Abstract
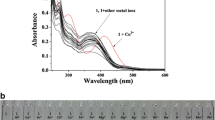
A new Schiff-base colorimetric chemosensor 1 was developed for the detection of Cu2+, Co2+ and S2−. Sensor 1 could simply monitor Cu2+ and Co2+ by a color change from colorless to yellow. The binding modes of 1 to Cu2+ and Co2+ were determined to be a 2: 1 complexation stoichiometry through Job’s plot and ESI-mass spectrometry analysis. The detection limits (0.02 μM and 0.63 μM) for Cu2+ and Co2+ were lower than the recommended values (31.5 μM and 1.7 μM) by the World Health Organization (WHO) for Cu2+ and the Environmental Protection Agency (EPA) for Co2+, respectively. Importantly, 1 could detect and quantify Cu2+ in real water samples. In addition, the Cu2+-2·1 complex could be used as a highly selective colorimetric sensor for S2− in the presence of other anions without any interference. Moreover, the sensing mechanisms of Cu2+ and Co2+ by 1 were explained by theoretical calculations.
Similar content being viewed by others
References
J. A. Cotruvo Jr., A. T. Aron, K. M. Ramos-Torres, and C. J. Chang, Synthetic fluorescent probes for studying copper in biological systems, Chem. Soc. Rev., 2015, 44, 4400–4414.
K. Kaur, R. Saini, A. Kumar, V. Luxami, N. Kaur, P. Singh, and S. Kumar, Chemodosimeters: An approach for detection and estimation of biologically and medically relevant metal ions, anions and thiols, Coord. Chem. Rev., 2012, 256, 1992–2028.
W. Wang, A. Fu, J. You, G. Gao, J. Lan, and L. Chen, Squaraine-based colorimetric and fluorescent sensors for Cu2+-specific detection and fluorescence imaging in living cells, Tetrahedron., 2010, 66, 3695–3701.
P. Kaur, S. Kaur, and K. Singh, Colorimetric detection of cyanide in water using a highly selective Cu2+ chemosensor, Inorg. Chem. Commun., 2009, 12, 978–981.
K. J. Barnham, C. L. Masters, and A. I. Bush, Neurodegenerative diseases and oxidative stress, Nat. Rev. Drug Discovery., 2004, 3, 205–214.
Y. K. Jang, U. C. Nam, H. L. Kwon, I. H. Hwang, and C. Kim, A selective colorimetric and fluorescent chemosensor based-on naphthol for detection of Al3+ and Cu2+, Dyes Pigm., 2013, 99, 6–13.
S. Goswami, D. Sen, N. K. Das, and G. Hazra, Highly selective colorimetric fluorescence sensor for Cu2+: cation-induced “switching on” of fluorescence due to excited state internal charge transfer in the red/near-infrared region of emission spectra, Tetrahedron Lett., 2010, 51, 5563–5566.
S. Yin, V. Leen, S. Van Snick, N. Boens, and W. Dehaen, A highly sensitive, selective, colorimetric and near-infrared fluorescent turn-on chemosensor for Cu2+ based on BODIPY, Chem. Commun., 2010, 46, 6329–6331.
H. S. Jung, P. S. Kwon, J. W. Lee, J. Il Kim, C. S. Hong, J. W. Kim, S. Yan, J. Y. Lee, J. H. Lee, T. Joo, and J. S. Kim, Coumarin-Derived Cu2+ -Selective Fluorescence Sensor: Synthesis, Mechanisms, and Applications in Living Cells, J. Am. Chem. Soc., 2009, 131, 2008–2012.
D. Maity, A. Raj, D. Karthigeyan, T. K. Kundu, T. Govindaraju, C. J. McKenzie, K. D. Karlin, and C. J. Fahrni, Reaction-based probes for Co(II) and Cu(I) with dual output modes: fluorescence live cell imaging, RSC Adv., 2013, 3, 16788–16794.
C.-Y. Li, X.-B. Zhang, Z. Jin, R. Han, G.-L. Shen, and R.-Q. Yu, A fluorescent chemosensor for cobalt ions based on a multi-substituted phenol-ruthenium(II) tris(bipyridine) complex, Anal. Chim. Acta., 2006, 580, 143–148.
K. Al-Habsi, E. H. Johnson, I. T. Kadim, A. Srikandakumar, K. Annamalai, R. Al-Busaidy, and O. Mahgoub, Effects of low concentrations of dietary cobalt on liveweight gains, haematology, serum vitamin B12 and biochemistry of Omani goats, Vet. J., 2007, 173, 131–137.
A. Frank, J. McPartlin, and R. Danielsson, Nova Scotia moose mystery-a moose sickness related to cobalt- and vitamin B12 deficiency, Sci. Total Environ., 2004, 318, 89–100.
K. Y. Ryu, S. Y. Lee, D. Y. Park, S. Y. Kim, and C. Kim, A novel colorimetric chemosensor for detection of Co2+ and S2− in an aqueous environment, Sens. Actuators, B., 2017, 242, 792–800.
E. J. Song, G. J. Park, J. J. Lee, S. Lee, I. Noh, Y. Kim, S. J. Kim, C. Kim, and R. G. Harrison, A fluorescence sensor for Zn2+ that also acts as a visible sensor for Co2+ and Cu2+, Sens. Actuators, B., 2015, 213, 268–275.
J. H. Ye, L. J. Duan, and L. L. Jin, A Fluorescence Sensor for Cu2+ and Co2+ Based on Click-Generated Triazole Moiety, Adv. Mater. Res., 2012, 554–556, 2045–2048.
L. Tang, M. Cai, Z. Huang, K. Zhong, S. Hou, Y. Bian, and R. Nandhakumar, Rapid and highly selective relay recognition of Cu(II) and sulfide ions by a simple benzimidazole-based fluorescent sensor in water, Sens. Actuators, B., 2013, 185, 188–194.
J. J. Lee, Y. W. Choi, G. R. You, S. Y. Lee, and C. Kim, A phthalazine-based two-in-one chromogenic receptor for detecting Co2+ and Cu2+ in an aqueous environment, Dalton Trans., 2015, 44, 13305–13314.
H. J. Jang, T. G. Jo, and C. Kim, A single colorimetric sensor for multiple targets: the sequential detection of Co2+ and cyanide and the selective detection of Cu2+ in aqueous solution, RSC Adv., 2017, 7, 17650–17659.
G. K. Patra, R. Chandra, A. Ghorai, and K. K. Shrivas, A highly selective benzildihydrazone based Schiff base chromogenic chemosensor for rapid detection of Cu2+ in aqueous solution, Inorg. Chim. Acta., 2017, 462, 315–322.
G. Dhaka, N. Kaur, and J. Singh, Spectral studies on benzimidazole-based “bare-eye” probe for the detection of Ni2+: Application as a solid state sensor, Inorg. Chim. Acta., 2017, 464, 18–22.
A. K. Mahapatra, G. Hazra, N. K. Das, and S. Goswami, A highly selective triphenylamine-based indolylmethane derivatives as colorimetric and turn-off fluorimetric sensor toward Cu2+ detection by deprotonation of secondary amines, Sens. Actuators, B., 2011, 156, 456–462.
D. Maity, A. K. Manna, D. Karthigeyan, T. K. Kundu, S. K. Pati, and T. Govindaraju, Visible-Near-Infrared and Fluorescent Copper Sensors Based on Julolidine Conjugates: Selective Detection and Fluorescence Imaging in Living Cells, Chem.–Eur. J., 2011, 17, 11152–11161.
D. Maity, and T. Govindaraju, Highly Selective Colorimetric Chemosensor for Co2+, Inorg. Chem., 2011, 50, 11282–11284.
D. Maity, B. Sarkar, S. Maiti, and T. Govindaraju, A Highly Selective Reaction-Based Two-Photon Probe for Copper(I) in Aqueous Media, ChemPlusChem., 2013, 78, 785–788.
D. Maity, A. Raj, D. Karthigeyan, T. K. Kundu, and T. Govindaraju, A switch-on near-infrared fluorescence-ready probe for Cu(I): live cell imaging, Supramol. Chem., 2015, 27, 589–594.
D. Maity, V. Kumar, and T. Govindaraju, Reactive Probes for Ratiometric Detection of Co2+ and Cu+ Based on ESIPT Mechanism, Org. Lett., 2012, 14, 6008–6011.
X. Cao, W. Lin, and L. He, A Near-Infrared Fluorescence Turn-On Sensor for Sulfide Anions, Org. Lett., 2011, 13, 4716–4719.
L. Tang, X. Dai, X. Wen, D. Wu, and Q. Zhang, A rhodamine-benzothiazole conjugated sensor for colorimetric, ratiometric and sequential recognition of copper(II) and sulfide in aqueous media, Spectrochim. Acta, Part A., 2015, 139, 329–334.
L. Tang, P. Zhou, Q. Zhang, Z. Huang, J. Zhao, and M. Cai, A simple quinoline derivatized thiosemicarbazone as a colorimetic and fluorescent sensor for relay recognition of Cu2+ and sulfide in aqueous solution, Inorg. Chem. Commun., 2013, 36, 100–104.
S. Y. Lee, and C. Kim, A colorimetric chemosensor for sulfide in a near-perfect aqueous solution: practical application using a test kit, RSC Adv., 2016, 6, 85091–85099.
J. Chen, and K. C. Teo, Determination of cadmium, copper, lead and zinc in water samples by flame atomic absorption spectrometry after cloud point extraction, Anal. Chim. Acta., 2001, 450, 215–222.
A.-C. Liu, D. Chen, C.-C. Lin, H.-H. Chou, and C. Chen, Application of Cysteine Monolayers for Electrochemical Determination of Sub-ppb Copper(II), Anal. Chem., 1999, 71, 1549–1552.
Y. Zheng, Q. Huo, P. Kele, F. M. Andreopoulos, S. M. Pham, and R. M. Leblanc, A new fluorescent chemosensor for copper ions based on tripeptide glycyl-histidyl-lysine (GHK), Org. Lett., 2001, 3, 3277–3280.
G. R. You, G. J. Park, J. J. Lee, and C. Kim, A colorimetric sensor for the sequential detection of Cu2+ and CN− in fully aqueous media: practical performance of Cu2+, Dalton Trans., 2015, 44, 9120–9129.
L. Jiao, J. Li, S. Zhang, C. Wei, E. Hao, M. G. H. Vicente, H. Zheng, Z. Zuo, C. Ouyang, H. Liu, Y. Li, and D. J. Zhu, A selective fluorescent sensor for imaging Cu2+ in living cells, New J. Chem., 2009, 33, 1888–1893.
X. Zhou, G. Li, P. Yang, L. Zhao, T. Deng, H. Shen, Z. Yang, Z. Tian, and Y. Chen, A switching sensor of C-H bond breakage/formation regulated by mediating copper(II)’s complexation, Sens. Actuators, B., 2017, 242, 56–62.
A. Perry, and D. Miles, An off-the-shelf sensor for colourimetric detection of sulfide, Tetrahedron Lett., 2016, 57, 5788–5793.
H. Tavallali, G. Deilamy-Rad, A. Moaddeli, and K. Asghari, A new pincer-type “naked-eye” colorimetric probe for Cu2+ determination in 80% water media and its application as a solid state sensor and an efficient antibacterial product, Sens. Actuators, B, 2017, 1121–1128.
I. J. Chang, M. G. Choi, Y. A. Jeong, S. H. Lee, and S.-K. Chang, Colorimetric determination of Cu2+ in simulated wastewater using naphthalimide-based Schiff base, Tetrahedron Lett., 2017, 58, 474–477.
G. Yu, Y. Cao, H. Liu, Q. Wu, Q. Hu, B. Jiang, and Z. Yuan, A spirobenzopyran-based multifunctional chemosensor for the chromogenic sensing of Cu2+ and fluorescent sensing of hydrazine with practical applications, Sens. Actuators, B., 2017, 245, 803–814.
P. X. Pei, J. H. Hu, Y. Chen, Y. Sun, and J. Qi, A novel dual-channel chemosensor for CN− using asymmetric double-azine derivatives in aqueous media and its application in bitter almond, Spectrochim. Acta, Part A., 2017, 181, 131–136.
Z. Yan, Q. Zhao, M. Wen, L. Hu, X. Zhang, and J. You, A novel polydentate ligand chromophore for simultaneously colorimetric detection of trace Ag+ and Fe3+, Spectrochim. Acta, Part A., 2017, 186, 17–22.
D. Maity, and T. Govindaraju, A turn-on NIR fluorescence and colourimetric cyanine probe for monitoring the thiol content in serum and the glutathione reductase assisted glutathione redox process, Org. Biomol. Chem., 2013, 11, 2098–2104.
D. Maity, A. Raj, P. K. Samanta, D. Karthigeyan, T. K. Kundu, S. K. Pati, and T. Govindaraju, A probe for ratiometric near-infrared fluorescence and colorimetric hydrogen sulfide detection and imaging in live cells, RSC Adv., 2014, 4, 11147–11151.
X. Qi, E. J. Jun, L. Xu, S.-J. Kim, J. S. J. Hong, Y. J. Yoon, and J. Yoon, New BODIPY derivatives as OFF-ON fluorescent chemosensor and fluorescent chemodosimeter for Cu2+: cooperative selectivity enhancement toward Cu2+, J. Org. Chem., 2006, 71, 2881–2884.
K. B. Kim, H. Kim, E. J. Song, S. Kim, I. Noh, and C. Kim, A cap-type Schiff base acting as a fluorescence sensor for zinc(II) and a colorimetric sensor for iron(II), copper(II), and zinc(II) in aqueous media, Dalton Trans., 2013, 42, 16569–16577.
C.-F. Lin, W.-S. Huang, H.-H. Chou, and J. T. Lin, Synthesis and characterization of cyclometalated iridium(III) complexes containing pyrimidine-based ligands, J. Organomet. Chem., 2009, 694, 2757–2769.
C. H. Min, S. Na, J. E. Shin, J. K. Kim, T. G. Jo, and C. Kim, A new Schiff-based chemosensor for chromogenic sensing of Cu2+, Co2+ and S2− in aqueous solution: experimental and theoretical studies, New J. Chem., 2017, 41, 3991–3999.
N. Roy, S. Nath, A. Dutta, P. Mondal, P. C. Paul, and T. S. Singh, A highly efficient and selective coumarin based fluorescent probe for colorimetric detection of Fe3+ and fluorescence dual sensing of Zn2+ and Cu2+, RSC Adv., 2016, 6, 63837–63847.
G. J. Park, J. J. Lee, G. R. You, L. Nguyen, I. Noh, and C. Kim, A dual chemosensor for Zn2+ and Co2+ in aqueous media and living cells: Experimental and theoretical studies, Sens. Actuators, B., 2016, 223, 509–519.
A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5652.
C. Lee, W. Yang, and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B: Condens. Matter., 1988, 37, 785–789.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, J. A. Montgomery Jr., T. Vreven, K. N. Kudin, J. C. Burant, J. M. Millam, S. S. Iyengar, J. Tomasi, V. Barone, B. Mennucci, M. Cossi, G. Scalmani, N. Rega, G. A. Petersson, H. Nakatsuji, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, M. Klene, X. Li, J. E. Knox, H. P. Hratchian, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, P. Y. Ayala, K. Morokuma, G. A. Voth, P. Salvador, J. J. Dannenberg, V. G. Zakrzewski, S. Dapprich, A. D. Daniels, M. C. Strain, O. Farkas, D. K. Malick, A. D. Rabuck, K. Raghavachari, J. B. Foresman, J. V. Ortiz, Q. Cui, A. G. Baboul, S. Clifford, J. Cioslowski, B. B. Stefanov, G. Liu, A. Liashenko, P. Piskorz, I. Komaromi, R. L. Martin, D. J. Fox, T. Keith, M. A. Al-Laham, C. Y. Peng, A. Nanayakkara, M. Challacombe, P. M. W. Gill, B. Johnson, W. Chen, M. W. Wong, C. Gonzalez and J. A. Pople, Gaussian 03, revision B.02, Gaussian, Inc., Wallingford CT, 2004.
P. C. Hariharan, and J. A. Pople, The influence of polarization functions on molecular orbital hydrogenation energies, Theor. Chim. Acta., 1973, 28, 213–222.
M. M. Francl, W. J. Pietro, W. J. Hehre, J. S. Binkley, M. S. Gordon, D. J. DeFrees, and J. A. Pople, Self-Consistent Molecular Orbital Methods. 23. A polarization-type basis set for 2nd-row elements, J. Chem. Phys., 1982, 77, 3654–3665.
P. J. Hay, and W. R. Wadt, Ab initio effective core potentials for molecular calculations. Potentials for the transition metal atoms Sc to Hg, J. Chem. Phys., 1985, 82, 270.
W. R. Wadt, and P. J. Hay, Ab initio effective core potentials for molecular calculations. Potentials for main group elements Na to Bi, J. Chem. Phys., 1985, 82, 284–298.
W. R. Wadt, and P. J. Hay, Ab initio effective core potentials for molecular calculations. Potentials for K to Au including the outermost core orbitals, J. Chem. Phys., 1985, 82, 299–310.
V. Barone, and M. Cossi, Quantum calculation of molecular energies and energy gradients in solution by a conductor solvent model, J. Phys. Chem. A., 1998, 102, 1995–2001.
M. Cossi, and V. Barone, Time-dependent density functional theory for molecules in liquid solutions, J. Chem. Phys., 2001, 115, 4708.
N. M. O’Boyle, A. L. Tenderholt, and K. M. Langner, cclib: a library for package-independent computational chemistry algorithms, J. Comput. Chem., 2008, 29, 839–845.
P. Job, Formation and Stability of Inorganic Complexes in Solution, Ann. Chim., 1928, 9, 113–203.
G. Grynkiewicz, M. Poenie, and R. Y. Tsien, A new generation of Ca2+ indicators with greatly improved fluorescence properties, J. Biol. Chem., 1985, 260, 3440–3450.
L. Tang, J. Zhao, M. Cai, P. Zhou, K. Zhong, S. Hou, and Y. Bian, An efficient sensor for relay recognition of Zn2+ and Cu2+ through fluorescence “off–on–off” functionality, Tetrahedron Lett., 2013, 54, 6105–6109.
S. Goswami, S. Maity, A. K. Das, and A. C. Maity, Single chemosensor for highly selective colorimetric and fluorometric dual sensing of Cu(II) as well as “NIRF” to acetate ion, Tetrahedron Lett., 2013, 54, 6631–6634.
Y.-K. Tsui, S. Devaraj, and Y.-P. Yen, Azo dyes featuring with nitrobenzoxadiazole (NBD) unit: A new selective chromogenic and fluorogenic sensor for cyanide ion, Sens. Actuators, B., 2012, 161, 510–519.
B. Gordon, P. Callan, and C. Vickers, WHO guidelines for drinking-water quality, WHO Chron., 2008, 38, 564.
F. A. Abebe, C. S. Eribal, G. Ramakrishna, and E. Sinn, A “turn-on” fluorescent sensor for the selective detection of cobalt and nickel ions in aqueous media, Tetrahedron Lett., 2011, 52, 5554–5558.
C.-Y. Tsai, and Y.-W. Lin, A highly selective and sensitive fluorescence assay for determination of copper(II) and cobalt(II) ions in environmental water and toner samples, Analyst., 2013, 138, 1232–1238.
J. M. Jung, S. Y. Lee, and C. Kim, A novel colorimetric chemosensor for multiple target metal ions Fe2+, Co2+, and Cu2+ in a near-perfect aqueous solution: Experimental and theoretical studies, Sens. Actuators, B., 2017, 251, 291–301.
D. Mahendiran, R. S. Kumar, V. Viswanathan, D. Velmurugan, and A. K. Rahiman, Targeting of DNA molecules, BSA/c-Met tyrosine kinase receptors and anti-proliferative activity of bis(terpyridine)copper(II) complexes, Dalton Trans., 2016, 45, 7794–7814.
R. Sahu, and V. Manivannan, Syntheses and molecular structures of Co3+-Na+ and Co3+-K+ coordination polymers constructed using mono- and bis-chelated cobalt(III) complexes of bis(2-pyridylcarbonyl)amide ion, Inorg. Chim. Acta., 2010, 363, 4008–4016.
G. J. Park, I. H. Hwang, E. J. Song, H. Kim, and C. Kim, A colorimetric and fluorescent sensor for sequential detection of copper ion and cyanide, Tetrahedron., 2014, 70, 2822–2828.
J. H. Kang, S. Y. Lee, H. M. Ahn, and C. Kim, Sequential detection of copper(II) and cyanide by a simple colorimetric chemosensor, Inorg. Chem. Commun., 2016, 74, 62–65.
As we proved with several mechanistic studies, the Co2+-2·1 complex is oxidized to the Co3+-2·1 complex in air. Co3+ would bind more strongly to sensor 1 than Co2+ or Cu2+. Therefore, it might be difficult for S2− to bind to Co3+. Based on this assumption, the cobalt complex of 1 is unable to detect S2−
H. A. Benesi, and J. H. Hildebrand, A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons, J. Am. Chem. Soc., 1949, 71, 2703–2707.
World Health Organization, pH in Drinking-water Background document for development of, pH Drink., 1996, 2, 7.
Acknowledgments
Financial support from the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2015R1A2A2A09001301) is gratefully acknowledged. This work was also supported by Korea Environment Industry & Technology Institute (KEITI) through “The Chemical Accident Prevention Technology Development Project”, funded by Korea Ministry of Environment (MOE) (no. 2016001970001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, M.S., Lee, S.Y., Jung, J.M. et al. A new Schiff-base chemosensor for selective detection of Cu2+ and Co2+ and its copper complex for colorimetric sensing of S2− in aqueous solution. Photochem Photobiol Sci 16, 1677–1689 (2017). https://doi.org/10.1039/c7pp00229g
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c7pp00229g