Abstract
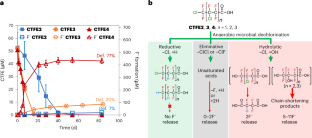
Chlorinated polyfluorocarboxylic acids (Cl-PFCAs) derived from the widely used chlorotrifluoroethylene oligomers and polymers may enter and influence the aquatic environment. Here we report the substantial defluorination of Cl-PFCAs by an anaerobic microbial community via novel pathways triggered by anaerobic microbial dechlorination. Cl-PFCAs first underwent microbial reductive, hydrolytic and eliminative dechlorination, with the hydrolytic dechlorination leading to the highest spontaneous defluorination. Hydrolytic dechlorination was favoured with increased Cl substitutions. An isolated, highly enriched, anaerobic defluorinating culture was dominated by two genomes that were most similar to those of Desulfovibrio aminophilus and Sporomusa sphaeroides, both of which exhibited defluorination activity towards chlorotrifluoroethylene tetramer acid. The results imply that anaerobic non-respiratory hydrolytic dechlorination plays a critical role in the fate of chlorinated polyfluoro chemicals in natural and engineered water environments. The greatly enhanced biodegradability by Cl substitution also sheds light on the design of cost-effective treatment biotechnologies, as well as alternative polyfluoroalkyl substances that are readily biodegradable and less toxic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The metagenomic sequencing dataset has been deposited in the Sequence Read Archive under accession number PRJNA838587. Source data are provided with this paper. The draft genomes of the two dominant bacterial species in the isolated defluorinating colonies have been deposited in GenBank under the accession numbers JAMHFZ000000000 and JAMHGA000000000. All other data supporting the findings in this study are available within the paper and its Supplementary Information. Source data are provided with this paper.
References
Dolbier, W. R. Fluorine chemistry at the millennium. J. Fluor. Chem. 126, 157–163 (2005).
Lohmann, R. et al. Are fluoropolymers really of low concern for human and environmental health and separate from other PFAS. Environ. Sci. Technol. 54, 12820–12828 (2020).
Evich, M. G. et al. Per- and polyfluoroalkyl substances in the environment. Science 375, eabg9065 (2022).
Xiao, F. Emerging poly- and perfluoroalkyl substances in the aquatic environment: a review of current literature. Water Res. 124, 482–495 (2017).
Kotthoff, M. & Bucking, M. Four chemical trends will shape the next decade’s directions in perfluoroalkyl and polyfluoroalkyl substances research. Front. Chem. 6, 103 (2018).
Dams, R. & Hintzer, K. in Fluorinated Polymers: Applications Vol. 2 1–31 (The Royal Society of Chemistry, 2017).
Vanbrocklin, C. Military applications of chlorotrifluoroethylene oligomer base nonflammable hydraulic fluid. J. Fire Sci. 11, 232–241 (1993).
Gschweder, L., Mattie, D., Syder, C., Warer, W. & van Brocklin, C. Chlorotrifluoroethylene oligomer based nonflammable hydraulic fluid. 1 Fluid, additive, and elastomer development. J. Synth. Lubr. 9, 187–203 (1992).
Ellis, D. A., Mabury, S. A., Martin, J. W. & Muir, D. C. Thermolysis of fluoropolymers as a potential source of halogenated organic acids in the environment. Nature 412, 321–324 (2001).
Myers, A. L., Jobst, K. J., Mabury, S. A. & Reiner, E. J. Using mass defect plots as a discovery tool to identify novel fluoropolymer thermal decomposition products. J. Mass Spectrom. 49, 291–296 (2014).
Dias, J. et al. Thermal degradation behavior of ionic liquid/fluorinated polymer composites: effect of polymer type and ionic liquid anion and cation. Polymer 229, 123995 (2021).
DelRaso, N. J., Auten, K. L., Higman, H. C. & Leahy, H. F. Evidence of hepatic conversion of C6 and C8 chlorotrifluoroethylene (CTFE) oligomers to their corresponding CTFE acids. Toxicol. Lett. 59, 41–49 (1991).
Brashear, W. T., Greene, R. J. & Mahle, D. A. Structural determination of the carboxylic acid metabolites of polychlorotrifluoroethylene. Xenobiotica 22, 499–506 (1992).
Song, X., Vestergren, R., Shi, Y., Huang, J. & Cai, Y. Emissions, transport, and fate of emerging per- and polyfluoroalkyl substances from one of the major fluoropolymer manufacturing facilities in China. Environ. Sci. Technol. 52, 9694–9703 (2018).
Fujiwara, M., Jodry, J. J. & Narizuka, S. Process for producing alkoxycarbonylfluoroalkanesulfonates. US patent US20080108846A1 (2008).
Yin, H., Anders, M. W. & Jones, J. P. Metabolism of 1,2-dichloro-1-fluoroethane and 1-fluoro-1,2,2-trichloroethane: electronic factors govern the regioselectivity of cytochrome P450-dependent oxidation. Chem. Res. Toxicol. 9, 50–57 (1996).
Harris, J. W. & Anders, M. W. Metabolism of the hydrochlorofluorocarbon 1,2-dichloro-1,1-difluoroethane. Chem. Res. Toxicol. 4, 180–186 (1991).
Liu, J. & Mejia Avendano, S. Microbial degradation of polyfluoroalkyl chemicals in the environment: a review. Environ. Int. 61, 98–114 (2013).
Yu, Y. et al. Microbial cleavage of C–F bonds in two C6 per- and polyfluorinated compounds via reductive defluorination. Environ. Sci. Technol. 54, 14393–14402 (2020).
Yu, Y. et al. Microbial defluorination of unsaturated per- and polyfluorinated carboxylic acids under anaerobic and aerobic conditions: a structure specificity study. Environ. Sci. Technol. 56, 4894–4904 (2022).
Wackett, L. P. Nothing lasts forever: understanding microbial biodegradation of polyfluorinated compounds and perfluorinated alkyl substances. Microb. Biotechnol. 15, 773–792 (2021).
Adrian, L. & Löffler, F. E. Organohalide-Respiring Bacteria (Springer, 2016).
Zhang, B. et al. Novel and legacy poly- and perfluoroalkyl substances (PFASs) in indoor dust from urban, industrial, and e-waste dismantling areas: the emergence of PFAS alternatives in China. Environ. Pollut. 263, 114461 (2020).
Liu, W. et al. Atmospheric chlorinated polyfluorinated ether sulfonate and ionic perfluoroalkyl acids in 2006 to 2014 in Dalian, China. Environ. Toxicol. Chem. 36, 2581–2586 (2017).
Wang, Y. et al. Suspect and nontarget screening of per- and polyfluoroalkyl substances in wastewater from a fluorochemical manufacturing park. Environ. Sci. Technol. 52, 11007–11016 (2018).
Liu, Y., Qian, M., Ma, X., Zhu, L. & Martin, J. W. Nontarget mass spectrometry reveals new perfluoroalkyl substances in fish from the Yangtze River and Tangxun Lake, China. Environ. Sci. Technol. 52, 5830–5840 (2018).
Washington, J. W. et al. Nontargeted mass-spectral detection of chloroperfluoropolyether carboxylates in New Jersey soils. Science 368, 1103–1107 (2020).
MacInnis, J. J., Lehnherr, I., Muir, D. C. G., Quinlan, R. & De Silva, A. O. Characterization of perfluoroalkyl substances in sediment cores from High and Low Arctic lakes in Canada. Sci. Total Environ. 666, 414–422 (2019).
Lin, Y., Ruan, T., Liu, A. & Jiang, G. Identification of novel hydrogen-substituted polyfluoroalkyl ether sulfonates in environmental matrices near metal-plating facilities. Environ. Sci. Technol. 51, 11588–11596 (2017).
Yi, S., Zhu, L. & Mabury, S. A. First report on in vivo pharmacokinetics and biotransformation of chlorinated polyfluoroalkyl ether sulfonates in rainbow trout. Environ. Sci. Technol. 54, 345–354 (2020).
Yi, S., Yang, D., Zhu, L. & Mabury, S. A. Significant reductive transformation of 6:2 chlorinated polyfluorooctane ether sulfonate to form hydrogen-substituted polyfluorooctane ether sulfonate and their toxicokinetics in male Sprague–Dawley rats. Environ. Sci. Technol. 56, 6123–6132 (2021).
Lieberman, I. & Barker, H. A. β-Keto acid formation and decomposition by preparations of Clostridium kluyveri. J. Bacteriol. 68, 329–333 (1954).
Buschmann, J., Angst, W. & Schwarzenbach, R. P. Iron porphyrin and cysteine mediated reduction of ten polyhalogenated methanes in homogeneous aqueous solution: product analyses and mechanistic considerations. Environ. Sci. Technol. 33, 1015–1020 (1999).
Che, S. et al. Structure-specific aerobic defluorination of short-chain fluorinated carboxylic acids by activated sludge communities. Environ. Sci. Technol. Lett. 8, 668–674 (2021).
Chen, G. et al. Anaerobic microbial metabolism of dichloroacetate. mBio https://doi.org/10.1128/mBio.00537-21 (2021).
Möller, B., Oßmer, R., Howard, B. H., Gottschalk, G. & Hippe, H. Sporomusa, a new genus of gram-negative anaerobic bacteria including Sporomusa sphaeroides spec. nov. and Sporomusa ovata spec. nov. Arch. Microbiol. 139, 388–396 (1984).
Temme, H. R., Carlson, A. & Novak, P. J. Presence, diversity, and enrichment of respiratory reductive dehalogenase and non-respiratory hydrolytic and oxidative dehalogenase genes in terrestrial environments. Front. Microbiol. 10, 1258 (2019).
Duhamel, M. & Edwards, E. A. Microbial composition of chlorinated ethene-degrading cultures dominated by Dehalococcoides. FEMS Microbiol. Ecol. 58, 538–549 (2006).
Baena, S. et al. Desulfovibrio aminophilus sp. nov., a novel amino acid degrading and sulfate reducing bacterium from an anaerobic dairy wastewater lagoon. Syst. Appl. Microbiol. 21, 498–504 (1998).
Yang, Y., Pesaro, M., Sigler, W. & Zeyer, J. Identification of microorganisms involved in reductive dehalogenation of chlorinated ethenes in an anaerobic microbial community. Water Res. 39, 3954–3966 (2005).
Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio fischeri (Luminescent Bacteria Test)—Part 3: Method Using Freeze-Dried Bacteria ISO 11348-3:2007 (ISO, 2007).
Newton, S. et al. Novel polyfluorinated compounds identified using high resolution mass spectrometry downstream of manufacturing facilities near Decatur, Alabama. Environ. Sci. Technol. 51, 1544–1552 (2017).
Evich, M. G. et al. Environmental fate of Cl-PFPECAs: predicting the formation of PFAS transformation products in New Jersey soils. Environ. Sci. Technol. 56, 7779–7788 (2022).
Men, Y. et al. Sustainable syntrophic growth of Dehalococcoides ethenogenes strain 195 with Desulfovibrio vulgaris Hildenborough and Methanobacterium congolense: global transcriptomic and proteomic analyses. ISME J. 6, 410–421 (2012).
Schymanski, E. L. et al. Identifying small molecules via high resolution mass spectrometry: communicating confidence. Environ. Sci. Technol. 48, 2097–2098 (2014).
Tonomura, K., Futai, F., Tanabe, O. & Yamakoka, T. Defluorination of monofluoroacetate by bacteria. Part I. Isolation of bacteria and their activity of defluorination. Agric. Biol. Chem. 29, 124–128 (1965).
Davis, C. K., Webb, R. I., Sly, L. I., Denman, S. E. & McSweeney, C. S. Isolation and survey of novel fluoroacetate-degrading bacteria belonging to the phylum Synergistetes. FEMS Microbiol. Ecol. 80, 671–684 (2012).
Acknowledgements
This work was supported by the Strategic Environmental Research and Development Program (project no. ER20-1541, for B.J., Y.Y., J.G., J.L. and Y.M.) and the National Institute of Environmental Health Sciences (award no. R01ES032668, for H.L., S.C. and Y.M.). M. Elsner at the Technical University of Munich provided insightful discussion on the biodefluorination pathways.
Author information
Authors and Affiliations
Contributions
Y.M. and B.J. conceived and designed the project, analysed the data and prepared the paper. B.J. conducted the anaerobic biotransformation experiments using the anaerobic microbial community and pure cultures and analysed the LC–HRMS/MS and sequencing data. S.C. performed the aerobic biotransformation experiments and contributed to the anaerobic biotransformation set-up. H.L. performed the anaerobic isolation and sample preparation for metagenomic sequencing. Y.Y. contributed to the analysis of the analytical and sequencing data. J.G. contributed to the calculation of the BDEs. J.L. contributed to the PFAS compound selection and mechanistic discussion.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Water thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Tables 1–4 and Figs. 1–15.
Supplementary Data 1
Source data for supplementary figures.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Fig. 7
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jin, B., Liu, H., Che, S. et al. Substantial defluorination of polychlorofluorocarboxylic acids triggered by anaerobic microbial hydrolytic dechlorination. Nat Water 1, 451–461 (2023). https://doi.org/10.1038/s44221-023-00077-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s44221-023-00077-6