Abstract
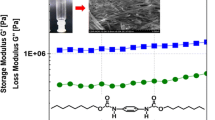
In this article we present dual-component charge-transfer interaction (CT) induced organogel formation with bile acid anthracene conjugates as donors and 2,4,7-trinitrofluorenone (TNF) as the acceptor. The use of TNF (1) as a versatile electron acceptor in the formation of gels is demonstrated through the formation of gels with different steroidal groups on the anthracene moiety in a variety of solvents ranging from aromatic hydrocarbons to long chain alcohols. Thermal stability and variable temperature fluorescence experiments were performed on these CT gels. Dynamic rheological experiments conducted on these gels suggest that these are viscoelastic soft materials and with the gel strength can be modulated by varying the donor/acceptor ratios.
Similar content being viewed by others
Notes and references
Molecular Gels, Materials with Self-assembled Fibrillar Networks, ed. R. G. Weiss and P. Terech, Springer, 2006.
S. Banerjee, R. K. Das and U. Maitra, J. Mater. Chem. 2009, 19, 6649.
T. Kitahara, M. Shirakawa, S.-I. Kawano, U. Beginn, N. Fujita and S. Shinkai, J. Am. Chem. Soc. 2005, 127, 14980.
T. Katakabe, T. Kaneko, M. Watanabe, T. Fukushima and T. Aida, J. Electrochem. Soc. 2005, 152, A1913.
J. P. Luis, A. P. del Pino, E. Laukhina, J. Esquena, V. Laukhin, C. Rovira, J. V. Gancedo, A. G. Kanaras, R. J. Nichols, M. Brust and D. B. Amabilino, Angew. Chem., Int. Ed. 2008, 47, 1861.
K. Sugiyasu, S. Kawano, N. Fujita and S. Shinkai, Chem. Mater. 2008, 20, 2863.
H. Maeda, Chem. Eur. J. 2008, 14, 11274.
J. A. Foster, M. M. Piepenbrock, G. O. Lloyd, N. Clarke, J. A. K. Howard and J. W. Steed, Nature 2010, 2, 1037.
G. O. Lloyd and J. W. Steed, Nature 2009, 1, 2009.
S. Dutta, A. Shome, T. Kar and P. K. Das, Langmuir 2011, 27, 5000.
X. Li, J. Li, Y. Gao, Y. Kuang, J. Shi and B. Xu, J. Am. Chem. Soc. 2010, 132, 17707.
Y. Gao, Y. Kuang, Z. Guo, I. J. Krauss and B. Xu, J. Am. Chem. Soc. 2009, 131, 13576.
B. Xu, Langmuir 2009, 25, 8375.
Y. Qiao, Y. Wang, Z. Yang, Y. Lin and J. B. Huang, Chem. Mater. 2011, 23, 1182.
Y. Qiao, Y. Lin, Z. Yang, H. Chen, S. Zhang, Y. Yan and J. B. Huang, J. Phys. Chem. B 2010, 114, 11725.
Y. Qiao, H. Chen, Y. Lin, Z. Yang, X. Cheng and J. B. Huang, J. Phys. Chem. C 2011, 15, 7323.
P. Terech and R. G. Weiss, Chem. Rev. 1997, 97, 3133.
N. M. Sangeetha and U. Maitra, Chem. Soc. Rev. 2005, 34, 821.
B. A. Simmons, C. E. Taylor, F. A. Landis, V. T. John, G. L. McPherson, D. K. Schwartz and R. Moore, J. Am. Chem. Soc. 2001, 123, 2414.
K. Sada, M. Miyata and K. Hanabusa, Chem. Lett. 2000 1170.
H. M. Willemen, T. Vermonden, A. T. M. Marcelis and E. J. R. Sudhölter, Langmuir 2002, 18, 7102.
S. W. Jeong and S. Shinkai, Nanotechnology 1997, 8, 179.
M. George and R. G. Weiss, J. Am. Chem. Soc. 2001, 123, 10393.
M. George and R. G. Weiss, Langmuir 2002, 18, 7124.
A. R. Hirst, D. K. Smith, M. C. Feiters, H. P. M. Geurts and A. C. Wright, J. Am. Chem. Soc. 2001, 123, 10393.
K. Hanabusa, T. Miki, Y. Taguchi, K. Koyama and H. Shirai, J. Chem. Soc., Chem. Commun. 1993 1382.
G. L. Tata, V. T. John, Y. Y. Waguespack and G. L. McPherson, J. Am. Chem. Soc. 1994, 116, 9464.
K. Inoue, Y. Ono, Y. Kanekiyo, T. Ishi-i, K. Yoshihara and S. Shinkai, J. Org. Chem. 1999, 64, 2933.
A. R. Hirst, D. K. Smith, M. C. Feiters, H. P. M. Geurts and A. C. Wright, J. Am. Chem. Soc. 2003, 125, 9010.
P. V. Kumar, N. Chandra, L. J. D’Souza, M. D. Prasanna, A. R. Raju and U. Maitra, Chem. Commun. 1999 595.
K. V. Rao, K. Jayaramulu, T. K. Maji and S. J. George, Angew. Chem., Int. Ed. 2010, 49, 4218.
X. Mei and J. Ouyang, Langmuir 2011, 27, 10953.
C. Wang, D. Zhang and D. Zhu, J. Am. Chem. Soc. 2005, 127, 16372.
B. Legouin, P. Uriac, S. Tomasi, L. Toupet, A. Bondon and P. van de Weghe, Org. Lett. 2009, 11, 745.
P. Babu, N. M. Sangeetha, P. V. Kumar, U. Maitra, K. Rissanen and A. R. Raju, Chem.–Eur. J. 2003 1922.
M. Orchin and E. O. Woolfolk, J. Am. Chem. Soc. 1946, 68, 1727.
N. Varghese, A. Ghosh, R. Voggu, S. Ghosh and C. N. R. Rao, J. Phys. Chem. C 2009, 113, 16855.
K. K. Kartha, S. S. Babu, S. Srinivasan and A. Ajayaghosh, J. Am. Chem. Soc. 2012, 134, 4834.
T. Brotin, R. Utermöhlen, F. Fages, H. Bouas-Laurent and J.-P. Desvergne, J. Chem. Soc., Chem. Commun. 1991 416.
P. Terech, H. Bouas-Laurent and J.-P. Desvergne, J. Colloid Interface Sci. 1995, 174, 258.
F. Placin, J.-P. Desvergne, C. Belin, T. Buffeteau, B. Desbat, L. Ducasse and J.-C. Lassègues, Langmuir 2003, 19, 4563.
A. Del Guerzo, A. G. L. Olive, J. Reichwagen, H. Hopf and J.-P. Desvergne, J. Am. Chem. Soc. 2005, 127, 17984.
N. M. Sangeetha, S. Bhat, G. Raffy, C. Belin, A. Loppinet-Serani, C. Aymonier, P. Terech, U. Maitra, J.-P. Desvergne and A. Del Guerzo, Chem. Mater. 2009, 21, 3424.
R. K. Das, S. Bhat, S. Banerjee, C. Aymonier, A. Loppinet-Serani, P. Terech, U. Maitra, G. Raffy, J.-P. Desvergne and A. Del Guerzo, J. Mater. Chem. 2011, 21, 2740.
H. Bouas-Laurent, A. Castellan, J.-P. Desvergne and R. Lapouyade, Chem. Soc. Rev. 2000, 29, 43.
H. Bouas-Laurent, A. Castellan, J.-P. Desvergne and R. Lapouyade, Chem. Soc. Rev. 2001, 30, 248.
F. Placin, J.-P. Desvergne and F. Cancell, J. Mater. Chem. 2000, 10, 2147.
R. K. Das, S. Banerjee, G. Raffy, A. Del Guerzo, J.-P. Desvergne and U. Maitra, J. Mater. Chem. 2010, 20, 7227.
S. Mukhopadhyay and U. Maitra, Curr. Sci. 2004, 87, 1666.
S. Mukhopadhyay, A. Sarkar, P. Rao, S. S. Indi and U. Maitra, Angew. Chem., Int. Ed. 2001, 40, 2281.
N. M. Sangeetha, S. Bhat, A. R. Choudhury, U. Maitra and P. Terech, J. Phys. Chem. B 2004, 108, 16056.
P. Terech, N. M. Sangeetha and U. Maitra, J. Phys. Chem. B 2006, 110, 15224.
N. M. Sangeetha and U. Maitra, Macromol. Symp. 2006, 241, 60.
P. Babu, D. Chopra, T. N. Guru Row and U. Maitra, Org. Biomol. Chem. 2005, 3, 3695.
Y. Hishikawa, K. Sada, R. Watanabe, M. Miyata and K. Hanabusa, Chem. Lett. 1998 795.
H. M. Willemen, T. A. Vermonden, T. M. Marcelis and E. J. R. Sudhölter, Eur. J. Org. Chem. 2001 2329.
H. M. Willemen, T. A. Vermonden, T. M. Marcelis and E. J. R. Sudhölter, Langmuir 2002, 18, 7102.
H. M. Willemen, T. M. Marcelis, E. J. R. Sudhölter, W. G. Bouwman, B. Demé and P. Terech, Langmuir 2004, 20, 2075.
V. Noponen, Nonappa, M. Lahtinen, A. Valkonen, H. Salo, E. Kolehmainen and E. Sievänen, Soft Matter 2010, 6, 3789.
Nonappa and U. Maitra, Soft Matter 2007, 3, 1728.
S. Banerjee, V. M. Vidya, A. J. Savyasachi and U. Maitra, J. Mater. Chem. 2011, 21, 14693.
N. M. Sangeetha, S. Bhat, A. R. Choudhury, U. Maitra and P. Terech, J. Phys. Chem. B 2004, 108, 16056.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published as part of a themed issue in honour of Jean-Pierre Desvergne on the occasion of his 65th birthday.
Electronic supplementary information (ESI) available: Characterization data for 2, 3 and 4 are available. Details of gel preparation for Tgel, absorption, emission, rheological and SEM studies are also mentioned. See DOI: 10.1039/c2pp25088h
Rights and permissions
About this article
Cite this article
Kandanelli, R., Maitra, U. Charge-transfer interaction mediated organogels from bile acid appended anthracenes: rheological and microscopic studies. Photochem Photobiol Sci 11, 1724–1729 (2012). https://doi.org/10.1039/c2pp25088h
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c2pp25088h