Abstract
Exposure to the UV component of sunlight is the principal factor leading to skin cancer development. Cyclobutane pyrimidine dimers (CPD) are considered to be the most important pre-mutagenic type of DNA damage involved in skin carcinogenesis. To better understand the biological mechanisms of UV carcinogenesis, it is critical to understand the CPD distribution between the four types of dipyrimidine sites. Most of our knowledge regarding CPD distribution comes from in vitro studies or from investigations using UVC, even though we are not naturally exposed to these UV wavelengths. We exposed normal human fibroblasts and purified DNA to UVB. Using ligation-mediated PCR, we quantified the CPD formation at 952 dipyrimidine sites among the PGK1 (phosphoglycerate kinase 1), JUN, HRAS, KRAS, NRAS and TP53 genes. In cellulo, we found a CPD distribution of 27:27:25:21 for TT:CC:TC:CT. This distribution is similar to that observed in vitro. In the analysed genes, we observed some extremely frequently damaged dipyrimidine sites and many of these occurred at potentially frequently mutated sites, i.e. at dipyrimidine sites containing cytosine. Also, most of the frequently damaged dipyrimidine sites in cellulo that are not frequently damaged in vitro are found on TP53 and NRAS. This indicates that many of the frequently damaged dipyrimidine sites in cellulo are on genes frequently mutated in skin cancer. All these results support the view that CPD are the main UVB-induced mutagenic photoproducts and provide evidence of the importance of CPD formation at sites containing cytosine.
Similar content being viewed by others
Abbreviations
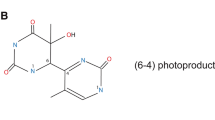
- 6-4 Photoproducts:
-
Pyrimidine (6-4) pyrimidone photoproducts
- CPD:
-
Cyclobutane pyrimidine dimers
- LMPCR:
-
Ligation-mediated polymerase chain reaction
- PGK1:
-
Phosphoglycerate kinase 1
- UV:
-
Ultraviolet
References
H. N. Ananthaswamy, W. E. Pierceall, Molecular mechanisms of ultraviolet radiation carcinogenesis, Photochem. Photobiol., 1990, 52, 1119–1136.
D. E. Brash, UV mutagenic photoproducts in Escherichia coli and human cells: a molecular genetics perspective on human skin cancer, Photochem. Photobiol., 1988, 48, 59–66.
Z. Kuluncsics, D. Perdiz, E. Brulay, B. Muel, E. Sage, Wavelength dependence of ultraviolet-induced DNA damage distribution: involvement of direct or indirect mechanisms and possible artefacts, J. Photochem. Photobiol., B, 1999, 49, 71–80.
T. Douki, D. Perdiz, P. Grof, Z. Kuluncsics, E. Moustacchi, J. Cadet, E. Sage, Oxidation of guanine in cellular DNA by solar UV radiation: biological role, Photochem. Photobiol., 1999, 70, 184–190.
D. Perdiz, P. Grof, M. Mezzina, O. Nikaido, E. Moustacchi, E. Sage, Distribution and repair of bipyrimidine photoproducts in solar UV-irradiated mammalian cells. Possible role of Dewar photoproducts in solar mutagenesis, J. Biol. Chem., 2000, 275, 26732–26742.
D. L. Mitchell, J. Jen, J. E. Cleaver, Sequence specificity of cyclobutane pyrimidine dimers in DNA treated with solar (ultraviolet B) radiation, Nucleic Acids Res., 1992, 20, 225–229.
P. J. Rochette, J. P. Therrien, R. Drouin, D. Perdiz, N. Bastien, E. A. Drobetsky, E. Sage, UVA-induced cyclobutane pyrimidine dimers form predominantly at thymine-thymine dipyrimidines and correlate with the mutation spectrum in rodent cells, Nucleic Acids Res., 2003, 31, 2786–2794.
T. Douki, A. Reynaud-Angelin, J. Cadet, E. Sage, Bipyrimidine photoproducts rather than oxidative lesions are the main type of DNA damage involved in the genotoxic effect of solar UVA radiation, Biochemistry, 2003, 42, 9221–9226.
S. Mouret, C. Baudouin, M. Charveron, A. Favier, J. Cadet, T. Douki, Cyclobutane pyrimidine dimers are predominant DNA lesions in whole human skin exposed to UVA radiation, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 13765–13770.
A. R. Young, C. S. Potten, O. Nikaido, P. G. Parsons, J. Boenders, J. M. Ramsden, C. A. Chadwick, Human melanocytes and keratinocytes exposed to UVB or UVA in vivo show comparable levels of thymine dimers, J. Invest. Dermatol., 1998, 111, 936–940.
J. Cadet, S. Mouret, J. L. Ravanat, T. Douki, Photoinduced damage to cellular DNA: direct and photosensitized reactions, Photochem. Photobiol., 2012, 88, 1048–1065.
P. M. Girard, S. Francesconi, D. Graindorge, P. J. Rochette, R. Drouin, E. Sage, UVA-induced Damage to DNA and proteins: direct versus indirect photochemical processes, J. Phys.: Conf. Ser., 2011, 261, 012002. 10.1088/1742-6596/261/1/012002
Y. Jiang, M. Rabbi, M. Kim, C. Ke, W. Lee, R. L. Clark, P. A. Mieczkowski, P. E. Marszalek, UVA generates pyrimidine dimers in DNA directly, Biophys. J., 2009, 96, 1151–1158.
S. Mouret, C. Philippe, J. Gracia-Chantegrel, A. Banyasz, S. Karpati, D. Markovitsi, T. Douki, UVA-induced cyclobutane pyrimidine dimers in DNA: a direct photochemical mechanism?, Org. Biomol. Chem., 2010, 8, 1706–1711.
W. J. Thilly, Induced mutagenesis: molecular mechanisms and their implications for environmental protection, Plenum Press, New York, 1983.
R. Drouin, J. P. Therrien, UVB-induced cyclobutane pyrimidine dimer frequency correlates with skin cancer mutational hotspots in p53, Photochem. Photobiol., 1997, 66, 719–726.
G. P. Pfeifer, Formation and processing of UV photoproducts: effects of DNA sequence and chromatin environment, Photochem. Photobiol., 1997, 65, 270–283.
D. E. Brash, S. Seetharam, K. H. Kraemer, M. M. Seidman, A. Bredberg, Photoproduct frequency is not the major determinant of UV base substitution hot spots or cold spots in human cells, Proc. Natl. Acad. Sci. U. S. A., 1987, 84, 3782–3786.
S. Tornaletti, D. Rozek, G. P. Pfeifer, The distribution of UV photoproducts along the human p53 gene and its relation to mutations in skin cancer, Oncogene, 1993, 8, 2051–2057.
G. P. Holmquist, S. Gao, Somatic mutation theory, DNA repair rates, and the molecular epidemiology of p53 mutations, Mutat. Res., 1997, 386, 69–101.
H. Ikehata, T. Ono, The mechanisms of UV mutagenesis, J. Radiat. Res., 2011, 52, 115–125.
D. E. Brash, J. A. Rudolph, J. A. Simon, A. Lin, G. J. McKenna, H. P. Baden, A. J. Halperin, J. Ponten, A role for sunlight in skin cancer: UV-induced p53 mutations in squamous cell carcinoma, Proc. Natl. Acad. Sci. U. S. A., 1991, 88, 10124–10128.
E. A. Drobetsky, A. J. Grosovsky, B. W. Glickman, The specificity of UV-induced mutations at an endogenous locus in mammalian cells, Proc. Natl. Acad. Sci. U. S. A., 1987, 84, 9103–9107.
A. S. Jonason, S. Kunala, G. J. Price, R. J. Restifo, H. M. Spinelli, J. A. Persing, D. J. Leffell, R. E. Tarone, D. E. Brash, Frequent clones of p53-mutated keratinocytes in normal human skin, Proc. Natl. Acad. Sci. U. S. A., 1996, 93, 14025–14029.
Q. Song, V. J. Cannistraro, J. S. Taylor, Rotational position of a 5-methylcytosine-containing cyclobutane pyrimidine dimer in a nucleosome greatly affects its deamination rate, J. Biol. Chem., 2011, 286, 6329–6335.
D. H. Lee, G. P. Pfeifer, Deamination of 5-methylcytosines within cyclobutane pyrimidine dimers is an important component of UVB mutagenesis, J. Biol. Chem., 2003, 278, 10314–10321.
N. Jiang, J. S. Taylor, In vivo evidence that UV-induced C->T mutations at dipyrimidine sites could result from the replicative bypass of cis-syn cyclobutane dimers or their deamination products, Biochemistry, 1993, 32, 472–481.
V. J. Cannistraro, J. S. Taylor, Acceleration of 5-methylcytosine deamination in cyclobutane dimers by G and its implications for UV-induced C-to-T mutation hotspots, J. Mol. Biol., 2009, 392, 1145–1157.
T. Douki, J. Cadet, Individual determination of the yield of the main UV-induced dimeric pyrimidine photoproducts in DNA suggests a high mutagenicity of CC photolesions, Biochemistry, 2001, 40, 2495–2501.
V. T. Tormanen, G. P. Pfeifer, Mapping of UV photoproducts within ras proto-oncogenes in UV-irradiated cells: correlation with mutations in human skin cancer, Oncogene, 1992, 7, 1729–1736.
P. J. Rochette, S. Lacoste, J. P. Therrien, N. Bastien, D. E. Brash, R. Drouin, Influence of cytosine methylation on ultraviolet-induced cyclobutane pyrimidine dimer formation in genomic DNA, Mutat. Res., 2009, 665, 7–13.
S. Tommasi, M. F. Denissenko, G. P. Pfeifer, Sunlight induces pyrimidine dimers preferentially at 5-methylcytosine bases, Cancer Res., 1997, 57, 4727–4730.
Y. H. You, C. Li, G. P. Pfeifer, Involvement of 5-methylcytosine in sunlight-induced mutagenesis, J. Mol. Biol., 1999, 293, 493–503.
S. Tornaletti, G. P. Pfeifer, UV damage and repair mechanisms in mammalian cells, Bioessays, 1996, 18, 221–228.
E. A. Drobetsky, E. Sage, UV-induced G:C->A:T transitions at the APRT locus of Chinese hamster ovary cells cluster at frequently damaged 5’-TCC-3’ sequences, Mutat. Res., 1993, 289, 131–138.
M. H. Patrick, Studies on thymine-derived UV photoproducts in DNA - I. Formation and biological role of pyrimidine adducts in DNA, Photochem. Photobiol., 1977, 25, 357–372.
I. D. Podmore, M. S. Cooke, K. E. Herbert, J. Lunec, Quantitative determination of cyclobutane thymine dimers in DNA by stable isotope-dilution mass spectrometry, Photochem. Photobiol., 1996, 64, 310–315.
G. P. Pfeifer, R. Drouin, A. D. Riggs, G. P. Holmquist, Binding of transcription factors creates hot spots for UV photoproducts in vivo, Mol. Cell. Biol., 1992, 12, 1798–1804.
G. P. Pfeifer, A. D. Riggs, Genomic footprinting by ligation mediated polymerase chain reaction, Methods Mol. Biol., 1993, 15, 153–168.
G. P. Pfeifer, A. D. Riggs, Genomic sequencing, Methods Mol. Biol., 1993, 23, 169–181.
S. Gao, R. Drouin, G. P. Holmquist, DNA repair rates mapped along the human PGK1 gene at nucleotide resolution, Science, 1994, 263, 1438–1440.
S. Tornaletti, G. P. Pfeifer, Slow repair of pyrimidine dimers at p53 mutation hotspots in skin cancer, Science, 1994, 263, 1436–1438.
H. Rodriguez, R. Drouin, G. P. Holmquist, T. R. O’Connor, S. Boiteux, J. Laval, J. H. Doroshow, S. A. Akman, Mapping of copper/hydrogen peroxide-induced DNA damage at nucleotide resolution in human genomic DNA by ligation-mediated polymerase chain reaction, J. Biol. Chem., 1995, 270, 17633–17640.
D. W. Brown, L. J. Libertini, C. Suquet, E. W. Small, M. J. Smerdon, Unfolding of nucleosome cores dramatically changes the distribution of ultraviolet photoproducts in DNA, Biochemistry, 1993, 32, 10527–10531.
J. M. Gale, K. A. Nissen, M. J. Smerdon, UV-induced formation of pyrimidine dimers in nucleosome core DNA is strongly modulated with a period of 10.3 bases, Proc. Natl. Acad. Sci. U. S. A., 1987, 84, 6644–6648.
J. R. Pehrson, Thymine dimer formation as a probe of the path of DNA in and between nucleosomes in intact chromatin, Proc. Natl. Acad. Sci. U. S. A., 1989, 86, 9149–9153.
J. R. Pehrson, Probing the conformation of nucleosome linker DNA in situ with pyrimidine dimer formation, J. Biol. Chem., 1995, 270, 22440–22444.
S. Tornaletti, G. P. Pfeifer, UV light as a footprinting agent: modulation of UV-induced DNA damage by transcription factors bound at the promoters of three human genes, J. Mol. Biol., 1995, 249, 714–728.
G. P. Pfeifer, R. Drouin, A. D. Riggs, G. P. Holmquist, In vivo mapping of a DNA adduct at nucleotide resolution: detection of pyrimidine (6-4) pyrimidone photoproducts by ligation-mediated polymerase chain reaction, Proc. Natl. Acad. Sci. U. S. A., 1991, 88, 1374–1378.
R. Drouin, N. Bastien, J. F. Millau, F. Vigneault, I. Paradis, In cellulo DNA analysis (LMPCR footprinting), Methods Mol. Biol., 2009, 543, 293–336.
R. Drouin, J. P. Therrien, M. Angers, S. Ouellet, In vivo DNA analysis, Methods Mol. Biol., 2001, 148, 175–219.
H. Rodriguez, S. A. Akman, Mapping oxidative DNA damage at nucleotide level, Free Radic. Res., 1998, 29, 499–510.
D. Rozek, G. P. Pfeifer, In vivo protein-DNA interactions at the c-jun promoter: preformed complexes mediate the UV response, Mol. Cell. Biol., 1993, 13, 5490–5499.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bastien, N., Therrien, JP. & Drouin, R. Cytosine containing dipyrimidine sites can be hotspots of cyclobutane pyrimidine dimer formation after UVB exposure. Photochem Photobiol Sci 12, 1544–1554 (2013). https://doi.org/10.1039/c3pp50099c
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c3pp50099c