Summary
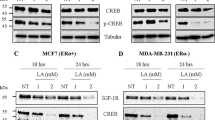
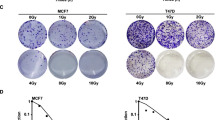
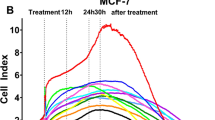
Recent data have demonstrated that the anti-oestrogen tamoxifen (TAM) is able to facilitate apoptosis in cancer cells not expressing oestrogen receptor (ER). In an attempt to identify the biochemical pathway for this phenomenon, we investigated the role of TAM as an oxidative stress agent. In two ER-negative human cancer cell lines, namely T-leukaemic Jurkat and ovarian A2780 cancer cells, we have demonstrated that TAM is able to generate oxidative stress, thereby causing thiol depletion and activation of the transcriptional factor NF-κB. As described for other oxidative agents, TAM was able to induce either cell proliferation or apoptosis depending on the dose. When used at the lowest dose tested (0.1 μM), a slight proliferative effect of TAM was noticed in terms of cell counts and DNA synthesis rate, whereas at higher doses (10 μM) a consistent occurrence of apoptosis was detected. Importantly, the induction of apoptosis by TAM is not linked to down-regulation or functional inactivation by phosphorylation of the antiapoptotic bcl-2 protein.
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bass, D. A., Parce, J. W., Dechatelet, L. R., Szejda, P., Seeds, M. C. & Thomas, M. (1983). Flow cytometric studies of oxidative product formation by neutrophils: a graded response to membrane stimulation. J Immunol 130: 1910–1917.
Berman, E., Adams, M., Duigou-Ostendorf, R., Godfrey, L., Clarkson, B. & Andreef, M. (1991). Effect of tamoxifen on cell lines displaying the multidrug-resistant phenotype. Blood 77: 818–825.
Couldwell, W. T., Hinton, D. R., He, S., Chen, T. C., Sebat, I., Weiss, M. H. & Law, R. E. (1994). Protein kinase C inhibitors induce apoptosis in human malignant glioma cell lines. FEBS Lett 345: 43–46.
De Vincenzo, R., Scambia, G., Benedetti Panici, P., Fattorossi, A., Bonanno, G., Ferlini, C., Isola, G., Pernisco, S. & Mancuso, S. (1996). Modulatory effect of tamoxifen and ICI 182,780 on adriamycin resistance in MCF-7 human breast cancer cells. Int J Cancer 68: 340–348.
Ferlini, C., Biselli, R., Scambia, G. & Fattorossi, A. (1996). Probing chromatin structure in the early phases of apoptosis. Cell Prolif 29: 427–436.
Ferlini, C., Scambia, G., Distefano, M., Bombardelli, E., Riva, A., Fattorossi, A., Filippini, P., Isola, G., Benedetti Panici, P. & Mancuso, S. (1997). Synergistic antiproliferative activity of tamoxifen and docetaxel on three oestrogen receptor negative cancer cell lines is mediated by the induction of apoptosis. Br J Cancer 75: 884–891.
Gosland, M., Lum, B., Schimmelpfennig, J., Baker, J. & Doukas, M. (1996). Insights into mechanisms of cisplatin resistance and potential for its clinical reversal. Pharmacotherapy 16: 16–39.
Gundimeda, U., Chen, Z. H. & Gopalakrishna, R. (1996). Tamoxifen modulates protein kinase C via oxidative stress in estrogen receptor-negative breast cancer cells. J Biol Chem 271: 13504–13514.
Haldar, S., Jena, N. & Croce, C. M. (1995). Inactivation of Bcl-2 by phosphorylation. Proc Natl Acad Sci USA 92: 4507–4511.
Haldar, S., Chintapalli, J. & Croce, C. M. (1996). Taxol induces bcl-2 phosphorylation and death of prostate cancer cells. Cancer Res 56: 1253–1255.
Hockenbery, D. M., Oltvai, Z. N., Xiao-Ming, Y., Milliman, C. L. & Korsmeyer, S. J. (1993). Bcl-2 functions in an antioxidant pathway to prevent apoptosis. Cell 75: 241–251.
Issandou, M., Faucher, C., Bayard, F. & Darbon, J. M. (1990). Opposite effects of tamoxifen on in vitro protein kinase C activity and endogenous protein phosphorylation in intact MCF-7 cells. Cancer Res 50: 5845–5850.
Jaiyesimi, I. A., Buzdar, A. U., Decker, D. A. & Hortobagyi, G. N. (1995). Use of tamoxifen for breast cancer: twenty-eight years later. J Clin Oncol 13: 513–529.
Janssen, Y. M., Barchowsky, A., Treadwell, M., Driscoll, K. E. & Mossman, B. T. (1995). Asbestos induces nuclear factor kappa B (NF-kappa B) DNA-binding activity and NF-kappa B-dependent gene expression in tracheal epithelial cells. Proc Natl Acad Sci 92: 8458–8462.
Kang, Y. & Perry, R. R. (1993). Modulatory effects of tamoxifen and recombinant human α-interferon on doxorubicin resistance. Cancer Res 53: 3040–3045.
Kang, Y., Cortina, R. & Perry, R. R. (1996). Role of c-myc in tamoxifen-induced apoptosis in estrogen-independent breast cancer cells. J Natl Cancer Inst 88: 279–284.
Kirk, K., Houlbrook, S., Stuart, N. S. A., Stratford, I. J., Harris, A. L. & Carmichael, J. (1993). Differential modulation of doxorubicin to multidrug and intrinsically drug-resistant cell lines by anti-oestrogens and their major metabolites. Br J Cancer 67: 1189–1195.
Leonessa, F., Jacobson, M., Boyle, B., Lippman, J., McGarvey, M. & Clarke, R. (1994). Effect of tamoxifen on the multidrug-resistant phenotype in human breast cancer cells: isobologram, drug accumulation and Mr 170 000 glycoprotein (gp 170) binding studies. Cancer Res 54: 441–447.
Lopes, M. C. F., Vale, M. G. P. & Caravalho, A. P. (1990). Ca-dependent binding of tamoxifen to calmodulin isolated from bovine brain. Cancer Res 50: 2753–2758.
Los, M., Droge, W., Stricker, K., Bauerle, P. A. & Schulze-Osthoff, K. (1995). Hydrogen peroxide as a potent activator of T lymphocyte functions. Eur J Immunol 25: 159–165.
McClay, E. F., Albright, K. D., Jones, J. A., Christen, R. D. & Howell, S. B. (1993). Tamoxifen modulation of cisplatin sensitivity in human malignant melanoma cells. Cancer Res 53: 1571–1576.
McClay, E. F., Jones, J. A., Winski, P. J., Albright, K. D., Christen, R. D. & Howell, S. B. (1996). Determinants of tamoxifen sensitivity control the nature of the synergistic interaction between tamoxifen and cisplatin. Cancer Res 56: 3993–3997.
Murrell, G. A., Francis, M. J. & Bromley, L. (1990). Modulation of fibroblast proliferation by oxygen free radicals. Biochem J 265: 659–665.
Perry, R. R., Kang, Y. & Greaves, B. (1995). Effects of tamoxifen on growth and apoptosis of oestrogen-dependent and -independent human breast cancer cells. Ann Surg Oncol 2: 238–245.
Poot, M., Kavanagh, T. J., Kang, H. C., Haugland, R. P. & Rabinovitch, P. S. (1991). Flow cytometric analysis of cell-cycle dependent changes in thiol level by combining a new laser dye with Hoechst 33342. Cytometry 12: 184–187.
Rao, G. N. & Berk, B. C. (1992). Active oxygen species stimulate vascular smooth muscle cell growth and proto-oncogene expression. Circ Res 70: 593–599.
Sen, C. K. & Packer, L. (1996). Antioxidant and redox regulation of gene transcription. FASEB J 10: 709–720.
Wiseman, H. (1993). Protective actions of tamoxifen and 4-hydroxytamoxifen against oxidative damage to human low-density lipoproteins: a mechanism accounting for the cardioprotective action of tamoxifen?. Biochem J 292: 635–638.
Wiseman, H., Laughton, M. J., Arnstein, H. R. V., Cannon, M. & Halliwell, B. (1990). The antioxidant action of tamoxifen. Inhibition of lipid peroxidation. FEBS Lett 263: 192–194.
Ye, Q. & Bodell, W. J. (1996). Production of 8-hydroxy-2′-deoxyguanosine in DNA by microsomal activation of tamoxifen and 4-hydroxytamoxifen. Carcinogenesis 17: 1747–1750.
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Ferlini, C., Scambia, G., Marone, M. et al. Tamoxifen induces oxidative stress and apoptosis in oestrogen receptor-negative human cancer cell lines. Br J Cancer 79, 257–263 (1999). https://doi.org/10.1038/sj.bjc.6690042
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bjc.6690042
Keywords
This article is cited by
-
Potentiating the anti-cancer profile of tamoxifen-loaded graphene using deep eutectic solvents as functionalizing agents
Applied Nanoscience (2020)
-
Simultaneous defeat of MCF7 and MDA-MB-231 resistances by a hypericin PDT–tamoxifen hybrid therapy
npj Breast Cancer (2019)
-
Potential anti-genotoxic effect of sodium butyrate to modulate induction of DNA damage by tamoxifen citrate in rat bone marrow cells
Cytotechnology (2017)
-
Oxidative stress contributes to the tamoxifen-induced killing of breast cancer cells: implications for tamoxifen therapy and resistance
Scientific Reports (2016)
-
Sensitization of estrogen receptor-positive breast cancer cell lines to 4-hydroxytamoxifen by isothiocyanates present in cruciferous plants
European Journal of Nutrition (2016)