Abstract
Purpose
To quantitatively examine changes in macular oedema after phacoemulsification surgery in eyes with diabetic retinopathy (DR) and in eyes without DR.
Methods
Thirty-four eyes with DR and 34 eyes with no DR scheduled for phacoemulsification surgery were recruited. Foveal thickness and macular volume were measured using optical coherence tomography preoperatively and at 3, 6, and 12 months postoperatively. Grade of diabetic macular oedema was also examined.
Results
Preoperatively, there were no significant differences between groups in foveal thickness and macular volume. The foveal thickness increased by 20.3% in the DR group and by 6.0% in the no DR group at 3 months after surgery, but thereafter decreased gradually. When comparing the groups, the foveal thickness and macular volume were significantly greater in the DR group than in the no DR group at 3 months postoperatively. The grade of macular oedema worsened in eight eyes (23.5%) in the DR group, and in one (2.9%) in the no DR group: the incidence was significantly greater in the DR group ( P=0.0272). However, the oedema that occurred after surgery resolved spontaneously in three (33.3%) of the nine eyes.
Conclusions
The degree of diabetic macular oedema increases up to 3 months after cataract surgery, but thereafter decreases gradually. Grade of diabetic macular oedema also worsens up to 3 months, but certain percent of macular oedema that occurs after surgery resolves spontaneously. These changes are more prominent in eyes with DR than in eyes with no DR.
Similar content being viewed by others
Introduction
A number of studies have shown that visual outcome following cataract surgery in diabetic patients depends primarily on the status of macular oedema.1, 2, 3, 4, 5, 6, 7 Previous reports have described many diabetic patients who developed severe maculopathy, retinopathy, and/or neovascular glaucoma following cataract surgery.5, 8, 9, 10, 11, 12 Because it is important to be able to predict long-term visual effects before cataract surgery is performed, surgeons need to have a better understanding of the natural course of diabetic macular oedema in addition to diabetic retinopathy (DR) after cataract surgery.
It has been demonstrated that DR progresses in approximately 10–30% of patients after cataract surgery,2, 6, 13, 14, 15, 16, 17 although some authors have hypothesized that the progression of DR after cataract surgery is due simply to the natural course of the condition, and that the progression is independent of the surgery.18, 19, 20, 21 In these studies, the most significant predictor for progression of DR has been the status of the DR at the time of the cataract surgery.13, 14, 15, 16, 22, 23
Similarly, diabetic macular oedema has been shown to worsen after cataract surgery,24, 25, 26 although controversy remains as to the incidence of this worsening.7, 27 Some studies have shown that there are two types of worsening of macular oedema after surgery: transient pseudophakic oedema that spontaneously resolves such as Irvine–Gass syndrome28, 29, 30 and actual progression of diabetic maculopathy. It has also been suggested that macular oedema tends to show actual worsening in eyes afflicted with DR at the time of cataract surgery.14 Distinguishing transient oedema from substantial progression of maculopathy is important to the timing of treatment for the macular oedema, including laser photocoagulation,31 vitrectomy,32, 33, 34 and triamcinolone injection.35, 36, 37 However, until recently, there had been no quantitative study to examine the progression of diabetic macular oedema after cataract surgery. A recent study described by Kim et al38 showed a short-term increase of macular thickness after cataract surgery.
The purpose of the study described herein was to investigate changes in macular oedema after phacoemulsification surgery in eyes with DR and in eyes with no DR. To compare the degree of macular oedema quantitatively, we measured periodically the foveal thickness and macular volume using optical coherence tomography.
Materials and methods
Patients
All patients with diabetes who were scheduled for phacoemulsification surgery and intraocular lens (IOL) implantation between May 2004 and July 2005 were consecutively screened for inclusion in this study. In those patients in whom both eyes underwent surgery, only the eye operated on first was included. Exclusion criteria were cataract due to causes other than age or diabetes, planned extracapsular cataract extraction, a history of ocular inflammation or surgery, any pathology of the macula or optic nerve due to causes other than diabetes, any anticipated difficulties with the analysis, any expected problems with follow-up because the patient resided in distant place or had been referred from another clinic, and patient' refusal to participate. Screening was continued until 35 eyes with DR and 35 eyes with no DR were recruited for this study. At the time of cataract surgery, all patients underwent implantation of a hydrophobic acrylic IOL (MA60AC or MA50BM; Alcon Laboratories, Fort Worth, TX, USA). The study protocol was approved by the Hospital's Institutional Review Board, and informed consent was obtained from each patient.
Surgical procedures
All surgeries were performed by one surgeon (KH) using the same surgical procedures as described previously.39 Briefly, a 3.5-mm scleral incision was made; after incision, a continuous curvilinear capsulorhexis measuring approximately 5.0 mm in diameter was accomplished using a bent needle. The intent was to make a round capsulorhexis of proper size (slightly smaller than optic diameter) so that the capsulorhexis edge would circumferentially overlie the IOL optic. After hydrodissection, phacoemulsification of the nucleus and cortical aspiration were performed. Using a keratome, the wound was enlarged to 4.1 mm for IOL implantation. The lens capsule was then inflated with sodium hyaluronate 1% (Healon; Advanced Medical Optics, Santa Ana, CA, USA), after which the acrylic IOL was placed in the capsular bag. After insertion, the viscoelastic material was thoroughly evacuated. All surgeries included in the analysis were uneventful and the IOLs were implanted accurately within the capsular bag.
Main outcome measures
The central retinal (foveal) thickness and macular volume of all patients were measured by Stratus optical coherence tomography (OCT; Carl Zeiss Meditec, Dublin, CA, USA) a day before surgery, and at 3, 6, and 12 months after surgery. Best-corrected visual acuity and grade of diabetic macular oedema were determined at each examination. Best-corrected visual acuity on decimal charts was converted to the logarithm of minimal angle of resolution (logMAR) scale for statistical analysis. Systemic status of the diabetes mellitus, including percentage of haemoglobin A1C, duration of diabetes (years), and types of treatment at the time of cataract surgery were also examined.
During the OCT procedure, each eye underwent six radial scans centred on the fovea. Each radial scan line consisted of 100 samples of 5.92 mm in length, yielding a total of 600 samples from the six radial scans. Macular volume was calculated as follows. A central macular thickness map measuring 3.45 mm in diameter was generated. The circular map was subdivided into nine quadrants. The diameters of the middle and inner circles were 2.22 and 1.00 mm, respectively. Mean retinal thickness was calculated for each of the nine quadrants from the previously obtained radial scans. Multiplying the mean retinal thickness by the area of the quadrant generated the volume for each of the nine quadrants, and the total macular volume was thus determined as the sum of the volume of the nine quadrants.
A fundus examination with full pupil dilation was performed before surgery and at each scheduled examination, and eyes with DR underwent fluorescein angiography to determine the presence of proliferative changes before surgery and at 12 months after surgery. The macular oedema was assigned one of the four following grades: 0=no macular oedema, 1=focal macular oedema, 2=diffuse macular oedema, and 3=cystoid macular oedema. Details of the macular oedema grade have been described previously.26 In brief, focal macular oedema was defined as retinal thickening that occupied less than 1 disc area, while diffuse macular oedema was defined as retinal thickening of 1 disc area or greater. The grade of macular oedema was determined by a single physician (KH), who was also the surgeon; other examinations were performed by ophthalmic technicians.
Statistical analysis
To compare data between the DR and no DR groups, the Mann–Whitney U test was used for foveal thickness, macular volume, logMAR visual acuity, and other continuous variables. Discrete variables were compared using the Fisher exact probability test or the χ2 test for independence. Simple correlations between the foveal thickness and macular volume and logMAR visual acuity, and between these and haemoglobin A1C, the duration of diabetes, or the types of treatment were evaluated using the Pearson correlation coefficient. Any differences showing a P-value of <0.05 were considered statistically significant.
Results
Of the 35 eyes originally enrolled, one in each group was lost to follow-up. One patient in the DR group refused the examination, and one in the no DR group did not appear for examination because of a scheduling conflict. Therefore, 34 patients (97%) in each group completed the 1-year follow-up and were included for analysis.
Patient characteristics are shown in Table 1. The average age of the patients (±standard deviation [SD]) was 67.9±7.0 years, with a range of 51–81 years; there were 39 men and 29 women. No statistically significant differences were found between the DR and no DR groups in age, the ratio of left to right eyes, or the presence of hypertension and nephropathy; the duration of diabetes in the DR group, however, was significantly longer than that in the no DR group (P=0.0318), and the percentage of haemoglobin A1C in the DR group was significantly greater than that in the no DR group (P<0.0001). Operative factors are shown in Table 2. No significant differences were found between the two groups in grade of the nucleus, ultrasound time, ultrasound power emitted, or infusion volume. Twelve eyes in the DR group underwent photocoagulation for the peripheral retina, but no patients underwent retinal photocoagulation for macular oedema.
The mean (±SD) foveal thickness and macular volume in the DR and no DR groups are shown in Figure 1 and 2, respectively. The percentage increases in the foveal thickness and increases in macular volume from baseline are provided in Table 3. There were no significant differences between the two groups in foveal thickness or in macular volume before surgery. Foveal thickness increased by 20.3% in the DR group and by 6.0% in the no DR group at 3 months after surgery, and macular volume increased by 7.8% in the DR group and 3.0% in the no DR group, but thereafter these changes decreased. When comparing groups at 3 months after surgery, the foveal thickness, macular volume, and the percent increases in the DR group were significantly greater than those in the no DR group. The foveal thickness, macular volume, and percentage increases also tended to be greater in the DR group than in the no DR group at 6 and 12 months after surgery, but the difference did not reach statistical significance.
Comparison of the mean (±standard deviation [SD]) foveal thickness between the diabetic retinopathy (DR) group and no DR group before and after cataract surgery. Before surgery, there was no significant difference in foveal thickness between the two groups. At 3 months after surgery, the mean foveal thickness increased in both groups, but then decreased gradually. When comparing the two groups, postoperative foveal thickness in the DR group was greater than that in the no DR group, although the difference was statistically significant only at 3 months after surgery.
Comparison of the mean (±SD) macular volume between the DR and no DR groups before and after cataract surgery. Before surgery, no significant difference was found in macular volume between the two groups. At 3 months after surgery, the macular volume increased in both groups, but then decreased gradually. When comparing the two groups, postoperative macular volume in the DR group was greater than that in the no DR group, although a significant difference was found only at 3 months after surgery.
Figure 3 shows the comparison of mean (±SD) logMAR visual acuity between the two groups. Mean visual acuity in both groups improved significantly by 3 months after surgery (P<0.0001), and did not deteriorate postoperatively (P=0.9104 in the DR group and P=0.9687 in the no DR group). Only two eyes (5.9%) in the DR group lost more than two decimal lines of visual acuity because of progression of maculopathy. When comparing the two groups, mean visual acuity in the DR group was significantly worse than that in the no DR group at 3, 6, and 12 months after surgery, despite the fact that there had been no difference before surgery. In addition, logMAR visual acuity was significantly correlated with both foveal thickness and macular volume at 3, 6, and 12 months after surgery.
Comparison of the mean (±SD) visual acuity between the DR and no DR groups before and after cataract surgery. Mean visual acuity improved significantly in both groups by cataract surgery, but did not change postoperatively. When comparing the groups, mean visual acuity in the DR group was significantly worse than that in the no DR group postoperatively, although there was no significant difference preoperatively.
The grade of macular oedema is shown in Figure 4. The grade of macular oedema worsened in eight eyes (23.5%) in the DR group but in only one (2.9%) in the no DR group; the incidence of progression in the DR group was significantly greater than that in the no DR group (P=0.0272). However, in three of the nine eyes (33.3%) that had developed macular oedema postoperatively, the oedema resolved virtually by 12 months after surgery.
Grade of macular oedema before surgery, and at 3, 6, and 12 months after surgery in the DR and no DR groups. The grade of macular oedema worsened in eight eyes (23.5%) in the DR group but in only one (2.9%) in the no DR group; the incidence of progression in the DR group was significantly greater than that in the no DR group (P=0.0272). G0=no macular oedema, G1=focal macular oedema, G2=diffuse macular oedema, and G3=cystoid macular oedema.
Table 4 shows the simple correlation coefficients between haemoglobin A1C and the foveal thickness and macular volume. Significant association was found between the haemoglobin A1C at the time of surgery and foveal thickness throughout the follow-up, whereas the associations between the haemoglobin A1C and macular volume did not reach statistical significance. Furthermore, no significant correlation was found between duration of diabetes and foveal thickness or macular volume. In addition, there were no significant differences in the foveal thickness or macular volume between the types of treatment, the presence of hypertension, or the presence of nephropathy.
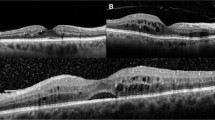
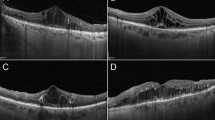
Figure 5 and 6 show the horizontal images and maps of the OCT as well as a fundus photograph of a representative eye that shows transient macular oedema or progression of maculopathy after surgery, respectively. In the eye shown in Figure 5, in which no apparent macular oedema was seen before surgery, marked cystoid macular oedema occurred at 3 months after surgery. However, this decreased gradually and only a slight oedema was observed at 12 months after surgery. In the eye shown in Figure 6, diffuse macular oedema with dot and blot haemorrhages and hard exudates was present before surgery. Although the macular thickening gradually decreased, there was accumulation of hard exudates in the macula, which resulted in severe macular deposition of the exudates at 12 months after surgery.
Horizontal images and maps of the optical coherence tomography (OCT) and fundus photograph of a representative eye that shows transient macular oedema. Marked cystoid macular oedema occurred at 3 months after surgery, but then decreased gradually until 12 months. Only a slight macular oedema was observed either before surgery or at 12 months after surgery.
Horizontal images and maps of the optical coherence tomography (OCT), and fundus photograph of a representative eye that shows actual progression of diabetic maculopathy. Diffuse macular oedema with dot haemorrhages and hard exudates was present before surgery. The macular oedema decreased gradually, but the hard exudates accumulated in the macula, and resulted in their severe deposition in the macula at 12 months after surgery.
Discussion
Our study have shown that the foveal thickness and macular volume in diabetic patients increases after small incision cataract surgery in eyes both with and without DR: the percent increase from baseline was greatest at 3 months after surgery, and then decreased gradually. Furthermore, the increase in foveal thickness and in macular volume was greater in eyes with DR than in eyes without DR. These results indicate that, on average, diabetic macular oedema worsens after cataract surgery, and the worsening is more pronounced in eyes with DR.
The grade of diabetic macular oedema also progressed in 24% of the eyes with DR, but in only 3% of the eyes with no DR. However, three cases of nine (33.3%) of macular oedema that occurred after surgery in eyes with DR had resolved spontaneously by 12 months. These results suggest that the change in macular oedema in some diabetic patients is transient, while in other diabetics, the macular oedema is substantial, with marked progression of maculopathy.
Mean visual acuity improved significantly after cataract surgery in eyes both with and without DR, and did not worsen in the postoperative period. Only two eyes ultimately lost more than 2 decimal lines of vision because of marked progression of maculopathy. This indicates that worsening of the macular oedema after small incision cataract surgery decreases visual acuity only in some cases.
Among the parameters studied that reflected the systemic status of diabetes, the haemoglobin A1C level at the time of surgery in eyes with DR was significantly greater than that in eyes without DR, and was correlated significantly with the foveal thickness. In contrast, duration of diabetes, types of treatment, and the presence of hypertension or nephropathy were not related to the degree of macular oedema. Although previous studies did not find a significant correlation between haemoglobin A1C and progression of macular oedema,26, 27 our quantitative study suggests that the percentage of haemoglobin A1C present at the time of surgery may be a significant predictor for progression of macular oedema.
Previous studies showed that diabetic macular oedema progressed in approximately 20–40% of eyes that underwent cataract surgery, but, in a considerable percentage of these eyes, the macular oedema resolved spontaneously.24, 25, 26, 27 Accordingly, these studies suggested that progression of diabetic macular oedema may be classified as follows: a transient pseudophakic macular oedema such as Irvine–Gass syndrome, or substantial progression of diabetic maculopathy.24, 25, 27 Our study quantitatively confirmed these two types of macular oedema. That is, macular thickening generally occurs after cataract surgery, but this resolves with time in most diabetic patients. In some diabetic patients, however, diabetic maculopathy actually progresses. Both types of macular change are more prominent in eyes with DR than in eyes with no DR. In addition, Funatsu et al26 reported the amount of vascular endothelial growth factor in the aqueous humour to be a significant predictor for progression of macular oedema. Our study showed that haemoglobin A1C is associated with an increased risk for progression of diabetic macular oedema.
Our study showed that macular thickening occurs in most diabetic eyes after cataract surgery, some of which develop clinically detectable oedema including cystoid macular oedema. Macular thickening after cataract surgery is greater, however, in eyes of diabetics, specifically in eyes with DR, than that in nondiabetic eyes (unpublished data). This may be explained biologically as follows. It is thought that pseudophakic cystoid macular oedema is caused by cytokines including prostaglandin or vascular endothelial growth factor, which are released from blood–ocular barrier after cataract extraction. Breakdown of the blood–ocular barrier in diabetic eyes, particularly in eyes with DR, is known to be greater than that in nondiabetic eyes.40, 41, 42 Indeed, the mean intensity of flare in the DR group was significantly greater than that in the no DR group throughout the follow-up (unpublished data). Accordingly, macular thickening may be more pronounced in eyes with DR.
We acknowledge some limitations to this study. First, the number of eyes with active DR was small. However, surgeons currently tend to perform retinal photocoagulation when retinopathy is in the early stages. Accordingly, distribution of the stage of DR in this study may actually reflect more closely the clinical situation. Secondly, the grade of macular oedema and the stage of DR evaluated in this study were relatively simple. However, the goal of the present study was to quantify the degree of macular oedema, so minute grading of macular oedema was not the primary end point.
In conclusion, we have demonstrated that the degree of diabetic macular oedema generally worsens after cataract surgery, and that the worsening of macular oedema is more prominent in eyes with DR than in eyes without DR at the time of surgery. However, macular oedema that occurs after cataract surgery resolves spontaneously in some patients for up to a year. Furthermore, percentage of haemoglobin A1C present at the time of surgery may be predictor for progression of macular oedema. Using a larger sample size, further study is warranted to examine the changes in macular oedema that occur in eyes with active retinopathy.
References
Dowler JGF, Hykin PG, Lightman SL, Hamilton AM . Visual acuity following extracapsular cataract extraction in diabetes: a meta-analysis. Eye 1995; 9: 313–317.
Zaczek A, Olivestedt G, Zetterstrom C . Visual outcome after phacoemulsification and IOL implantation in diabetic patients. Br J Ophthalmol 1999; 83: 1036–1041.
Cunliffe IA, Flanagan DW, George NDL, Aggarwaal RJ, Moore AT . Extracapsular cataract surgery with lens implantation in diabetics with and without proliferative retinopathy. Br J Ophthalmol 1991; 75: 9–12.
Hykin PG, Gregson RMC, Stevens JD, Hamilton PAM . Extracapsular cataract extraction in proliferative diabetic retinopathy. Ophthalmology 1993; 100: 394–399.
Benson WE, Brown GC, Tasman W, McNamara JA, Vander JF . Extracapsular cataract extraction with placement of a posterior chamber lens in patients with diabetic retinopathy. Ophthalmology 1993; 100: 730–738.
Antcliff RJ, Poulson A, Flanagan DW . Phacoemulsification in diabetics. Eye 1996; 10: 737–741.
Early Treatment Diabetic Retinopathy Study Research Group. Results after lens extraction in patients with diabetic retinopathy: Early Treatment Diabetic Retinopathy Study report number 25. Arch Ophthalmol 1999; 117: 1600–1606.
Poliner LS, Christianson DJ, Escoffery RF, Kolker AE, Gordon ME . Neovascular glaucoma after intracapsular and extracapsular cataract extraction in diabetic patients. Am J Ophthalmol 1985; 100: 637–643.
Pavese T, Insler MS . Effects of extracapsular cataract extraction with posterior chamber lens implantation on the development of neovascular glaucoma in diabetics. J Cataract Refract Surg 1987; 13: 197–201.
Jaffe GJ, Burton TC . Progression of nonproliferative diabetic retinopathy following cataract extraction. Arch Ophthalmol 1988; 106: 745–749.
Jaffe GJ, Burton TC, Kuhn E, Prescott A, Hartz A . Progression of nonproliferative diabetic retinopathy and visual outcome after extracapsular cataract extraction and intraocular lens implantation. Am J Ophthalmol 1992; 114: 448–456.
Schatz H, Atienza D, McDonald HR, Johnson RN . Severe diabetic retinopathy after cataract surgery. Am J Ophthalmol 1994; 117: 314–321.
Pollack A, Dotan S, Oliver M . Progression of diabetic retinopathy after cataract extraction. Br J Ophthalmol 1991; 75: 547–551.
Henricsson M, Heijl A, Janzon L . Diabetic retinopathy before and after cataract surgery. Br J Ophthalmol 1996; 80: 789–793.
Kato S, Fukada Y, Hori S, Tanaka Y, Oshika T . Influence of phacoemulsification and intraocular lens implantation on the course of diabetic retinopathy. J Cataract Refract Surg 1999; 25: 788–793.
Mittra RA, Borrillo JL, Dev S, Mieler WF, Koenig SB . Retinopathy progression and visual outcomes after phacoemulsification in patients with diabetes mellitus. Arch Ophthalmol 2000; 118: 912–917.
Chung J, Kim MY, Kim HS, Yoo JS, Lee YC . Effect of cataract surgery on the progression of diabetic retinopathy. J Cataract Refract Surg 2002; 25: 626–630.
Tsujikawa A, Otani A, Takanashi T, Ogura Y . Long-term prognosis of extracapsular cataract extraction and intraocular lens implantation in diabetic patients. Jpn J Ophthalmol 1997; 41: 319–323.
Krepler K, Biowski R, Schrey S, Jandrasits K, Wedrich A . Cataract surgery in patients with diabetic retinopathy: visual outcome, progression of diabetic retinopathy, and incidence of diabetic macular edema. Graefes Arch Clin Exp Ophthalmol 2002; 240: 735–738.
Schrey S, Krepler K, Biowski R, Wedrich A . Midterm visual outcome and progression of diabetic retinopathy following cataract surgery. Midterm outcome of cataract surgery in diabetes. Ophthalmologica 2002; 216: 337–340.
Romero-Aroca P, Fernandez-Ballart J, Almena-Garcia M, Mendez-Marin I, Salvat-Serra M, Buil-Calvo JA . Nonproliferative diabetic retinopathy and macular edema progression after phacoemulsification: prospective surgery. J Cataract Refract Surg 2006; 321: 1438–1444.
Pollack A, Leiba H, Bukelman A, Abrahami S, Oliver M . The course of diabetic retinopathy following cataract surgery in eyes previously treated by laser photocoaguration. Br J Ophthalmol 1992; 76: 228–231.
Somaiya MD, Burns JD, Mintz R, Warren RE, Uchida T, Godley BF . Factors affecting visual outcomes after small-incision phacoemulsification in diabetic patients. J Cataract Refract Surg 2002; 28: 1364–1371.
Dowler JGF, Sehmi KS, Hykin PG, Hamilton AMP . The natural history of macular edema cataract surgery in diabetes. Ophthalmology 1999; 106: 663–668.
Dowler JGF, Hykin PG, Hamilton AMP . Phacoemulsification versus extracapsular cataract extraction in patients with diabetes. Ophthalmology 2000; 107: 457–462.
Funatsu H, Yamashita H, Noma H, Shimizu E, Mimura T, Hori S . Prediction of macular edema exacerbation after phacoemulsification in patients with nonproliferative diabetic retinopathy. J Cataract Refract Surg 2002; 28: 1355–1363.
Squirrell D, Bhola R, Bush J, Winder S, Talbot JF . A prospective, case controlled study of the natural history of diabetic retinopathy and maculopathy after uncomplicated phacoemulsification cataract surgery in patients with type 2 diabetes. Br J Ophthalmol 2002; 86: 565–571.
Gass JDM, Norton EWD . Cystoid macular edema and papilledema following cataract extraction. A fluorescein fundoscopic and angiographic study. Arch Ophthalmol 1966; 76: 646–661.
Tolentino FI, Schepens CL . Edema of posterior pole after cataract extraction. A biomicroscopic study. Arch Ophthalmol 1965; 74: 781–786.
Schepens CL, Avila MP, Jalkh AE, Trempe CL . Role of the vitreous in cystoid macular edema. Surv Ophthalmol 1984; 28(Suppl): 499–504.
Early Treatment Diabetic Retinopathy Study Research Group. Photocoagulation for diabetic macular edema: Early Treatment Diabetic Retinopathy Study report number 1. Arch Ophthalmol 1985; 103: 1796–1806.
Lewis H, Abrams GW, Blumenkranz MS, Campo RV . Vitrectomy for diabetic macular traction and edema associated with posterior hyaloidal traction. Ophthalmology 1992; 99: 753–759.
Harbour JW, Smiddy WE, Flynn Jr HW, Rubsamen PE . Vitrectomy for diabetic macular edema associated with a thickened and taut posterior hyaloid membrane. Am J Ophthalmol 1996; 121: 405–413.
Tachi N, Ogino N . Vitrectomy for diffuse macular edema in cases of diabetic retinopathy. Am J Ophthalmol 1996; 122: 258–260.
Jonas JB, Sofker A . Intraocular injection of crystalline cortisone as adjunctive treatment of diabetic macular edema. Am J Ophthalmol 2001; 132: 425–427.
Martidis A, Duker JS, Greenberg PB, Rogers AH, Puliafito CA, Reichel E et al. Intravitreal triamcinolone for refractory diabetic macular edema. Ophthalmology 2002; 109: 920–927.
Jonas JB, Kreissig I, Sofker A, Degenring RF . Intravitreal injection of triamcinolone for diffuse diabetic macular edema. Arch Ophthalmol 2003; 121: 57–61.
Kim SJ, Equi R, Bressler NM . Analysis of macular edema after cataract surgery in patients with diabetes using optical coherence tomography. Ophthalmology 2007; 114: 881–889.
Hayashi K, Hayashi H, Nakao F, Hayashi F . Posterior capsule opacification after cataract surgery in patients with diabetes mellitus. Am J Ophthalmol 2002; 134: 10–16.
Zaczek A, Zetterstrom C . Aqueous flare intensity after phacoemulsification in patients with diabetes mellitus. J Cataract Refract Surg 1998; 24: 1099–1104.
Oshika T, Kato S, Funatsu H . Quantitative assessment of aqueous flare intensity in diabetes. Graefes Arch Clin Exp Ophthalmol 1989; 227: 518–520.
Moriarty AP, Spalton DJ, Moriarty BJ, Shilling JS, Ffytche TJ, Bulsara M . Studies of the blood-aqueous barrier in diabetes mellitus. Am J Ophthalmol 1994; 117: 768–771.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors have no proprietary interest in any of the materials described in this article and received no financial support.
Rights and permissions
About this article
Cite this article
Hayashi, K., Igarashi, C., Hirata, A. et al. Changes in diabetic macular oedema after phacoemulsification surgery. Eye 23, 389–396 (2009). https://doi.org/10.1038/sj.eye.6703022
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.eye.6703022
Keywords
This article is cited by
-
Cataract surgery with combined versus deferred intravitreal dexamethasone implant for diabetic macular edema: long-term outcomes from a real-world setting
Acta Diabetologica (2020)
-
Antagonising Wnt/β-catenin signalling ameliorates lens-capsulotomy-induced retinal degeneration in a mouse model of diabetes
Diabetologia (2018)
-
Benchmarking cataract surgery outcomes in an ethnically diverse and diabetic population: final post-operative visual acuity and rates of post-operative cystoid macular oedema
Eye (2017)