Abstract
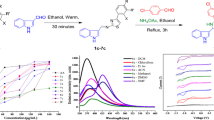
A series of three substituted triazole appended NDI-derivatives, 2,7-bis(3,5-di(pyridin-X-yl)-4H-1,2,4-triazol-4-yl)benzo[lmn][3,8]phenanthroline-1,3,6,8(2H,7H)-tetraone (where X = 2, NDI-PyTz-1; 3, NDI-PyTz-2; and 4, NDI-PyTz-3), were designed, synthesized and well characterized using various analytical and spectroscopic techniques. All the three NDI-PyTz derivatives exhibit decent electronic properties as suggested by DFT, cyclic voltammetry and fluorescence studies. In particular, NDI-PyTz-1 demonstrated the generation of a stable anion radical [NDI-PyTz-1]•−.
Similar content being viewed by others
References
N. Tamai and H. Miyasaka, Ultrafast Dynamics of Photochromic Systems, Chem. Rev., 2000, 100,1875–1890.
R. Pardo, M. Zayat and D. Levy, Photochromic organic-inorganic hybrid materials, Chem. Soc. Rev., 2011, 40, 672–687.
H. Dong, H. Zhu, Q. Meng, X. Gong and W. Hu, Organic photoresponse materials and devices, Chem. Soc. Rev., 2012, 41, 1754–1808.
A. Fihey, A. Perrier, W. R. Browne and D. Jacquemin, Multiphotochromic molecular systems, Chem. Soc. Rev., 2015, 44, 3719–3759.
G. Kumar and R. Gupta, Molecularly designed architectures -the metalloligand way, Chem. Soc. Rev., 2013, 42, 9403–9453.
S. Kawata and Y. Kawata, Three-Dimensional Optical Data Storage Using Photochromic Materials, Chem. Rev., 2000, 100, 1777–1788.
H. Tian and S. Yang, Recent progresses on diarylethene based photochromic switches, Chem. Soc. Rev., 2004, 33, 85–97.
S. Qu and H. Tian, Diketopyrrolopyrrole (DPP)-based materials for organic photovoltaics, Chem. Commun., 2012, 48, 3039–3051.
E. Jacques, M. Romain, A. Yassin, S. Bebiche, M. Harnois, T. Mohammed-Brahim, J. Rault-Berthelot, C. Poriel, J.-W. Park, N. Audebrand, L. Vignau, N. Huby, G. Wantz and L. Hirsch, An electron deficient dicyanovinylene-ladder-type pentaphenylene derivative for n-type organic field effect transistors, J. Mater. Chem. C, 2014, 2, 3292–3302.
Y. Wang and T. Michinobu, Benzothiadiazole and its n-extended, heteroannulated derivatives: useful acceptor building blocks for high-performance donor-acceptor polymers in organic electronics, J. Mater. Chem. C, 2016, 4, 6200–6214.
H. Li, T. L. Tam, Y. M. Lam, S. G. Mhaisalkar and A. C. Grimsdale, Synthesis of Low Band Gap [1,2,5]-Thiadiazolo[3,4 g]quinoxaline and Pyrazino[2,3 g]quinoxa-line Derivatives by Selective Reduction of Benzo[1,2-c;4,5-c'] bis[1,2,5]thiadiazole, Org. Lett., 2011, 13, 46–49.
E. Kozma and M. Catellani, Perylene diimides based materials for organic solar cells, Dyes Pigm., 2013, 98, 160–179.
D. Zhao, Q. Wu, Z. Cai, T. Zheng, W. Chen, J. Lu and L. Yu, Electron Acceptors Based on a-Substituted Perylene Diimide (PDI) for Organic Solar Cells, Chem. Mater., 2016, 28, 1139–1146.
W. Chen, X. Yang, G. Long, X. Wan, Y. Chen and Q. Zhang, A perylene diimide (PDI)-based small molecule with tetrahedral configuration as a non-fullerene acceptor for organic solar cells, J. Mater. Chem. C, 2015, 3, 4698–4705.
R. Stalder, J. Mei, K. R. Graham, L. A. Estrada and J. R. Reynolds, Isoindigo, a Versatile Electron-Deficient Unit For High-Performance Organic Electronics, Chem. Mater., 2014, 26, 664–678.
S. V. Bhosale, C. H. Jani and S. J. Langford, Chemistry of naphthalene diimides, Chem. Soc. Rev., 2008, 37, 331–342.
T. Šolomek, N. E. Powers-Riggs, Y.-L. Wu, R. M. Young, M. D. Krzyaniak, N. E. Horwitz and M. R. Wasielewski, Electron Hopping and Charge Separation within a Naphthalene-1,4:5,8-bis(dicarboximide) Chiral Covalent Organic Cage, J. Am. Chem. Soc., 2017, 139, 3348–3351.
E. Ahmed, G. Ren, F. S. Kim, E. C. Hollenbeck and S. A. Jenekhe, Design of New Electron Acceptor Materials for Organic Photovoltaics: Synthesis, Electron Transport, Photophysics, and Photovoltaic Properties of Oligothiophene-Functionalized Naphthalene Diimides, Chem. Mater., 2011, 23, 4563–4577.
J. Choi, K.-H. Kim, H. Yu, C. Lee, H. Kang, I. Song, Y. Kim, J. H. Oh and B. J. Kim, Importance of Electron Transport Ability in Naphthalene Diimide-Based Polymer Acceptors for High-Performance, Additive-Free, All-Polymer Solar Cells, Chem. Mater., 2015, 27, 5230–5237.
M. Poddar, V. Sharma, S. M. Mobin and R. Misra, 1,8-Naphthalimide-Substituted BODIPY Dyads: Synthesis, Structure, Properties, and Live-Cell Imaging, Chem. - Asian J., 2018, 13, 2881–2890.
T. S. Reddy, R. Maragani, B. Dhokale, S. M. Mobin and R. Misra, Heteroatom-connected ferrocenyl substituted naphthalimides, RSC Adv., 2016, 6, 7746–7754.
B. Dhokale, T. Jadhav, Y. Patil and R. Misra, Symmetrical and unsymmetrical ferrocenyl perylenediimides: Design, synthesis and properties, Dyes Pigm., 2016, 134, 164–170.
M. Al Kobaisi, S. V. Bhosale, K. Latham, A. M. Raynor and S. V. Bhosale, Functional Naphthalene Diimides: Synthesis, Properties, and Applications, Chem. Rev., 2016, 116, 11685–11796.
A. Baron, C. Herrero, A. Quaranta, M.-F. Charlot, W. Leibl, B. Vauzeilles and A. Aukauloo, Efficient electron transfer through a triazole link in ruthenium(II) polypyridine type complexes, Chem. Commun., 2011, 47, 11011–11013.
E.-C. Yang, Z.-Y. Liu, C.-H. Zhang, Y.-L. Yang and X.-J. Zhao, Structural diversity directed by switchable coordination of substitute groups in a ternary Cu(n)-triazole-sulfoisophthalate self-assembly system: synthesis, crystal structures and magnetic behavior, Dalton Trans., 2013, 42, 1581–1590.
J. M. Rawson, L. Donato and E. Zysman-Colman, Triazole-directed hydrogen-bonded structures of cationic iridium(III) complexes, CrystEngComm, 2014, 16, 8531–8536.
B. Schulze and U. S. Schubert, Beyond click chemistry - supramolecular interactions of 1,2,3-triazoles, Chem. Soc. Rev., 2014, 43, 2522–2571.
M. Pan, X.-M. Lin, G.-B. Li and C.-Y. Su, Progress in the study of metal-organic materials applying naphthalene diimide (NDI) ligands, Coord. Chem. Rev., 2011, 255, 1921–1936.
D. D. Perrin, W. L. F. Armarego and D. R. Perrin, Purification of Laboratory Chemicals, Pergamon Press, Oxford, 1980.
SAINT ver. 8.34A, Bruker AXS Inc., Madison, WI, 2014.
G. M. Sheldrick, A short history of SHELX, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112–122.
A. L. Spek, Structure validation in chemical crystallography, Acta Crystallogr., Sect. D: Biol. Crystallogr., 2009, 65, 148–155.
C. Lee, W. Yang and R. G. Parr, Development of the Colle-Salvetti correlation-energy formula into a functional of the electron density, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785–789.
A. D. Becke, Density-functional thermochemistry. III. The role of exact exchange, J. Chem. Phys., 1993, 98, 5648–5652.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, GAUSSIAN 09, Gaussian, Inc., Wallingford, CT, 2009.
V. Barone and M. Cossi, Quantum Calculation of Molecular Energies and Energy Gradients in Solution by a Conductor Solvent Model, J. Phys. Chem. A, 1998, 102, 1995–2001.
M. Cossi, N. Rega, G. Scalmani and V. Barone, Energies, structures, and electronic properties of molecules in solution with the C-PCM solvation model, J. Comput. Chem., 2003, 24, 669–681.
A. Klamt and G. Schuurmann, COSMO: a new approach to dielectric screening in solvents with explicit expressions for the screening energy and its gradient, Perkin Trans. 2, 1993, 799–805.
A. Schafer, A. Klamt, D. Sattel, J. C. W. Lohrenz and F. Eckert, COSMO Implementation in TURBOMOLE: Extension of an efficient quantum chemical code towards liquid systems, Phys. Chem. Chem. Phys., 2000, 2, 2187–2193.
P. H. Dinolfo, M. E. Williams, C. L. Stern and J. T. Hupp, Rhenium-Based Molecular Rectangles as Frameworks for Ligand-Centered Mixed Valency and Optical Electron Transfer, J. Am. Chem. Soc., 2004, 126, 12989–13001.
M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, P. R. Spackman, D. Jayatilaka and M. A. Spackman, CrystalExplorer17, University of Western Australia, 2017.
R. Rybakiewicz, E. D. Glowacki, L. Skorka, S. Pluczyk, P. Zassowski, D. H. Apaydin, M. Lapkowski, M. Zagorska and A. Pron, Low and High Molecular Mass Dithienopyrrole-Naphthalene Bisimide Donor-Acceptor Compounds: Synthesis, Electrochemical and Spectroelectrochemical Behaviour, Chem. - Eur.J., 2017, 23, 2839–2851.
S. Guha and S. Saha, Fluoride Ion Sensing by an Anion-n Interaction, J. Am. Chem. Soc., 2010, 132, 17674–17677.
A. T. R. Williams, S. A. Winfield and J. N. Miller, Relative fluorescence quantum yields using a computer-controlled luminescence spectrometer, Analyst, 1983, 108, 1067–1071.
E. M. Kosower, H. Dodiuk, K. Tanizawa, M. Ottolenghi and N. Orbach, Intramolecular donor-acceptor systems. Radiative and nonradiative processes for the excited states of 2-N-arylamino-6-naphthalenesulfonates, J. Am. Chem. Soc., 1975, 97, 2167–2178.
M. F. Broglia, M. L. Gómez, S. G. Bertolotti, H. A. Montejano and C. M. Previtali, Photophysical properties of safranine and phenosafranine: A comparative study by laser flash photolysis and laser induced optoacoustic spectroscopy, J. Photochem. Photobiol., A, 2005, 173, 115–120.
S. Jockusch, H.-J. Timpe, W. Schnabel and N. J. Turro, Photoinduced Energy and Electron Transfer between Ketone Triplets and Organic Dyes, J. Phys. Chem. A, 1997, 101, 440–445.
M. Y. Berezin and S. Achilefu, Fluorescence Lifetime Measurements and Biological Imaging, Chem. Rev., 2010, 110, 2641–2684.
J. René Albani, Fluorescence lifetimes of tryptophan: Structural origin and relation with So 1Lb and So 1La transitions, J. Fluoresc., 2009, 19, 1061–1071.
H. Lee, M. Y. Berezin, M. Henary, L. Strekowski and S. Achilefu, Fluorescence lifetime properties of near-infrared cyanine dyes in relation to their structures, J. Photochem. Photobiol., A, 2008, 200, 438–444.
C. M. Cardona, L. Wei, A. E. Kaifer, D. Stockdale and G. C. Bazan, Electrochemical Considerations for Determining Absolute Frontier Orbital Energy Levels of Conjugated Polymers for Solar Cell Applications, Adv. Mater., 2011, 23, 2367–2371.
B. W. D'Andrade, S. Datta, S. R. Forrest, P. Djurovich, E. Polikarpov and M. E. Thompson, Relationship between the ionization and oxidation potentials of molecular organic semiconductors, Org. Electron., 2005, 6, 11–20.
A. Mallick, B. Haldar, S. Maiti, S. C. Bera and N. Chattopadhyay, Photophysical Study of 3-Acetyl-4-oxo-6,7-dihydro-12H-indolo[2,3-a]quinolizine in Biomimetic Reverse Micellar Nanocavities: A Spectroscopic Approach, J. Phys. Chem. B, 2005, 109, 14675–14682.
Acknowledgements
G. Kumar thanks the Department of Science and Technology (DST), Government of India for the financial assistance in the form of the INSPIRE Faculty Award with Ref. No. [DST/ INSPIRE/04/2015/001300]. The authors are grateful to the UGC networking resource centre, School of Chemistry, University of Hyderabad, Hyderabad for providing the NMR, CHN, single crystal X-ray diffraction and XRD powder facilities. Dr Praveen Kumar, Department of Physics, DAV University, Jalandhar is gratefully acknowledged for providing the support for fluorescence studies. We thank Dr Mohd Afzal, Department of Chemistry, Jadavpur University, Kolkata for valuable discussions and help with the fluorescence data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, G., Guda, R., Husain, A. et al. Synthesis and photophysical properties of pyridyl conjugated triazole appended naphthalenediimide derivatives. Photochem Photobiol Sci 18, 1333–1341 (2019). https://doi.org/10.1039/c9pp00003h
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c9pp00003h