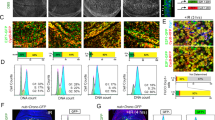
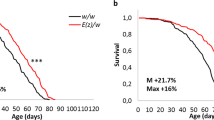
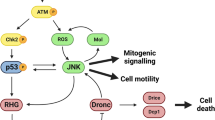
Abstract
p53 is a fundamental determinant of cancer susceptibility and other age-related pathologies. Similar to mammalian counterparts, Drosophila p53 integrates stress signals and elicits apoptotic responses that maintain genomic stability. To illuminate core-adaptive functions controlled by this gene family, we examined the Drosophila p53 regulatory network at a genomic scale. In development, the absence of p53 impacted constitutive expression for a surprisingly broad scope of genes. By contrast, stimulus-dependent responses governed by Drosophila p53 were limited in scope. The vast majority of stress responders were induced and p53 dependent (RIPD) genes. The signature set of 29 ‘high stringency’ RIPD genes identified here were enriched for intronless loci, with a non-uniform distribution that includes a recently evolved cluster unique to Drosophila melanogaster. Two RIPD genes, with known and unknown biochemical activities, were functionally examined. One RIPD gene, designated XRP1, maintains genome stability after genotoxic challenge and prevents cell proliferation upon induced expression. A second gene, RnrL, is an apoptogenic effector required for caspase activation in a model of p53-dependent killing. Together, these studies identify ancient and convergent features of the p53 regulatory network.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Abbreviations
- IR:
-
ionizing radiation
- RnrL:
-
ribonucleotide reductase large subunit
- RnrS:
-
ribonucleotide reductase small subunit
- RIPD:
-
radiation-induced p53-dependent
References
Bae BI, Xu H, Igarashi S, Fujimuro M, Agrawal N, Taya Y et al. (2005). p53 mediates cellular dysfunction and behavioral abnormalities in Huntington's disease. Neuron 47: 29–41.
Bauer JH, Poon PC, Glatt-Deeley H, Abrams JM, Helfand SL . (2005). Neuronal expression of p53 dominant-negative proteins in adult Drosophila melanogaster extends life span. Curr Biol 15: 2063–2068.
Blanchette M, Labourier E, Green RE, Brenner SE, Rio DC . (2004). Genome-wide analysis reveals an unexpected function for the Drosophila splicing factor U2AF50 in the nuclear export of intronless mRNAs. Mol Cell 14: 775–786.
Brodsky MH, Nordstrom W, Tsang G, Kwan E, Rubin GM, Abrams JM . (2000). Drosophila p53 binds a damage response element at the reaper locus. Cell 101: 103–113.
Brodsky MH, Weinert BT, Tsang G, Rong YS, McGinnis NM, Golic KG et al. (2004). Drosophila melanogaster MNK/Chk2 and p53 regulate multiple DNA repair and apoptotic pathways following DNA damage. Mol Cell Biol 24: 1219–1231.
Chai J, Du C, Wu JW, Kyin S, Wang X, Shi Y . (2000). Structural and biochemical basis of apoptotic activation by Smac/DIABLO. Nature 406: 855–862.
Chen P, Ho SI, Shi Z, Abrams JM . (2004). Bifunctional killing activity encoded by conserved reaper proteins. Cell Death Differ 11: 704–713.
Chew SK, Akdemir F, Chen P, Lu WJ, Mills K, Daish T et al. (2004). The apical caspase dronc governs programmed and unprogrammed cell death in Drosophila. Dev Cell 7: 897–907.
Colombani J, Polesello C, Josue F, Tapon N . (2006). Dmp53 activates the Hippo pathway to promote cell death in response to DNA damage. Curr Biol 16: 1453–1458.
Courgeon AM . (1972). Effects of - and -ecdysone on in vitro diploid cell multiplication in Drosophila melanogaster. Nat New Biol 238: 250–251.
Daish TJ, Mills K, Kumar S . (2004). Drosophila caspase DRONC is required for specific developmental cell death pathways and stress-induced apoptosis. Dev Cell 7: 909–915.
Dorstyn L, Colussi PA, Quinn LM, Richardson H, Kumar S . (1999). DRONC, an ecdysone-inducible Drosophila caspase. Proc Natl Acad Sci USA 96: 4307–4312.
Echalier G, Ohanessian A . (1970). In vitro culture of Drosophila melanogaster embryonic cells. In Vitro 6: 162–172.
Jin S, Kalkum M, Overholtzer M, Stoffel A, Chait BT, Levine AJ . (2003). CIAP1 and the serine protease HTRA2 are involved in a novel p53-dependent apoptosis pathway in mammals. Genes Dev 17: 359–367.
Kauppila S, Maaty WS, Chen P, Tomar RS, Eby MT, Chapo J et al. (2003). Eiger and its receptor, Wengen, comprise a TNF-like system in Drosophila. Oncogene 22: 4860–4867.
Lee CY, Clough EA, Yellon P, Teslovich TM, Stephan DA, Baehrecke EH . (2003a). Genome-wide analyses of steroid- and radiation-triggered programmed cell death in Drosophila. Curr Biol 13: 350–357.
Lee JH, Lee E, Park J, Kim E, Kim J, Chung J . (2003b). In vivo p53 function is indispensable for DNA damage-induced apoptotic signaling in Drosophila. FEBS Lett 550: 5–10.
Lu WJ, Abrams JM . (2006). Lessons from p53 in non-mammalian models. Cell Death Differ 13: 909–912.
Martins LM, Iaccarino I, Tenev T, Gschmeissner S, Totty NF, Lemoine NR et al. (2002). The serine protease Omi/HtrA2 regulates apoptosis by binding XIAP through a reaper-like motif. J Biol Chem 277: 439–444.
Nakano K, Balint E, Ashcroft M, Vousden KH . (2000). A ribonucleotide reductase gene is a transcriptional target of p53 and p73. Oncogene 19: 4283–4289.
Nordstrom W, Abrams JM . (2000). Guardian ancestry: fly p53 and damage-inducible apoptosis. Cell Death Differ 7: 1035–1038.
Nordstrom W, Chen P, Steller H, Abrams JM . (1996). Activation of the reaper gene during ectopic cell killing in Drosophila. Dev Biol 180: 213–226.
Peters M, DeLuca C, Hirao A, Stambolic V, Potter J, Zhou L et al. (2002). Chk2 regulates irradiation-induced, p53-mediated apoptosis in Drosophila. Proc Natl Acad Sci USA 99: 11305–11310.
Sakharkar MK, Kangueane P . (2004). Genome SEGE: a database for ‘intronless’ genes in eukaryotic genomes. BMC Bioinformatics 5: 67.
Sharpless NE, DePinho RA . (2002). p53: good cop/bad cop. Cell 110: 9–12.
Sogame N, Kim M, Abrams JM . (2003). Drosophila p53 preserves genomic stability by regulating cell death. Proc Natl Acad Sci USA 100: 4696–4701.
Tanaka H, Arakawa H, Yamaguchi T, Shiraishi K, Fukuda S, Matsui K et al. (2000). A ribonucleotide reductase gene involved in a p53-dependent cell-cycle checkpoint for DNA damage. Nature 404: 42–49.
Verhagen AM, Silke J, Ekert PG, Pakusch M, Kaufmann H, Connolly LM et al. (2002). HtrA2 promotes cell death through its serine protease activity and its ability to antagonise inhibitor of apoptosis proteins. J Biol Chem 277: 445–454.
Vousden KH, Prives C . (2005). P53 and prognosis: new insights and further complexity. Cell 120: 7–10.
Wu G, Chai J, Suber TL, Wu JW, Du C, Wang X et al. (2000). Structural basis of IAP recognition by Smac/DIABLO. Nature 408: 1008–1012.
Acknowledgements
We thank Nichole Link, Muskaan Behl and Brad Riek for technical assistance and Margaret Hickson for administrative excellence. This work was supported by the NIH (RO1GMO72124) and a Research Scholar award to JMA from the American Cancer Society. Raw data and related files are available at GEO using accession numbers GSE2780 and GSE3072.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Akdemir, F., Christich, A., Sogame, N. et al. p53 directs focused genomic responses in Drosophila. Oncogene 26, 5184–5193 (2007). https://doi.org/10.1038/sj.onc.1210328
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210328
Keywords
This article is cited by
-
Towards reconstructing the dipteran demise of an ancient essential gene: E3 ubiquitin ligase Murine double minute
Development Genes and Evolution (2020)
-
Xrp1 is a transcription factor required for cell competition-driven elimination of loser cells
Scientific Reports (2018)
-
Chronic activation of JNK JAK/STAT and oxidative stress signalling causes the loser cell status
Nature Communications (2017)
-
A platform for interrogating cancer-associated p53 alleles
Oncogene (2017)
-
Dissecting cellular senescence and SASP in Drosophila
Inflammation and Regeneration (2016)