Abstract
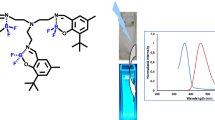
Although boron-based fluorophores incorporating nitrogenous chelating ligands have received much attention, there has been little work on examples of boron-salphen and azine derivatives. In this report, we present several it expanded boron salphen type complexes, incorporating both bis(2-hydroxy-naphthaldehyde)azine as well as ortho, meta and para variants of the analogous 2-hydroxynaphthalde-hyde salphen compounds. For the azine, we observed only the formation of a single BF2 adduct, while for the naphth-phen compounds dimeric BF2 binding was observed. All new compounds were fully characterized via X-ray diffraction, and both DFT. and TDDFT. studies were carried out to probe the electronic structures of these fluorophores
Similar content being viewed by others
References
M. S. T. Goncalves, Chem. Rev., 2009, 109, 190–212.
M. Vendrell, D. Zhai, J. C. Er and Y. Chang, Chem. Rev., 2012, 112, 439–4420.
Y. Jeong and J. Yoon, Inorg. Chim. Acta, 2012, 381, 2–14.
A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo and H. Pettersson, Chem. Rev., 2010, 110, 6595–6663.
H. Zheng, Z.-Q. Zhan, Q.-N. Bian and X.-J. Zhang, Chem. Commun., 2013, 49, 429–447.
J. Han and K. Burgess, Chem. Rev., 2010, 110, 2709–2728.
L. D. Lavis and R. T. Raines, ACS. Chem. Biol, 2008, 3, 142–155.
A. Mishra, M. K. R. Fischer and R. Bauerle, Angew. Chem., Int. Ed., 2009, 48, 2474–2499.
A. Loudet and K. Burgess, Chem. Rev., 2007, 107, 4891–4932.
R. Ziessel, G. Ulrich and A. Harriman, New J. Chem., 2007, 31, 496–501.
G. Ulrich, R. Ziessel and A. Harriman, Angew. Chem., Int. Ed., 2008, 47, 1184–1201.
N. Boens, V. Leen and W. Dehaen, Chem. Soc. Rev., 2012, 41, 1130.
A. Y. Bochkov, I. O. Akchurin, O. A. Dyachenko and V. F. Traven, Chem. Commun., 2013, 49, 11653–11655.
H. Lu, J. Mack, Y. Yang and Z. Shen, Chem. Soc. Rev., 2014, 43, 4778–4823.
S. M. Barbon, J. T. Price, U. Yogarajah and J. B. Gilroy, RSC. Adv., 2015, 5, 56316–56324.
M. Nakamura, H. Tahara, K. Takahashi, T. Nagata, H. Uoyama, D. Kuzuhara, S. Mori, T. Okujima, H. Yamada and H. Uno, Org. Biomol. Chem., 2012, 10, 6840–8649.
M. T. Whited, N. M. Patel, S. T. Roberts, K. Allen, P. I. Djurovich, S. E. Bradforth and M. E. Thompson, Chem. Commun., 2012, 48, 284–286.
L. Wang, I.-S. Tamgho, L. Crandall, J. Rack and C. Ziegler, Phys. Chem. Chem. Phys., 2014, 4–6.
C. Yu, L. Jiao, P. Zhang, Z. Feng, C. Cheng, Y. Wei, X. Mu and E. Hao, Org. Lett, 2014, 16, 3048–3051.
H. M. Rhoda, K. Chanawanno, A. J. King, Y. V. Zatsikha, C. J. Ziegler and V. N. Nemykin, Chem. - Eur. J., 2015, 21, 18043–18046.
J. Wang, Q. Wu, C. Yu, Y. Wei, X. Mu, E. Hao and L. Jiao, J. Org. Chem., 2016, 81, 11316–11323.
L. Zhou, D. Xu, H. Gao, C. Zhang, F. Ni, W. Zhao, D. Cheng, X. Liu and A. Han,J. Org. Chem., 2016, 81, 7439–7447.
I. S. Tamgho, A. Hasheminasab, J. T. Engle, V. N. Nemykin and C. J. Ziegler, J. Am. Chem. Soc, 2014, 136, 5623–5626.
H.-J. Li, W.-F. Fu, L. Li, X. Gan, W.-H. Mu, W. Chen, X.-M. Duan and H.-B. Song, Org. Lett, 2010, 12, 2924–2927.
G. Nawn, S. R. Oakley, M. B. Majewski, R. McDonald, B. O. Patrick and R. G. Hicks, Chem. Sci., 2013, 4, 612–621.
A. Combes, C. R. Acad. Fr., 1889, 108, 1252.
L. Canali and D. C. Sherrington, Chem. Soc. Rev., 1999, 28, 85–93.
P. G. Cozzi, Chem. Soc. Rev., 2004, 33, 410–421.
C. J. Whiteoak, G. Salassa and A. Kleij, Chem. Soc. Rev., 2012, 41, 622–631.
R. M. Clarke and T. Storr, Dalton Trans., 2014, 43, 9380–9391.
S. J. Wezenberg and A. W. Kleij, Angew. Chem., Int. Ed., 2008, 47, 2354–2364.
H. Kunkely and A. Vogler, Inorg. Chim. Acta, 2001, 321, 171–174.
S. Dagorne and D. A. Atwood, Chem. Rev., 2008, 108, 4037–4071.
P. Wei and D. A. Atwood, Inorg. Chem., 1997, 36, 4060–4065.
F. Umland, E. Hohaus and K. Brodte, Chem. Ber., 1973, 106, 2427–2437.
P. Gong, H. Yang, J. Sun, Z. Zhang, J. Sun, P. Xue and R. Lu, J. Mater. Chem. C, 2015, 3, 10302–10308.
Q. Hou, L. Zhao, H. Zhang, Y. Wang and S. Jiang,J. Lumin., 2007, 126, 447–451.
D. Frath, S. Azizi, G. Ulrich and R. Ziessel, Org. Lett, 2012, 14, 4774–4777.
X. Ma, J. Cheng, J. Liu, X. Zhou and H. Xiang, New J. Chem., 2014, 39, 492–500.
D. A. Saffln, K. Robeyns and Y. Garcia, RSC. Adv., 2012, 2, 11379–11388.
M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, 6. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision B.0.1, Gaussian, Inc., Wallingford CT, 2009.
Author information
Authors and Affiliations
Corresponding author
Additional information
Electronic supplementary information (ESI) available: Spectroscopic, and crystallographic data for compounds. CCDC 1523987-1523991 for 1-5. For ESI and crystallographic data in CIF. or other electronic format see DOI: 10.1039/C6pp00479b
Rights and permissions
About this article
Cite this article
Crandall, L.A., Dawadi, M.B., Burrell, T. et al. Structure and electronics in dimeric boron π expanded azine and salphen complexes. Photochem Photobiol Sci 16, 627–632 (2017). https://doi.org/10.1039/c6pp00479b
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c6pp00479b