Abstract
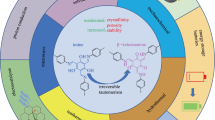
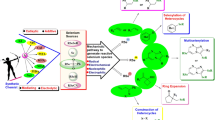
ortho-Nitrobenzyl (oNB) triggers have been extensively used to release various molecules of interest. However, the toxicity and reactivity of the spent chromophore, o-nitrosobenzaldehyde, remains an un-addressed difficulty. In this study we have applied the well-established supramolecular photochemical concepts to retain the spent trigger o-nitrosobenzaldehyde within the organic capsule after release of water-soluble acids and alcohols. The sequestering power of organic capsules for spent chromophores during photorelease from ortho-nitrobenzyl esters, ethers and alcohols is demonstrated with several examples.
Similar content being viewed by others
References
P. Klan, T. Solomek, C. G. Bochet, A. Blanc, R. Givens, M. Rubina, V. Popik, A. Kostikov and J. Wirz, Chem. Rev., 2013, 113, 119–191.
D. D. Young and A. Deiters, Org. Biomol. Chem., 2007, 5, 999–1005.
G. Mayer and A. Heckel, Angew. Chem., Int. Ed., 2006, 45, 4900–4921.
M. Goeldner and R. Givens, Dynamic Studies in Biology: Phototriggers, Photoswitches and Caged Molecules, Wiley- VCH, New York, 2005.
A. P. Pelliccioli and J. Wirz, Photochem. Photobiol. Sci., 2002, 1, 441–458.
M. Abe, Y. Chitosea, S. Jakkampudia, P. T. T. Thuya, Q. Lina, B. T. Vana, A. Yamadaa, R. Oyamaa, M. Sasakia and C. Katan, Synthesis, 2017, 49, 3337–3346.
C. Bochet, J. Chem. Soc., Perkin Trans. 1, 2002, 125–142.
R. S. Givens, M. Rubina and J. Wirz, Photochem. Photobiol., 2012, 11, 472–488.
J. Vazquez, M. A. Romero, R. N. Dsouza and U. Pischel, Chem. Commun., 2016, 52, 6245–6248.
Molecular Encapsulation, ed. U. H. Brinker and J.-L. Mieusset, John Wiley & Sons, Chichester, 2010.
P. Jagadesan, J. P. Da Silva, R. S. Givens and V. Ramamurthy, Org. Lett., 2015, 17, 1276–1279.
N. Jayaraj, P. Jagadesan, S. R. Samanta, J. P. Da Silva and V. Ramamurthy, Org. Lett., 2015, 17, 1276–1279.
N. Kamatham, J. P. Da Silva, R. S. Givens and V. Ramamurthy, Org. Lett., 2017, 19, 3588–3591.
N. Kamatham, D. C. Mendes, J. P. Da Silva, R. S. Givens and V. Ramamurthy, Org. Lett., 2016, 18, 5480–5483.
J. E. T. Corrie, in Dynamic Studies in Biology, ed. M. Goeldner and R. Givens, Wiley-VCH Verlag GmbH & Co., Weinheim, 2003, ch. 1.1, pp. 1–28.
J. E. T. Corrie and D. R. Trentham, in Biological Applications of Switches, ed. H. Morrison, John Wiley & Sons, Inc., New York, 1993, vol. 2, ch. 5, pp. 243–305.
J. A. McCray and D. R. Trentham, Annu. Rev. Biophys. Biophys. Chem., 1989, 18, 239–270.
P. De Mayo and S. T. Reid, Q. Rev., Chem. Soc., 1961, 15, 393–417.
J. A. Barltrop, P. J. Plant and P. Schofield, Chem. Commun., 1966, 822–823.
A. Patchornik, B. Amit and R. B. Woodward, J. Am. Chem. Soc., 1970, 92, 6333–6335.
Y. V. Il’ichev, M. A. Schworer and J. Wirz, J. Am. Chem. Soc., 2004, 126, 4581–4595.
M. Gaplovsky, Y. V. Il’ichev, Y. Kamdzhilov, S. V. Kombarova, M. Mac, M. A. Schworer and J. Wirz, Photochem. Photobiol. Sci., 2005, 4, 33–42.
C. L. D. Gibb and B. C. Gibb, J. Am. Chem. Soc., 2004, 126, 11408–11409.
G. Appendino, A. Minassi, N. Daddario, F. Bianchi and G. C. Tron, Org. Lett., 2002, 4, 3839–3841.
M. Porel, N. Jayaraj, L. S. Kaanumalle, M. V. S. N. Maddipatla, A. Parthasarathy and V. Ramamurthy, Langmuir, 2009, 25, 3473–3481.
N. Jayaraj, S. Jockusch, L. S. Kaanumalle, N. J. Turro and V. Ramamurthy, Can. J. Chem., 2011, 89, 203–213.
R. Choudhury, A. Barman, R. Prabhakar and V. Ramamurthy, J. Phys. Chem. B, 2013, 117, 398–407.
N. Jayaraj, Y. Zhao, A. Parthasarathy, M. Porel, R. S. H. Liu and V. Ramamurthy, Langmuir, 2009, 25, 10575–10586.
M. Schwörer and J. Wirz, Helv. Chim. Acta, 2001, 84, 1441–1458.
M. C. Pirrung, Y. R. Lee, K. Park and J. B. Springer, J. Org. Chem., 1999, 64, 5042–5047.
D. Beaudoin and J. D. Wuest, Chem. Rev., 2016, 116, 258–286.
I. Biljan and H. Vančik, Crystals, 2017, 7, 376.
H. Tomioka, N. Ichikawa and K. Komatsu, J. Am. Chem. Soc., 1992, 114, 8045–8053.
P. Jagadesan, B. Mondal, A. Parthasarathy, V. Jayathirtha Rao and V. Ramamurthy, Org. Lett., 2013, 15, 1326–1329.
T. Schmierer, S. Laimgruber, K. Haiser, K. Kiewisch, J. Neugebauer and P. Gilch, Phys. Chem. Chem. Phys., 2010, 12, 15653–15664.
R. W. Yip, Y. X. Wen, D. Gravel, R. Giasson and D. K. Sharma, J. Phys. Chem., 1991, 95, 6078–6081.
T. Schmierer, F. Bley, K. Schaper and P. Gilch, J. Photochem. Photobiol., A, 2011, 217, 363–368.
R. W. Yip, D. K. Sharma, R. Glasson and D. Gravel, J. Phys. Chem., 1985, 89, 5328–5330.
H. A. Morrison, in The Chemistry of Nitro and Nitroso Groups, ed. H. Feuer, John Wiley and Sons, Inc., New York, 1969, pp. 165–213.
T. Šolomek, S. Mercier, T. Bally and C. G. Bochet, Photochem. Photobiol. Sci., 2012, 11, 548–555.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: Experimental procedures, 1H NMR, UV and ESI-MS spectra for all new compounds. Irradiation procedures, 1H NMR titration spectra of host–guest complexes, progress of photoreactions as monitored by 1H NMR, LC-DAD-MS. See DOI: 10.1039/ c9pp00260j
Rights and permissions
About this article
Cite this article
Kamatham, N., Raj, A.M., Givens, R.S. et al. Supramolecular photochemistry of encapsulated caged ortho-nitrobenzyl triggers†. Photochem Photobiol Sci 18, 2411–2420 (2019). https://doi.org/10.1039/c9pp00260j
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00260j