurity index for each howl acoustic parameter. Higher values of mean decrease in accuracy indicate variables that are more important in classification for the random forest model. The proportion of variance of PC1, PC2, and PC3 was 0.39, 0.29, and 0.14, respectively. SD, standard deviation.
PC1 loadings, PC2 loadings, and the mean decrease in accuracy values
| Howl acoustic parameter . | Abbreviation . | PC1 loadings . | PC2 loadings . | Mean decrease in accuracy . |
|---|---|---|---|---|
| Mean frequency | Meanf | −0.52 | 0.014 | 96.15 |
| Maximum frequency | Maxf | −0.50 | −0.21 | 117.72 |
| Minimum frequency | Minf | −0.43 | 0.30 | 86.14 |
| End frequency | Endf | −0.44 | 0.30 | 96.14 |
| Duration | Duration | 0.13 | −0.31 | 131.56 |
| Range | Range | −0.27 | −0.49 | 120.36 |
| Coefficient of frequency variation [(SD/Meanf) × 100] | cofv | −0.12 | −0.54 | 109.24 |
| Position of maximum frequency | Posmax | −0.0105 | 0.29 | 75.15 |
| Position of minimum frequency | Posmin | −0.0089 | −0.25 | 62.83 |
| Howl acoustic parameter . | Abbreviation . | PC1 loadings . | PC2 loadings . | Mean decrease in accuracy . |
|---|---|---|---|---|
| Mean frequency | Meanf | −0.52 | 0.014 | 96.15 |
| Maximum frequency | Maxf | −0.50 | −0.21 | 117.72 |
| Minimum frequency | Minf | −0.43 | 0.30 | 86.14 |
| End frequency | Endf | −0.44 | 0.30 | 96.14 |
| Duration | Duration | 0.13 | −0.31 | 131.56 |
| Range | Range | −0.27 | −0.49 | 120.36 |
| Coefficient of frequency variation [(SD/Meanf) × 100] | cofv | −0.12 | −0.54 | 109.24 |
| Position of maximum frequency | Posmax | −0.0105 | 0.29 | 75.15 |
| Position of minimum frequency | Posmin | −0.0089 | −0.25 | 62.83 |
Notes: These values are based on gini impurity index for each howl acoustic parameter. Higher values of mean decrease in accuracy indicate variables that are more important in classification for the random forest model. The proportion of variance of PC1, PC2, and PC3 was 0.39, 0.29, and 0.14, respectively. SD, standard deviation.
Comparing all possible wolf subspecies combinations using a Kruskal–Wallis one-way analysis of variance under Dunn’s test with Bonferroni P value adjustments on PC1 showed that the Himalayan wolf was acoustically distinct from both all Holarctic and the two other basal wolf subspecies (Table 3). The North African wolf showed significant differences for PC1 among all wolf subspecies except the Arctic and Israeli wolf. In contrast, the Indian wolf showed no significant differences in howl acoustic structure from any of the Holarctic wolf subspecies. Within the Holarctic clade, various wolf subspecies were also acoustically distinct from each other, yet these differences were less marked than those between and within the 2 most basal wolf lineages (Table 3). For the PC2 comparison, the Himalayan and Indian wolf showed the most distinct howls across all wolf subspecies (Table 4).
PC1 comparison across all wolf subspecies using a post hoc Kruskal–Wallis one-way analysis of variance under Dunn’s tests with Bonferroni P value adjustments
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.004 | |
| North African | <0.001 | <0.001 | 0.43 | <0.001 | <0.001 | <0.001 | 0.17 | <0.001 | <0.001 | |
| Indian | <0.001 | <0.001 | 0.17 | 0.095 | 1.00 | 0.26 | 0.31 | 1.00 | 0.43 | |
| Arctic | <0.001 | 0.43 | 0.17 | <0.001 | 0.25 | 0.0016 | 1.00 | 0.26 | 0.0059 | |
| European | <0.001 | <0.001 | 0.095 | <0.001 | 0.015 | 1.00 | 0.0016 | 0.013 | 1.00 | |
| Iberian | <0.001 | <0.001 | 1.00 | 0.25 | 0.015 | 0.041 | 0.43 | 1.00 | 0.25 | |
| Mackenzie Valley | <0.001 | <0.001 | 0.26 | 0.0016 | 1.00 | 0.041 | 0.0052 | 0.035 | 1.00 | |
| Israeli | <0.001 | 0.17 | 0.31 | 1.00 | 0.0016 | 0.43 | 0.0052 | 0.43 | 0.016 | |
| Italian | <0.001 | <0.001 | 1.00 | 0.26 | 0.013 | 1.00 | 0.035 | 0.43 | 0.23 | |
| Mexican | 0.004 | <0.001 | 0.43 | 0.0059 | 1.00 | 0.25 | 1.00 | 0.016 | 0.23 |
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.004 | |
| North African | <0.001 | <0.001 | 0.43 | <0.001 | <0.001 | <0.001 | 0.17 | <0.001 | <0.001 | |
| Indian | <0.001 | <0.001 | 0.17 | 0.095 | 1.00 | 0.26 | 0.31 | 1.00 | 0.43 | |
| Arctic | <0.001 | 0.43 | 0.17 | <0.001 | 0.25 | 0.0016 | 1.00 | 0.26 | 0.0059 | |
| European | <0.001 | <0.001 | 0.095 | <0.001 | 0.015 | 1.00 | 0.0016 | 0.013 | 1.00 | |
| Iberian | <0.001 | <0.001 | 1.00 | 0.25 | 0.015 | 0.041 | 0.43 | 1.00 | 0.25 | |
| Mackenzie Valley | <0.001 | <0.001 | 0.26 | 0.0016 | 1.00 | 0.041 | 0.0052 | 0.035 | 1.00 | |
| Israeli | <0.001 | 0.17 | 0.31 | 1.00 | 0.0016 | 0.43 | 0.0052 | 0.43 | 0.016 | |
| Italian | <0.001 | <0.001 | 1.00 | 0.26 | 0.013 | 1.00 | 0.035 | 0.43 | 0.23 | |
| Mexican | 0.004 | <0.001 | 0.43 | 0.0059 | 1.00 | 0.25 | 1.00 | 0.016 | 0.23 |
Note: Each subspecies was treated as an independent unit. Bolded values indicate P values below 0.001.
PC1 comparison across all wolf subspecies using a post hoc Kruskal–Wallis one-way analysis of variance under Dunn’s tests with Bonferroni P value adjustments
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.004 | |
| North African | <0.001 | <0.001 | 0.43 | <0.001 | <0.001 | <0.001 | 0.17 | <0.001 | <0.001 | |
| Indian | <0.001 | <0.001 | 0.17 | 0.095 | 1.00 | 0.26 | 0.31 | 1.00 | 0.43 | |
| Arctic | <0.001 | 0.43 | 0.17 | <0.001 | 0.25 | 0.0016 | 1.00 | 0.26 | 0.0059 | |
| European | <0.001 | <0.001 | 0.095 | <0.001 | 0.015 | 1.00 | 0.0016 | 0.013 | 1.00 | |
| Iberian | <0.001 | <0.001 | 1.00 | 0.25 | 0.015 | 0.041 | 0.43 | 1.00 | 0.25 | |
| Mackenzie Valley | <0.001 | <0.001 | 0.26 | 0.0016 | 1.00 | 0.041 | 0.0052 | 0.035 | 1.00 | |
| Israeli | <0.001 | 0.17 | 0.31 | 1.00 | 0.0016 | 0.43 | 0.0052 | 0.43 | 0.016 | |
| Italian | <0.001 | <0.001 | 1.00 | 0.26 | 0.013 | 1.00 | 0.035 | 0.43 | 0.23 | |
| Mexican | 0.004 | <0.001 | 0.43 | 0.0059 | 1.00 | 0.25 | 1.00 | 0.016 | 0.23 |
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 0.004 | |
| North African | <0.001 | <0.001 | 0.43 | <0.001 | <0.001 | <0.001 | 0.17 | <0.001 | <0.001 | |
| Indian | <0.001 | <0.001 | 0.17 | 0.095 | 1.00 | 0.26 | 0.31 | 1.00 | 0.43 | |
| Arctic | <0.001 | 0.43 | 0.17 | <0.001 | 0.25 | 0.0016 | 1.00 | 0.26 | 0.0059 | |
| European | <0.001 | <0.001 | 0.095 | <0.001 | 0.015 | 1.00 | 0.0016 | 0.013 | 1.00 | |
| Iberian | <0.001 | <0.001 | 1.00 | 0.25 | 0.015 | 0.041 | 0.43 | 1.00 | 0.25 | |
| Mackenzie Valley | <0.001 | <0.001 | 0.26 | 0.0016 | 1.00 | 0.041 | 0.0052 | 0.035 | 1.00 | |
| Israeli | <0.001 | 0.17 | 0.31 | 1.00 | 0.0016 | 0.43 | 0.0052 | 0.43 | 0.016 | |
| Italian | <0.001 | <0.001 | 1.00 | 0.26 | 0.013 | 1.00 | 0.035 | 0.43 | 0.23 | |
| Mexican | 0.004 | <0.001 | 0.43 | 0.0059 | 1.00 | 0.25 | 1.00 | 0.016 | 0.23 |
Note: Each subspecies was treated as an independent unit. Bolded values indicate P values below 0.001.
PC2 comparisons across all wolf subspecies using a post hoc Kruskal–Wallis one-way analysis of variance under Dunn’s tests with Bonferroni P value adjustments
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| North African | <0.001 | <0.001 | 1.00 | 0.37 | 1.00 | <0.001 | 0.21 | 0.95 | 0.77 | |
| Indian | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| Arctic | <0.001 | 1.00 | <0.001 | 1.00 | 1.00 | 0.026 | 0.85 | 1.00 | 1.00 | |
| European | <0.001 | 0.37 | <0.001 | 1.00 | <0.001 | 0.67 | 1.00 | 1.00 | 1.00 | |
| Iberian | <0.001 | 1.00 | <0.001 | 1.00 | <0.001 | <0.001 | 0.0038 | <0.001 | 0.044 | |
| Mackenzie Valley | <0.001 | <0.001 | <0.001 | 0.026 | 0.67 | <0.001 | 1.00 | 0.0021 | 1.00 | |
| Israeli | <0.001 | 0.21 | <0.001 | 0.85 | 1.00 | 0.0038 | 1.00 | 1.00 | 1.00 | |
| Italian | <0.001 | 0.95 | <0.001 | 1.00 | 1.00 | <0.001 | 0.0021 | 1.00 | 1.00 | |
| Mexican | <0.001 | 0.77 | <0.001 | 1.00 | 1.00 | 0.044 | 1.00 | 1.00 | 1.00 |
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| North African | <0.001 | <0.001 | 1.00 | 0.37 | 1.00 | <0.001 | 0.21 | 0.95 | 0.77 | |
| Indian | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| Arctic | <0.001 | 1.00 | <0.001 | 1.00 | 1.00 | 0.026 | 0.85 | 1.00 | 1.00 | |
| European | <0.001 | 0.37 | <0.001 | 1.00 | <0.001 | 0.67 | 1.00 | 1.00 | 1.00 | |
| Iberian | <0.001 | 1.00 | <0.001 | 1.00 | <0.001 | <0.001 | 0.0038 | <0.001 | 0.044 | |
| Mackenzie Valley | <0.001 | <0.001 | <0.001 | 0.026 | 0.67 | <0.001 | 1.00 | 0.0021 | 1.00 | |
| Israeli | <0.001 | 0.21 | <0.001 | 0.85 | 1.00 | 0.0038 | 1.00 | 1.00 | 1.00 | |
| Italian | <0.001 | 0.95 | <0.001 | 1.00 | 1.00 | <0.001 | 0.0021 | 1.00 | 1.00 | |
| Mexican | <0.001 | 0.77 | <0.001 | 1.00 | 1.00 | 0.044 | 1.00 | 1.00 | 1.00 |
Note: Each subspecies was treated as an independent unit. Bolded values indicate P values below 0.001.
PC2 comparisons across all wolf subspecies using a post hoc Kruskal–Wallis one-way analysis of variance under Dunn’s tests with Bonferroni P value adjustments
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| North African | <0.001 | <0.001 | 1.00 | 0.37 | 1.00 | <0.001 | 0.21 | 0.95 | 0.77 | |
| Indian | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| Arctic | <0.001 | 1.00 | <0.001 | 1.00 | 1.00 | 0.026 | 0.85 | 1.00 | 1.00 | |
| European | <0.001 | 0.37 | <0.001 | 1.00 | <0.001 | 0.67 | 1.00 | 1.00 | 1.00 | |
| Iberian | <0.001 | 1.00 | <0.001 | 1.00 | <0.001 | <0.001 | 0.0038 | <0.001 | 0.044 | |
| Mackenzie Valley | <0.001 | <0.001 | <0.001 | 0.026 | 0.67 | <0.001 | 1.00 | 0.0021 | 1.00 | |
| Israeli | <0.001 | 0.21 | <0.001 | 0.85 | 1.00 | 0.0038 | 1.00 | 1.00 | 1.00 | |
| Italian | <0.001 | 0.95 | <0.001 | 1.00 | 1.00 | <0.001 | 0.0021 | 1.00 | 1.00 | |
| Mexican | <0.001 | 0.77 | <0.001 | 1.00 | 1.00 | 0.044 | 1.00 | 1.00 | 1.00 |
| . | Himalayan . | North African . | Indian . | Arctic . | European . | Iberian . | Mackenzie Valley . | Israeli . | Italian . | Mexican . |
|---|---|---|---|---|---|---|---|---|---|---|
| Himalayan | <0.001 | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| North African | <0.001 | <0.001 | 1.00 | 0.37 | 1.00 | <0.001 | 0.21 | 0.95 | 0.77 | |
| Indian | 1.00 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |
| Arctic | <0.001 | 1.00 | <0.001 | 1.00 | 1.00 | 0.026 | 0.85 | 1.00 | 1.00 | |
| European | <0.001 | 0.37 | <0.001 | 1.00 | <0.001 | 0.67 | 1.00 | 1.00 | 1.00 | |
| Iberian | <0.001 | 1.00 | <0.001 | 1.00 | <0.001 | <0.001 | 0.0038 | <0.001 | 0.044 | |
| Mackenzie Valley | <0.001 | <0.001 | <0.001 | 0.026 | 0.67 | <0.001 | 1.00 | 0.0021 | 1.00 | |
| Israeli | <0.001 | 0.21 | <0.001 | 0.85 | 1.00 | 0.0038 | 1.00 | 1.00 | 1.00 | |
| Italian | <0.001 | 0.95 | <0.001 | 1.00 | 1.00 | <0.001 | 0.0021 | 1.00 | 1.00 | |
| Mexican | <0.001 | 0.77 | <0.001 | 1.00 | 1.00 | 0.044 | 1.00 | 1.00 | 1.00 |
Note: Each subspecies was treated as an independent unit. Bolded values indicate P values below 0.001.
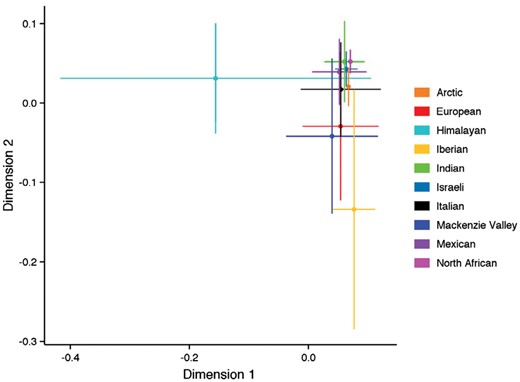
Classical MDS plot from the random forest algorithm using 9 acoustic parameters across the 10 wolf subspecies. Himalayan and Iberian wolves form distinct highly variable clusters, whereas North African and Indian wolves form tight clusters in the upper right section of the axis.
Percent of howls classified correctly to each wolf subspecies on 9 acoustic parameters
| Wolf subspecies . | Percent correctly classified . | Best guess . | Percent misclassified as best guess . |
|---|---|---|---|
| Himalayan wolf | 87.7 | Indian | 5.3 |
| North African wolf | 81.2 | Italian | 9.1 |
| Indian wolf | 68.4 | Himalayan | 15.4 |
| Arctic wolf | 34.6 | Italian | 34.6 |
| European wolf | 32.3 | Iberian | 29.2 |
| Iberian wolf | 75.6 | Italian | 9.6 |
| Israeli wolf | 6.7 | Italian | 30.0 |
| Italian wolf | 52.4 | Himalayan | 17.7 |
| Mackenzie Valley wolf | 62.2 | Himalayan | 18.1 |
| Mexican wolf | 54.8 | Iberian | 38.7 |
| Wolf subspecies . | Percent correctly classified . | Best guess . | Percent misclassified as best guess . |
|---|---|---|---|
| Himalayan wolf | 87.7 | Indian | 5.3 |
| North African wolf | 81.2 | Italian | 9.1 |
| Indian wolf | 68.4 | Himalayan | 15.4 |
| Arctic wolf | 34.6 | Italian | 34.6 |
| European wolf | 32.3 | Iberian | 29.2 |
| Iberian wolf | 75.6 | Italian | 9.6 |
| Israeli wolf | 6.7 | Italian | 30.0 |
| Italian wolf | 52.4 | Himalayan | 17.7 |
| Mackenzie Valley wolf | 62.2 | Himalayan | 18.1 |
| Mexican wolf | 54.8 | Iberian | 38.7 |
Notes: Best guess represents the specific wolf subspecies that was most commonly misclassified as the wolf subspecies being tested for. The overall estimate of error rate was 32.2%.
Percent of howls classified correctly to each wolf subspecies on 9 acoustic parameters
| Wolf subspecies . | Percent correctly classified . | Best guess . | Percent misclassified as best guess . |
|---|---|---|---|
| Himalayan wolf | 87.7 | Indian | 5.3 |
| North African wolf | 81.2 | Italian | 9.1 |
| Indian wolf | 68.4 | Himalayan | 15.4 |
| Arctic wolf | 34.6 | Italian | 34.6 |
| European wolf | 32.3 | Iberian | 29.2 |
| Iberian wolf | 75.6 | Italian | 9.6 |
| Israeli wolf | 6.7 | Italian | 30.0 |
| Italian wolf | 52.4 | Himalayan | 17.7 |
| Mackenzie Valley wolf | 62.2 | Himalayan | 18.1 |
| Mexican wolf | 54.8 | Iberian | 38.7 |
| Wolf subspecies . | Percent correctly classified . | Best guess . | Percent misclassified as best guess . |
|---|---|---|---|
| Himalayan wolf | 87.7 | Indian | 5.3 |
| North African wolf | 81.2 | Italian | 9.1 |
| Indian wolf | 68.4 | Himalayan | 15.4 |
| Arctic wolf | 34.6 | Italian | 34.6 |
| European wolf | 32.3 | Iberian | 29.2 |
| Iberian wolf | 75.6 | Italian | 9.6 |
| Israeli wolf | 6.7 | Italian | 30.0 |
| Italian wolf | 52.4 | Himalayan | 17.7 |
| Mackenzie Valley wolf | 62.2 | Himalayan | 18.1 |
| Mexican wolf | 54.8 | Iberian | 38.7 |
Notes: Best guess represents the specific wolf subspecies that was most commonly misclassified as the wolf subspecies being tested for. The overall estimate of error rate was 32.2%.
Discussion
Our quantitative study illustrates that the long-range vocalization of the wolf shows distinct acoustic differences across subspecies in the C.lupus clade. This complements the results from Kershenbaum et al. (2016) where howl-type usage varied among wolf subspecies. Acoustic differences in howls are most prominent in the Himalayan and North African wolf, which diverged before the radiation of the Holarctic clade across the Northern hemisphere (Sharma et al. 2004, Rueness et al. 2011). This suggests that geographic distance and/or genetic divergence may broadly be correlated with wolf howl acoustic structure. Additionally, we observe that smallest wolf subspecies, notably the North African, Indian, and Israeli wolf, have the highest mean frequencies in our study, indicating that body size may effect some acoustic parameters (Supplementary Table S1). Overall, our study contributes to a multi-step process in understanding the processes shaping howl acoustic variation in C.lupus.
In some species, acoustic divergence is associated with genetic or geographic distance, suggesting that stochastic forces play an important role in the divergence of acoustic signals (Percy et al. 2006; Irwin et al. 2008; Amezquita et al. 2009; Chen et al. 2009; Campbell et al. 2010; Thinh et al. 2011). For canids, previous studies have shown that red wolves C.rufus have similar howl types to coyotes C.latrans, despite the red wolf’s intermediate morphology between gray wolves and coyotes (Chambers et al. 2012; Kershenbaum et al. 2016). Although it is debated whether red wolves are hybrids between gray wolves and coyotes (vonHoldt et al. 2016) or if red wolves and coyotes diverged from a common ancestor around 150,000–300,000 years ago (Wilson et al. 2000), similarities in acoustic behavior between the two species may suggest evolutionary history playing a role in howl acoustic variation. In our study, strong acoustic differences in the howls of the 2 most basal wolf lineages may indicate that genetic divergence and/or geographic distance may be broadly correlated with acoustic divergence in C.lupus. Previous studies on the Himalayan, North African, and Indian wolf show that these lineages have been isolated since before the radiation across the Northern Hemisphere (Sharma et al. 2004; Rueness et al. 2011), and during that time may have accumulated acoustic differences in their howls due to stochastic forces, such as behavioral drift. It is notable that the most basal wolf subspecies, the Himalayan wolf, appears to have a divergent acoustic howl structure from other similarly sized wolf subspecies (Table 1), further suggesting that geographic or genetic distance may play important roles in patterns of howl variation. Obtaining howls from adjacent wolf subspecies, such as the Tibetan wolf C.lupuschanco in Northern China and the Steppe wolf C.lupuscampestris in Central Asia would aid in disentangling whether shared ancestry, geographic distance, and/or environmental differences is driving acoustic divergence in wolves.
Owing to the ability of gray wolves to inhabit almost every possible terrestrial environment (Mech and Boitani 2003), body sizes range widely across wolf subspecies and may have influenced some acoustic variables in our study (Table 1). Generally, our results show that the smallest wolf subspecies, such as the Israeli, Indian, and North African wolf, had the highest mean frequencies, a trend seen in previous studies on other taxa (Wallschager 1980; Fitch 1997; Barclay et al. 1999). Smaller canids also commonly show a greater diversity in howl types and favor howls that end in sharp frequency drops (Kershenbaum et al. 2016). While the smallest subspecies, the North African wolf, has the highest mean frequency and greatest mean frequency range, whether an overall trend between body size and howl acoustic structure exists is unclear. For instance, Mexican wolves had one of the lowest mean frequencies and narrowest frequency ranges, yet are also one of the smallest wolf subspecies, averaging between 23 and 41 kg (Hedrick et al. 1997). Although Himalayan wolves are a medium-sized wolf subspecies (Shrotriya et al. 2012), our study shows that Himalayan wolves have the lowest mean frequency of any wolf subspecies in our study. While acoustic differences can be associated to divergent body sizes (Heller and Helveren 1989; Fitch 1997; Jones 1999; Pfefferle and Fischer 2006), vocalizations are influenced by many factors, such as mate choice criteria, which may act as a stronger selective force on acoustic divergence (McComb 1991; Mitani et al. 1992; Charlton et al. 2009). A larger set of howls with associated wolf weights would further elucidate the influence of body size on patterns of wolf howl acoustic structure across wolf subspecies.
While environmental contexts have been shown to influence acoustic signals (Morton 1975; Badyaev and Leaf 1997; Slabbekoorn and Smith 2002), there are no striking differences in habitats of Indian and North African wolves to other wolf subspecies, such as the Israeli or the Mexican wolf subspecies that reside in similar dry scrubland habitats (Olson et al. 2001). In contrast, the Himalayan wolf lives in the highly mountainous and treeless Trans-Himalaya landscape, which may pose additional selective pressures on long-distance communication (Wilkins et al. 2013). Under the acoustic adaptation hypothesis, habitat differences may cause selection on vocal communication that optimizes propagation in specific environments (Morton 1975). This hypothesis is mostly applied to closed versus open habitats, which are commonly defined by the extent of vegetation structure (Morton 1975). Vocalizations in densely vegetated habitats show specific acoustic adjustments to increase sound propagation, such as lengthening vocal signals and limiting frequency modulations (Brown and Handford 2000; Ey and Fischer 2009). Thus, wolves in open, treeless habitats are expected to have howls with a shorter duration, higher frequency modulation, and higher mean frequencies compared to wolves living in closed habitats (Ey and Fischer 2009). Although both Himalayan and Arctic wolves live in primarily treeless and open habitats, they show highly contrasting acoustic structure in their howls, notably in their mean frequencies and frequency variation. Overall for mammals, there have only been a few examples of changes in vocal signals corresponding to habitat differences (but see Masters 1991; Mitani et al. 1999; Perla and Slobodchikoff 2002), suggesting that selective forces imposed by the environment may only weakly influence vocal communication in mammals (Ey and Fischer 2009).
Understanding possible behavioral differences in closely related species provides great insight into speciation (Mendelson and Shaw 2005; Arnegard et al. 2010; Wilkins et al. 2013) and aids in identifying cryptic species (Irwin et al. 2001; Thabah et al. 2006; Angulo and Riechle 2008; Braune et al. 2008; Ramasindrazana et al. 2011). Taxonomic confusion surrounding the Himalayan wolf has persisted for over 165 years, as the wolf was once first considered to be its own species C.laniger, yet later regarded as just another Tibetan wolf population C.lupuschanco (Shrotriya et al. 2012). Although the wolves are genetically unique based on mitochondrial DNA, the classification still remains controversial and understudied, hindering its conservation. This study is the first to provide detailed howl acoustic descriptions of the Himalayan wolf and distinguish that, although morphologically similar to other wolf subspecies, there may be underlying behavioral differences reflecting their basal position in the Canis clade.
Overall, our study shows that the acoustic structure of wolf howls varies across subspecies and provides insight into the possible causes shaping acoustic variation, particularly the role of stochastic forces in which howl variation reflects evolution history or geographic distance. Future research that includes a larger and more diverse sample of howls from current and additional wolf subspecies would further elucidate patterns and processes influencing wolf howl acoustic structure.
Acknowledgments
We thank Dawa Tashi, Stanzin Dorjay, Salvador Lyngdoh, Shivam Shrotriya, and members of Project Wolf for providing logistical support during fieldwork in India. Additionally, we thank the State Forest Departments of Himachal Pradesh, Jammu and Kashmir, Maharashtra, and various zoos in India for permissions to record wolf howls. Finally, we thank all who helped with the project: the staff at Colchester Zoo; the Wildwood Trust, the Borror Laboratory of Bioacoustics; the British Library; Lupus Laetus; Polish Mammal Research Institute; Tigress Productions; the BBC Natural History Unit; Longleat Safari Park; Tierstimmen Archiv; Wild Sweden; Wolf Park; the Macaulay Sound Library and the UK Wolf Conservation Trust; and Mike Collins, Teresa Palmer, Monty Sloan, Karl-Heinz Frommolt, Yorgos Iliopoulos, Christine Anhalt, Louise Gentle, Richard Yarnell, and Victoria Allison Hughes. We also thank Geraldine Werhahn and Bryan Maitland for helpful comments on the manuscript.
Funding
We are thankful for the Fulbright Program and United States-India Education Foundation for financial support to L.H. as well as the Wildlife Institute of India for funding the Ecology and Conservation of the Himalayan wolf project.
Supplementary material
Supplementary material can be found at http://www.cz.oxfordjournals.org/.
References