Abstract
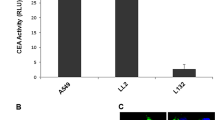
Gene therapy is a novel therapy for melanoma. To date, however, there is still no powerful tumor specific promoter (TSP) to restrict the transgene expression in melanoma cells. In order to define a useful TSP for targeting in the context of melanoma gene therapy, four promoters, the cyclooxygenase-2 (Cox-2), α-chemokine SDF-1 receptor (CXCR4), epithelial glycoprotein 2 (EGP-2), and survivin, were tested in both established melanoma cell lines and primary melanoma cells. We employed recombinant adenoviral vectors (reAds) each with a candidate TSP (the Cox-2, CXCR4, EGP-2, or survivin), a reporter luciferase gene, and a poly-A signal, all of which were inserted into the E1-deleted region. A reAdGL3Bcytomegalovirus (CMV), containing the CMV promoter and luciferase gene, was used as a positive control to normalize the luciferase activity. Luciferase activity was measured in multiple tumor cell lines and two primary melanoma cell cultures after infection with reAds. Human epithelial melanocytes, HEM, were used as normal control. In contrast to three other promoters, the survivin promoter exhibited the highest activities within both melanoma cell lines and primary melanoma cells, but not in HEMs. Additionally, the survivin promoter exhibited very low activities in major mouse organs including the liver, in vivo. EGP-2 is not active in melanoma; messenger RNA expressions were correlated to promoter activities both in melanoma cell lines and primary cell cultures. Thus, these data suggest that the survivin promoter achieved a ‘tumor-on/liver-off’ profile, and thus represents a potentially useful tumor-specific promoter with applications for transcriptional targeting of Ad vector-based cancer gene therapy or oncolysis to melanoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA . Cancer statistics, 1999. CA Cancer J Clin 1999; 49: 8–31.
Koh HK . Cutaneous melanoma. N Engl J Med 1991; 325: 171–182.
Eigentler TK, Caroli UM, Radny P, Garbe C . Palliative therapy of disseminated malignant melanoma: a systematic review of 41 randomised clinical trials. Lancet Oncol 2003; 4: 748–759.
Gomez-Navarro J, Curiel DT, Douglas JT . Gene therapy for cancer. Eur J Cancer 1999; 35: 2039–2057.
Kirn D, Martuza RL, Zwiebel J . Replication-selective virotherapy for cancer: biological principles, risk management and future directions. Nat Med 2001; 7: 781–787.
Brand K et al. Liver-associated toxicity of the HSV-tk/GCV approach and adenoviral vectors. Cancer Gene Ther 1997; 4: 9–16.
van der Eb MM et al. Severe hepatic dysfunction after adenovirus-mediated transfer of the herpes simplex virus thymidine kinase gene and ganciclovir administration. Gene Therapy 1998; 5: 451–458.
Haisma HJ et al. Targeting of adenoviral vectors through a bispecific single-chain antibody. Cancer Gene Ther 2000; 7: 901–904.
Heideman DA et al. Selective gene delivery toward gastric and esophageal adenocarcinoma cells via EpCAM-targeted adenoviral vectors. Cancer Gene Ther 2001; 8: 342–351.
Krasnykh V et al. Genetic targeting of an adenovirus vector via replacement of the fiber protein with the phage T4 fibritin. J Virol 2001; 75: 4176–4183.
Yamamoto M et al. Characterization of the cyclooxygenase-2 promoter in an adenoviral vector and its application for the mitigation of toxicity in suicide gene therapy of gastrointestinal cancers. Mol Ther 2001; 3: 385–394.
Adachi Y et al. A midkine promoter-based conditionally replicative adenovirus for treatment of pediatric solid tumors and bone marrow tumor purging. Cancer Res 2001; 61: 7882–7888.
Nettelbeck DM, Jerome V, Muller R . Gene therapy: designer promoters for tumour targeting. Trends Genet 2000; 16: 174–181.
Nettelbeck DM et al. Cox2 promoter for tumor-specific targeting of adenoviral vector to melanoma. Melanoma Res 2003; 13: 287–292.
Muller A et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001; 410: 50–56.
Altieri DC . Validating survivin as a cancer therapeutic target. Nat Rev Cancer 2003; 3: 46–54.
Adida C et al. Developmentally regulated expression of the novel cancer anti-apoptosis gene survivin in human and mouse differentiation. Am J Pathol 1998; 152: 43–49.
Chakravarti A et al. Quantitatively determined survivin expression levels are of prognostic value in human gliomas. J Clin Oncol 2002; 20: 1063–1068.
Tanaka K et al. Expression of survivin and its relationship to loss of apoptosis in breast carcinomas. Clin Cancer Res 2000; 6: 127–134.
Sarela AI et al. Expression of survivin, a novel inhibitor of apoptosis and cell cycle regulatory protein, in pancreatic adenocarcinoma. Br J Cancer 2002; 86: 886–892.
Bao R et al. Activation of cancer-specific gene expression by the survivin promoter. J Natl Cancer Inst 2002; 94: 522–528.
Andersen MH, thor SP . Survivin – a universal tumor antigen. Histol Histopathol 2002; 17: 669–675.
Balzar M, Winter MJ, de Boer CJ, Litvinov SV . The biology of the 17-1A antigen (Ep-CAM). J Mol Med 1999; 77: 699–712.
Linnenbach AJ et al. Sequence investigation of the major gastrointestinal tumor-associated antigen gene family, GA733. Proc Natl Acad Sci USA 1989; 86: 27–31.
Linnenbach AJ et al. Retroposition in a family of carcinoma-associated antigen genes. Mol Cell Biol 1993; 13: 1507–1515.
Litvinov SV et al. Ep-CAM: a human epithelial antigen is a homophilic cell–cell adhesion molecule. J Cell Biol 1994; 125: 437–446.
Momburg F, Moldenhauer G, Hammerling GJ, Moller P . Immunohistochemical study of the expression of a Mr 34 000 human epithelium-specific surface glycoprotein in normal and malignant tissues. Cancer Res 1987; 47: 2883–2891.
Alemany R, Balague C, Curiel DT . Replicative adenoviruses for cancer therapy. Nat Biotechnol 2000; 18: 723–727.
He TC et al. A simplified system for generating recombinant adenoviruses. Proc Natl Acad Sci USA 1998; 95: 2509–2514.
Zeng B Zhu et al. Transcriptional targeting of adenoviral vector through the CXCR4 tumor specific promoter. Gene Therapy 2004; 11: 645–648.
Zeng B Zhu et al. Transcriptional targeting of tumors with a novel tumor-specific survivin promoter. Cancer Gene Ther 2004; 11: 256–266.
McLaughlin PM et al. The epithelial glycoprotein 2 (EGP-2) promoter-driven epithelial-specific expression of EGP-2 in transgenic mice: a new model to study carcinoma-directed immunotherapy. Cancer Res 2001; 61: 4105–4111.
Marr RA et al. Tumour immunotherapy using an adenoviral vector expressing a membrane-bound mutant of murine TNF alpha. Gene Therapy 1997; 4: 1181–1188.
van der Eb MM et al. Severe hepatic dysfunction after adenovirus-mediated transfer of the herpes simplex virus thymidine kinase gene and ganciclovir administration. Gene Therapy 1998; 5: 451–458.
Southgate TD et al. Transcriptional targeting to anterior pituitary lactotrophic cells using recombinant adenovirus vectors in vitro and in vivo in normal and estrogen/sulpiride-induced hyperplastic anterior pituitaries. Endocrinology 2000; 141: 3493–3505.
Vile RG, Hart IR . Use of tissue-specific expression of the herpes simplex virus thymidine kinase gene to inhibit growth of established murine melanomas following direct intratumoral injection of DNA. Cancer Res 1993; 53: 3860–3864.
Lee SE et al. Development of a new plasmid vector with PSA-promoter and enhancer expressing tissue-specificity in prostate carcinoma cell lines. Anticancer Res 2000; 20: 417–422.
Tanaka S et al. Targeted killing of carcinoembryonic antigen (CEA)-producing cholangiocarcinoma cells by polyamidoamine dendrimer-mediated transfer of an Epstein–Barr virus (EBV)-based plasmid vector carrying the CEA promoter. Cancer Gene Ther 2000; 7: 1241–1250.
Dubois RN et al. Cyclooxygenase in biology and disease. FASEB J 1998; 12: 1063–1073.
Tippetts MT, Varnum BC, Lim RW, Herschman HR . Tumor promoter-inducible genes are differentially expressed in the developing mouse. Mol Cell Biol 1988; 8: 4570–4572.
Williams C, Shattuck-Brandt RL, DuBois RN . The role of COX-2 in intestinal cancer. Ann NY Acad Sci 1999; 889: 72–83.
Tsujii M, DuBois RN . Alterations in cellular adhesion and apoptosis in epithelial cells overexpressing prostaglandin endoperoxide synthase 2. Cell 1995; 83: 493–501.
Singh J, Hamid R, Reddy BS . Dietary fat and colon cancer: modulation of cyclooxygenase-2 by types and amount of dietary fat during the postinitiation stage of colon carcinogenesis. Cancer Res 1997; 57: 3465–3470.
Ristimaki A et al. Expression of cyclooxygenase-2 in human gastric carcinoma. Cancer Res 1997; 57: 1276–1280.
Denkert C et al. Expression of cyclooxygenase 2 in human malignant melanoma. Cancer Res 2001; 61: 303–308.
Wegner SA et al. Genomic organization and functional characterization of the chemokine receptor CXCR4, a major entry co-receptor for human immunodeficiency virus type 1. J Biol Chem 1998; 273: 4754–4760.
Robledo MM et al. Expression of functional chemokine receptors CXCR3 and CXCR4 on human melanoma cells. J Biol Chem 2001; 276: 45098–45105.
Payne AS, Cornelius LA . The role of chemokines in melanoma tumor growth and metastasis. J Invest Dermatol 2002; 118: 915–922.
Moore MA . The role of chemoattraction in cancer metastases. Bioessays 2001; 23: 674–676.
Taichman RS et al. Use of the stromal cell-derived factor-1/CXCR4 pathway in prostate cancer metastasis to bone. Cancer Res 2002; 62: 1832–1837.
Nasu S et al. Survivin mRNA expression in patients with breast cancer. Anticancer Res 2002; 22: 1839–1843.
Sarela AI et al. Expression of survivin, a novel inhibitor of apoptosis and cell cycle regulatory protein, in pancreatic adenocarcinoma. Br J Cancer 2002; 86: 886–892.
Ikeguchi M, Kaibara N . Survivin messenger RNA expression is a good prognostic biomarker for oesophageal carcinoma. Br J Cancer 2002; 87: 883–887.
Das A, Tan WL, Teo J, Smith DR . Expression of survivin in primary glioblastomas. J Cancer Res Clin Oncol 2002; 128: 302–306.
Zaffaroni N et al. Expression of the anti-apoptotic gene survivin correlates with taxol resistance in human ovarian cancer. Cell Mol Life Sci 2002; 59: 1406–1412.
Chiodino C et al. Communication: expression of the novel inhibitor of apoptosis survivin in normal and neoplastic skin. J Invest Dermatol 1999; 113: 415–418.
Gradilone A et al. Survivin, bcl-2, bax, and bcl-X gene expression in sentinel lymph nodes from melanoma patients. J Clin Oncol 2003; 21: 306–312.
Chakravarti A et al. Quantitatively determined survivin expression levels are of prognostic value in human gliomas. J Clin Oncol 2002; 20: 1063–1068.
Li F . Survivin Study: What is the next wave. J Cell Physiol 2003; 197: 8–29.
Asanuma K et al. A role for survivin in radioresistance of pancreatic cancer cells. Jpn J Cancer Res 2002; 93: 1057–1062.
Altieri DC . Validating survivin as a cancer therapeutic target. Nat Rev Cancer 2003; 3: 46–54.
Ambrosini G, Adida C, Sirugo G, Altieri DC . Induction of apoptosis and inhibition of cell proliferation by survivin gene targeting. J Biol Chem 1998; 273: 11177–11182.
Tamm I et al. IAP-family protein survivin inhibits caspase activity and apoptosis induced by Fas (CD95), Bax, caspases, and anticancer drugs. Cancer Res 1998; 58: 5315–5320.
Yamamoto T, Manome Y, Nakamura M, Tanigawa N . Downregulation of survivin expression by induction of the effector cell protease receptor-1 reduces tumor growth potential and results in an increased sensitivity to anticancer agents in human colon cancer. Eur J Cancer 2002; 38: 2316–2324.
Spizzo G et al. Correlation of COX-2 and Ep-CAM overexpression in human invasive breast cancer and its impact on survival. Br J Cancer 2003; 88: 574–578.
Gastl G et al. Ep-CAM overexpression in breast cancer as a predictor of survival. Lancet 2000; 356: 1981–1982.
Wimberger P et al. Efficient tumor cell lysis by autologous, tumor-resident T lymphocytes in primary ovarian cancer samples by an EP-CAM-/CD3-bispecific antibody. Int J Cancer 2003; 105: 241–248.
Trojan A et al. Functional detection of epithelial cell adhesion molecule specific cytotoxic T lymphocytes in patients with lung cancer, colorectal cancer and in healthy donors. Lung Cancer 2002; 36: 151–158.
Horn S et al. Expression of cell adhesion molecules in lung cancer cell lines. Exp Toxicol Pathol 1996; 48: 535–540.
Acknowledgements
This study was supported by research grants from the National Institutes of Health: RO1CA83821, RO1HL 67962, K12 HD01261-02 (WRHR) 5P50 CA83591 (Ovarian SPORE Developmental Grant). We thank Dr D Dieckmann for primary melanoma cells. DMN was supported by the Deutsche Forschungsgemeinschaft.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lu, B., Makhija, S., Nettelbeck, D. et al. Evaluation of tumor-specific promoter activities in melanoma. Gene Ther 12, 330–338 (2005). https://doi.org/10.1038/sj.gt.3302385
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302385
This article is cited by
-
Development of a carrier system containing hyaluronic acid and protamine for siRNA delivery in the treatment of melanoma
Investigational New Drugs (2021)
-
A novel approach for assessment of prostate cancer aggressiveness using survivin-driven tumour-activatable minicircles
Gene Therapy (2019)
-
Expression of HIF-1α ODD domain fused canine caspase 3 by EGFR promoter-driven adenovirus vector induces cytotoxicity in canine breast tumor cells under hypoxia
Veterinary Research Communications (2016)
-
Survivin: A Dual Player in Healthy and Diseased Skin
Journal of Investigative Dermatology (2012)
-
Potent, tumor-specific gene expression in an orthotopic hepatoma rat model using a Survivin-targeted, amplifiable adenoviral vector
Gene Therapy (2011)