Abstract
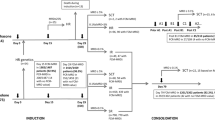
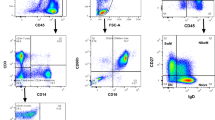
Sensitive and quantitative detection of minimal residual disease (MRD) in bone marrow (BM) samples of children with acute lymphoblastic leukemia (ALL) is essential for evaluation of early treatment response. In this study, we evaluated whether the traumatic BM samplings can be replaced by peripheral blood (PB) samplings. MRD levels were analyzed in follow-up samples of 62 children with precursor-B-ALL (532 paired BM-PB samples) and 22 children with T-ALL (149 paired BM-PB samples) using real-time quantitative PCR (RQ-PCR) analysis of immunoglobulin and T cell receptor gene rearrangements with sensitivities of 10−3 to 10−5 (one ALL cell in 103 to 105 normal cells). In 14 of the 22 T-ALL patients, detectable MRD levels were found in 67 paired BM-PB samples: in 47 pairs MRD was detected both in BM and PB, whereas in the remaining pairs very low MRD levels were detected in BM (n = 11) or PB (n = 9) only. The MRD levels in the paired BM-PB samples were very comparable and strongly correlated (rs = 0.849). Comparable results were obtained earlier by immunophenotyping in 26 T-ALL patients (321 paired BM-PB samples), which also showed a strong correlation between MRD levels in paired BM and PB samples (rs = 0.822). In 39 of the 62 precursor-B-ALL patients, MRD was detected in 107 BM-PB pairs: in 48 pairs MRD was detected in both BM and PB, in 47 pairs MRD was solely detected in BM (at variable levels), and in 12 pairs only the PB sample was MRD-positive at very low levels (≤10−4). Furthermore, in the 48 double-positive pairs, MRD levels in BM and PB varied enormously with MRD levels in BM being up to 1000 times higher than in the corresponding PB samples. Consequently, BM samples cannot easily be replaced by PB sampling for MRD analysis in childhood precursor-B-ALL, in line with their BM origin. In T-ALL, which are of thymic origin, BM sampling might be replaced by PB sampling, because the dissemination of T-ALL cells to BM and PB appears to be comparable.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Cave H, van der Werff ten Bosch J, Suciu S, Guidal C, Waterkeyn C, Otten J, Bakkus M, Thielemans K, Grandchamp B, Vilmer E . Clinical significance of minimal residual disease in childhood acute lymphoblastic leukemia. European Organization for Research and Treatment of Cancer - Childhood Leukemia Cooperative Group N Engl J Med 1998 339: 591–598
Coustan-Smith E, Sancho J, Hancock ML, Boyett JM, Behm FG, Raimondi SC, Sandlund JT, Rivera GK, Rubnitz JE, Ribeiro RC, Pui CH, Campana D . Clinical importance of minimal residual disease in childhood acute lymphoblastic leukemia Blood 2000 96: 2691–2696
van Dongen JJ, Seriu T, Panzer-Grmayer ER, Biondi A, Pongers-Willemse MJ, Corral L, Stolz F, Schrappe M, Masera G, Kamps WA, Gadner H, van Wering ER, Ludwig WD, Basso G, de Bruijn MA, Cazzaniga G, Hettinger K, van der Does-van den Berg A, Hop WC, Riehm H, Bartram CR . Prognostic value of minimal residual disease in acute lymphoblastic leukaemia in childhood Lancet 1998 352: 1731–1738
Panzer-Grmayer ER, Schneider M, Panzer S, Fasching K, Gadner H . Rapid molecular response during early induction chemotherapy predicts a good outcome in childhood acute lymphoblastic leukemia Blood 2000 95: 790–794
Knechtli CJ, Goulden NJ, Hancock JP, Grandage VL, Harris EL, Garland RJ, Jones CG, Rowbottom AW, Hunt LP, Green AF, Clarke E, Lankester AW, Cornish JM, Pamphilon DH, Steward CG, Oakhill A . Minimal residual disease status before allogeneic bone marrow transplantation is an important determinant of successful outcome for children and adolescents with acute lymphoblastic leukemia Blood 1998 92: 4072–4079
van der Velden VH, Joosten SA, Willemse MJ, van Wering ER, Lankester AW, van Dongen JJ, Hoogerbrugge PM . Real-time quantitative PCR for detection of minimal residual disease before allogeneic stem cell transplantation predicts outcome in children with acute lymphoblastic leukemia Leukemia 2001 15: 1485–1487
Szczepanski T, Orfao A, van der Velden VHJ, San Miguel JF, van Dongen JJM . Minimal residual disease in leukaemia patients Lancet Oncol 2001 2: 409–417
Van Dongen-Melman JE . Information booklet for parents of children surviving cancer Leukemia 1997 11: 1799–1806
van Dongen JJM, Hooijkaas H, Adriaansen HJ, HΩhlen K, van Zanen GE . Detection of minimal residual acute lymphoblastic leukemia by immunological marker analysis: possibilities and limitations. In: Hagenbeek A, Lowenberg B (eds) Minimal Residual Disease In Acute Leukemia Martinus Nijhoff: Dordrecht 1986 113–133
van Dongen JJ, Breit TM, Adriaansen HJ, Beishuizen A, Hooijkaas H . Detection of minimal residual disease in acute leukemia by immunological marker analysis and polymerase chain reaction Leukemia 1992 6: 47–59
Campana D, Coustan-Smith E . Detection of minimal residual disease in acute leukemia by flow cytometry Cytometry 1999 38: 139–152
Kluin-Nelemans HC, Bolhuis RL, Löwenberg B, Campana D, Sizoo W . Characterization of normal and regenerating bone marrow cells with a panel of monoclonal antibodies Scand J Haematol 1986 36: 71–78
van Lochem EG, Wiegers YM, van den Beemd R, Hählen K, van Dongen JJ, Hooijkaas H . Regeneration pattern of precursor-B-cells in bone marrow of acute lymphoblastic leukemia patients depends on the type of preceding chemotherapy Leukemia 2000 14: 688–695
Smedmyr B, Bengtsson M, Jakobsson A, Simonsson B, Oberg G, Totterman TH . Regeneration of CALLA (CD10+), TdT+ and double-positive cells in the bone marrow and blood after autologous bone marrow transplantation Eur J Haematol 1991 46: 146–151
van Wering ER, van der Linden-Schrever BE, Szczepanski T, Willemse MJ, Baars EA, van Wijngaarde-Schmitz HM, Kamps WA, van Dongen JJ . Regenerating normal B-cell precursors during and after treatment of acute lymphoblastic leukaemia: implications for monitoring of minimal residual disease Br J Haematol 2000 110: 139–146
Coustan-Smith E, Sancho J, Behm FG, Hancock ML, Razzouk B, Ribeiro RC, Rubnitz JE, Sanslund JT, Pui CH, Campana D . Comparative analysis of minimal residual disease in peripheral blood versus bone marrow in childhood acute lymphoblastic leukemia Blood 2001 98: 3497 (Abstr.)
van der Velden VHJ, Szczepanski T, van Dongen JJM . Polymerase chain reaction, real-time quantitative. In: S Brenner, JH Miller (eds) Encyclopedia of Genetics Academic Press: London 2001 1503–1506
Pongers-Willemse MJ, Verhagen OJ, Tibbe GJ, Wijkhuijs AJ, de Haas V, Roovers E, van der Schoot CE, van Dongen JJ . Real-time quantitative PCR for the detection of minimal residual disease in acute lymphoblastic leukemia using junctional region specific TaqMan probes Leukemia 1998 12: 2006–2014
Verhagen O, Willemse M, Breunis W, Wijkhuis A, Jacobs D, Joosten S, van Wering E, van Dongen J, van der Schoot C . Application of germline IGH probes in real-time quantitative PCR for the detection of minimal residual disease in acute lymphoblastic leukemia Leukemia 2000 14: 1426–1435
Brüggemann M, Droese J, Bolz I, Luth P, Pott C, von Neuhoff N, Scheuering U, Kneba M . Improved assessment of minimal residual disease in B cell malignancies using fluorogenic consensus probes for real-time quantitative PCR Leukemia 2000 14: 1419–1425
van der Velden VHJ, Willemse MJ, van der Schoot CE, Hählen K, van Wering ER, van Dongen JJM . Immunoglobulin kappa deleting element rearrangements in precursor-B-acute lymphoblastic leukemia are stable targets for detection of minimal residual disease by real-time quantitative PCR Leukemia 2002 16: 928–936
Brisco MJ, Sykes PJ, Hughes E, Story CJ, Rice MS, Schwarer AP, Morley AA . Molecular relapse can be detected in blood in a sensitive and timely fashion in B-lineage acute lymphoblastic leukemia Leukemia 2001 15: 1801–1802
Verhagen OJ, Wijkhuijs AJ, van der Sluijs-Gelling AJ, Szczepanski T, van der Linden-Schrever BE, Pongers-Willemse MJ, van Wering ER, van Dongen JJ, van der Schoot CE . Suitable DNA isolation method for the detection of minimal residual disease by PCR techniques Leukemia 1999 13: 1298–1299
Kamps WA, Veerman AJ, van Wering ER, van Weerden JF, Släter R, van der Does-van den Berg A . Long-term follow-up of Dutch Childhood Leukemia Study Group (DCLSG) protocols for children with acute lymphoblastic leukemia, 1984-1991 Leukemia 2000 14: 2240–2246
van Dongen JJ, Hooijkaas H, Comans-Bitter M, Hahlen K, de Klein A, van Zanen GE, van't Veer MB, Abels J, Benner R . Human bone marrow cells positive for terminal deoxynucleotidyl transferase (TdT), HLA-DR, and a T cell marker may represent prothymocytes J Immunol 1985 135: 3144–3150
Pongers-Willemse MJ, Seriu T, Stolz F, d'Aniello E, Gameiro P, Pisa P, Gonzalez M, Bartram CR, Panzer-Grmayer ER, Biondi A, San Miguel JF, van Dongen JJ . Primers and protocols for standardized detection of minimal residual disease in acute lymphoblastic leukemia using immunoglobulin and T cell receptor gene rearrangements and TAL1 deletions as PCR targets: report of the BIOMED-1 CONCERTED ACTION: investigation of minimal residual disease in acute leukemia Leukemia 1999 13: 110–118
Langerak AW, Szczepanski T, van der Burg M, Wolvers-Tettero IL, van Dongen JJ . Heteroduplex PCR analysis of rearranged T cell receptor genes for clonality assessment in suspect T cell proliferations Leukemia 1997 11: 2192–2199
Brisco MJ, Sykes PJ, Hughes E, Dolman G, Neoh SH, Peng LM, Toogood I, Morley AA . Monitoring minimal residual disease in peripheral blood in B-lineage acute lymphoblastic leukaemia Br J Haematol 1997 99: 314–319
Acknowledgements
This study was supported by the Dutch Cancer Foundation/Koningin Wilhelmina Fonds (grant SNWLK 97-1567 and grant SNWLK 2000-2268), the Ank Van Vlissingen Foundation, and the Rotary Zwolle. We acknowledge Daniëlle Korpershoek, Marcia Reintjes and Annella Boon for their secretarial support, Dr Ton Langerak for critically reading the manuscript, and Dr Wim CJ Hop (Department of Epidemiology and Biostatistics) for help with the statistical analyses. We thank Phary Hart for technical assistance, the pediatric oncologists of the Department of Pediatrics, Sophia Children‘s Hospital Rotterdam, for the collection of bone marrow and blood samples at diagnosis and during follow-up, and the Dutch Childhood Leukemia Study Group for kindly providing additional ALL cell samples.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
van der Velden, V., Jacobs, D., Wijkhuijs, A. et al. Minimal residual disease levels in bone marrow and peripheral blood are comparable in children with T cell acute lymphoblastic leukemia (ALL), but not in precursor-B-ALL. Leukemia 16, 1432–1436 (2002). https://doi.org/10.1038/sj.leu.2402636
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2402636
Keywords
This article is cited by
-
MRD in ALL: Optimization and Innovations
Current Hematologic Malignancy Reports (2022)
-
Comparison of minimal residual disease levels in bone marrow and peripheral blood in adult acute lymphoblastic leukemia
Leukemia (2020)
-
Flow Cytometric Minimal Residual Disease Analysis in Acute Leukemia: Current Status
Indian Journal of Hematology and Blood Transfusion (2020)
-
Residual methylation of tumor suppressor gene promoters, RASSF6 and RASSF10, as novel biomarkers for minimal residual disease detection in adult acute lymphoblastic leukemia
Annals of Hematology (2019)
-
Standardisation and consensus guidelines for minimal residual disease assessment in Philadelphia-positive acute lymphoblastic leukemia (Ph + ALL) by real-time quantitative reverse transcriptase PCR of e1a2 BCR-ABL1
Leukemia (2019)