Abstract
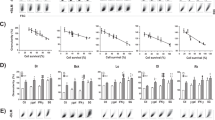
A recombinant MVMp of the fibrotropic strain of minute virus of mice (MVMp) expressing the chloramphenicol acetyltransferase reporter gene was used to infect a series of biologically relevant cultured cells, normal or tumor-derived, including normal melanocytes versus melanoma cells, normal mammary epithelial cells versus breast adenocarcinoma cells, and normal neurons or astrocytes versus glioma cells. As a reference cell system we used normal human fibroblasts versus the SV40-transformed fibroblast cell line NB324K. After infection, we observed good expression of the reporter gene in the different tumor cell types, but only poor expression if any in the corresponding normal cells. We also constructed a recombinant MVMp expressing the green fluorescent protein reporter gene and assessed by flow cytometry the efficiency of gene transduction into the different target cells. At a multiplicity of infection of 30, we observed substantial transduction of the gene into most of the tumor cell types tested, but only marginal transduction into normal cells under the same experimental conditions. Finally, we demonstrated that a recombinant MVMp expressing the herpes simplex virus thymidine kinase gene can, in vitro, cause efficient killing of most tumor cell types in the presence of ganciclovir, whilst affecting normal proliferating cells only marginally if at all. However, in the same experimental condition, breast tumor cells appeared to be resistant to GCV-mediated cytotoxicity, possibly because these cells are not susceptible to the bystander effect. Our data suggest that MVMp-based vectors could prove useful as selective vehicles for anticancer gene therapy, particularly for in vivo delivery of cytotoxic effector genes into tumor cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Marcel T, Grausz JD . The TMC Worldwide Gene Therapy Enrollment Report (June 1996) Hum Gene Ther 1996 7: 2025–2046
Yee D et al. Adenovirus-mediated gene transfer of herpes simplex virus thymidine kinase in an ascites model of human breast cancer Hum Gene Ther 1996 7: 1251–1257
Brand K et al. Liver-associated toxicity of the HSV-tk/GCV approach and adenoviral vectors Cancer Gene Ther 1997 4: 9–16
Miller N, Vile R . Targeted vectors for gene therapy FASEB J 1995 9: 190–199
Kasahara N, Dozy AM, Kan YW . Tissue-specific targeting of retroviral vectors through ligand–receptor interactions Science 1994 266: 1373–1376
Douglas JT et al. Targeted gene delivery by tropism-modified adenoviral vectors Nat Biotechnol 1996 14: 1574–1578
Culver KW et al. In vivo gene transfer with retroviral vector-producer cells for treatment of experimental brain tumors Science 1992 256: 1550–1552
Miller N, Whelan J . Progress in transcriptionally targeted and regulatable vectors for genetic therapy Hum Gene Ther 1997 8: 803–815
Miyatake S, Iyer A, Martuza RL, Rabkin SD . Transcriptional targeting of herpes simplex virus for cell-specific replication J Virol 1997 71: 5124–5132
Russell SJ . Lymphokine gene therapy for cancer Immunol Today 1990 11: 196–200
Cotmore SF, Tattersall P . The autonomously replicating parvoviruses of vertebrates Adv Virus Res 1987 33: 91–174
Berns KI . Parvovirus replication Microbiol Rev 1990 54: 316–329
Rommelaere J, Tattersall P . Oncosuppression by parvoviruses. In: Tijsen P (ed) Handbook of Parvoviruses CRC Press: Boca Raton 1990 41–57
Rommelaere J, Cornelis JJ . Antineoplastic activity of parvoviruses J Virol Meth 1991 33: 233–251
Perros M et al. Upstream CREs participate in the basal activity of minute virus of mice promoter P4 and in its stimulation in ras-transformed cells J Virol 1995 69: 5506–5515
Fuks F et al. ras oncogene-dependent activation of the P4 promoter of minute virus of mice through a proximal P4 element interacting with the Ets family of transcription factors J Virol 1996 70: 1331–1339
Cotmore SF, Tattersall P . DNA replication in the autonomous parvoviruses Semin Virol 1995 6: 271–281
Doerig C, Hirt B, Beard P, Antonietti JP . Minute virus of mice non-structural protein NS-1 is necessary and sufficient for trans-activation of the viral P39 promoter J Gen Virol 1988 69: 2563–2573
Christensen J, Cotmore SF, Tattersall P . Minute virus of mice transcriptional activator protein NS1 binds directly to the transactivation region of the viral P38 promoter in a strictly ATP-dependent manner J Virol 1995 69: 5422–5430
Brandenburger A, Legendre D, Avalosse B, Rommelaere J . NS-1 and NS-2 proteins may act synergistically in the cytopathogenicity of parvovirus MVMp Virology 1990 174: 576–584
Caillet-Fauquet P et al. Programmed killing of human cells by means of an inducible clone of parvoviral genes encoding non-structural proteins EMBO J 1990 9: 2989–2995
Russell SJ et al. Transformation-dependent expression of interleukin genes delivered by a recombinant parvovirus J Virol 1992 66: 2821–2828
Dupont F et al. Use of an autonomous parvovirus vector for selective transfer of a foreign gene into transformed human cells of different tissue origins and its expression therein J Virol 1994 68: 1397–1406
Naeger LK, Cater J, Pintel DJ . The small nonstructural protein (NS2) of the parvovirus minute virus of mice is required for efficient DNA replication and infectious virus production in a cell-type-specific manner J Virol 1990 64: 6166–6175
Spegelaere P et al. Initiation of transcription from the minute virus of mice P4 promoter is stimulated in rat cells expressing a c-Ha-ras oncogene J Virol 1991 65: 4919–4928
Rhode SL . Replication process of the parvovirus H-1. I. Kinetics in a parasynchronous cell system J Virol 1973 11: 856–861
Siegl G, Gautschi M . The multiplication of parvovirus Lu3 in a synchronized culture system. II. Biochemical characteristics of virus replication Arch Gesamte Virusforsch 1973 40: 119–127
Tattersall P, Bratton J . Reciprocal productive and restrictive virus–cell interactions of immunosuppressive and prototype strains of minute virus of mice J Virol 1983 46: 944–955
Deleu L et al. Opposite transcriptional effects of cyclic AMP-responsive elements in confluent or p27KIP-overexpressing cells versus serum-starved or growing cells Mol Cell Biol 1998 18: 409–419
Maron A et al. Gene therapy of rat C6 glioma using adenovirus-mediated transfer of the herpes simplex virus thymidine kinase gene: long-term follow-up by magnetic resonance imaging Gene Therapy 1996 3: 315–322
Maron A et al. Differential toxicity of ganciclovir for rat neurons and astrocytes in primary culture following adenovirus-mediated transfer of the HSVtk gene Gene Therapy 1997 4: 25–31
Siegl G . Biology and pathogenicity of autonomous parvoviruses. In: Berns KI (ed) The Parvoviruses Plenum Press: New York 1994 297–348
Studdert MJ . Tissue tropism of parvoviruses. In: Tijsen P (ed) Handbook of Parvoviruses CRC Press: Boca Raton 1990 3–27
Koering CE et al. Induced expression of the conditionally cytotoxic herpes simplex virus thymidine kinase gene by means of a parvoviral regulatory circuit Hum Gene Ther 1994 5: 457–463
Lee SW et al. Transcriptional downregulation of gap-junction proteins blocks junctional communication in human mammary tumor cell lines J Cell Biol 1992 118: 1213–1221
Nicolson GL . Breast cancer metastasis-associated genes: role in tumour progression to the metastatic state Biochem Soc Symp 1998 63: 231–243
Kumar NM, Gilula NB . The gap junction communication channel Cell 1996 84: 381–388
Bi WL, Parysek LM, Warnick R, Stambrook PJ . In vitro evidence that metabolic cooperation is responsible for the bystander effect observed with HSV tk retroviral gene therapy Hum Gene Ther 1993 4: 725–731
Vrionis FD et al. The bystander effect exerted by tumor cells expressing the herpes simplex virus thymidine kinase (HSVtk) gene is dependent on connexin expression and cell communication via gap junctions Gene Therapy 1997 4: 577–585
Dilber MS et al. Gap junctions promote the bystander effect of herpes simplex virus thymidine kinase in vivo Cancer Res 1997 57: 1523–1528
Rommelaere J, Ward DC . Effect of UV-irradiation on DNA replication of the parvovirus minute-virus-of-mice in mouse fibroblasts Nucleic Acids Res 1982 10: 2577–2596
Shein HM, Enders JF . Multiplication and cytopathogenicity of simian vacuolating virus 40 in cultures of human tissue Proc Soc Exp Biol Med 1962 40: 495–500
Heimann P et al. Chromosomal findings in cultured melanocytes from a giant congenital nevus Cancer Genet Cytogenet 1993 68: 74–77
Merchlinsky MJ et al. Construction of an infectious molecular clone of the autonomous parvovirus minute virus of mice J Virol 1983 47: 227–232
Colbere-Garapin F et al. Cloning of the active thymidine kinase gene of herpes simplex virus type 1 in Escherichia coli K-12 Proc Natl Acad Sci USA 1979 76: 3755–3759
Avalosse B et al. Method for concentrating and purifying recombinant autonomous parvovirus vectors designed for tumour-cell-targeted gene therapy J Virol Meth 1996 62: 179–183
Acknowledgements
We thank J-N Octave for providing normal rat neurones and astrocytes, and P Spegelaere for the gift of pSP116. We are grateful to T Dupressoir, C Bagnis, P Martiat, L Tenenbaum and JC Dumon for helpful discussions and critical reading of the manuscript. We also thank JC Dumon for the statistical analysis. This work was financially supported by the ‘Fonds National de la Recherche Scientifique’ (FNRS), the Région Bruxelles-Capitale, Z-Company, the Fondation Medic, and by grants from ‘Télévie’ and ‘les Amis de l'Institut Bordet’ (to AK).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dupont, F., Avalosse, B., Karim, A. et al. Tumor-selective gene transduction and cell killing with an oncotropic autonomous parvovirus-based vector. Gene Ther 7, 790–796 (2000). https://doi.org/10.1038/sj.gt.3301161
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301161
Keywords
This article is cited by
-
Novel adenovirus-based helper system to support production of recombinant parvovirus
Cancer Gene Therapy (2011)
-
TNF-α and the IFN-γ-inducible protein 10 (IP-10/CXCL-10) delivered by parvoviral vectors act in synergy to induce antitumor effects in mouse glioblastoma
Cancer Gene Therapy (2009)
-
Augmented transgene expression in transformed cells using a parvoviral hybrid vector
Cancer Gene Therapy (2008)
-
Oncolytic murine autonomous parvovirus, a candidate vector for glioma gene therapy, is innocuous to normal and immunocompetent mouse glial cells
Cell and Tissue Research (2006)
-
Suppression of metastatic hemangiosarcoma by a parvovirus MVMp vector transducing the IP-10 chemokine into immunocompetent mice
Cancer Gene Therapy (2002)