Abstract
Predicting potential distributions of species in new areas is challenging. Physiological data can improve interpretation of predicted distributions and can be used in directed distribution models. Nonnative species provide useful case studies. Panther chameleons (Furcifer pardalis) are native to Madagascar and have established populations in Florida, USA, but standard correlative distribution modeling predicts no suitable habitat for F. pardalis there. We evaluated commonly collected thermal traits– thermal performance, tolerance, and preference—of F. pardalis and the acclimatization potential of these traits during exposure to naturally-occurring environmental conditions in North Central Florida. Though we observed temperature-dependent thermal performance, chameleons maintained similar thermal limits, performance, and preferences across seasons, despite long-term exposure to cool temperatures. Using the physiological data collected, we developed distribution models that varied in restriction: time-dependent exposure near and below critical thermal minima, predicted activity windows, and predicted performance thresholds. Our application of commonly collected physiological data improved interpretations on potential distributions of F. pardalis, compared with correlative distribution modeling approaches that predicted no suitable area in Florida. These straightforward approaches can be applied to other species with existing physiological data or after brief experiments on a limited number of individuals, as demonstrated here.
Similar content being viewed by others
Introduction
A key aim throughout many subfields of ecology is predicting where organisms occur. A common approach is to extract landscape and climate data from known occurrences for use in correlative distribution modeling1,2 but these require appropriate sampling of environmental preferences and have other limitations3,4,5. For example, correlative distribution modeling relies on extensive sampling of species presences and an assumption that these presences reflect the existing fundamental niche for the species6,7. Physiological data provide a basis for more mechanistic models and better inform prediction and forecasting of future distribution changes, especially for cryptic, rare, or introduced species8,9,10,11. While collecting physiological data is resource-intensive, doing so can provide information about flexibility of traits, revealing more about the overall fundamental niche (rather than the existing fundamental niche, a best-case outcome for correlative models;6,7) and potential distribution. It is not uncommon for species with restricted distributions to have far broader physiological tolerances, allowing them to establish beyond their known current distribution5,12,13.
A popular approach to integrating physiological data in predictive frameworks are mechanistic niche models, which utilize biophysics to derive physiological constraints and thresholds that can then be projected to the landscape-scale14,15. The demands of many mechanistic modeling frameworks require strong knowledge of species ecophysiology16 and thus are most useful for testing hypotheses in well-studied, model systems7 or in well-funded species where reduced time to accrue the data may not be a priority. While predictions would admittedly be more accurate with comprehensive knowledge of a species, in practice, answers are often required under short timelines with limited prior data and funding. To try and address these demands, we aimed to investigate the utility of models based on easy-to-collect, well known physiological thresholds derived from commonly collected data on physiological tolerances.
We used a species that is cryptic, rare, and introduced to explore the potential seasonal plasticity of physiological tolerance and performance and the utility of different physiologically derived metrics in predicting potential distributions. Of the many potential physiological constraints, we focused on thermal tolerance as it is especially important for determining distributions of ectotherms, which comprise the majority of species on earth17. We assessed thermal traits in the panther chameleon, Furcifer pardalis, which occurs in several introduced populations in Florida, USA with the earliest report from 200818. Introduced populations of chameleons tend to go unreported and some have been “seeded” in areas for later collection due to their high value in the pet trade19. Lack of reporting obscures the knowledge of the current extent of introduced populations, presenting challenges for directing management and regulatory actions. Predicting the distribution of this species in particular is difficult for two main reasons. First, there is relatively little known on thermal limits of this taxon, owing to limited studies of their thermal biology and limited information from related species in the native range in Madagascar20,21. Second, the occurrence of populations in Florida at higher latitudes than the native range suggests a greater thermal tolerance than predicted based on its native range in Madagascar or invasive range of Réunion22. Panther chameleons have a fast life-history strategy, typically maturing within 14 months23, allowing for the possibility of rapid adaptation of introduced populations to local thermal regimes. Thus, we opted to test the thermal traits of chameleons from a population from the highest known latitude24, to account for potential extremes in thermal tolerance.
We assessed thermal tolerance, thermal performance, and thermal preference of F. pardalis exposed to seasonal fluctuations in climate. We hypothesized that chameleons would show a shift in thermal traits associated with seasonal exposure; in particular, we predicted that thermal preference may decrease and cold tolerance and performance at low temperatures would improve following exposure to winter conditions. We then used these trait data and fine-grain, daily temperature data to predict suitable winter habitats across Florida. We also constructed correlative niche models using native range occurrence data, which is often the only option for predicting suitable habitats in data-limited non-native species when physiological data are not available. We then evaluated which models and which thermal traits may be most useful for such predictions. We predicted that the correlative niche model would under-perform compared to models derived from physiological trait data, because of the apparent niche-shift in F. pardalis in Florida. Of the models integrating physiological trait data, we predicted that models integrating critical thermal minima would be most restrictive, followed by those integrating preference and performance.
Results
Thermal limits
Body size of chameleons was 13.6 + /− 2.5 cm snout to vent length (SVL) and 70.7 + /− 33.5 g. CTmin averaged 9.9 °C + /− 2.0 (SD), with an average rate of chameleon temperature change of -0.6 °C + /− 0.3 (SD) per minute. There was an effect of season on critical thermal minimum (CTmin; F2, 9 = 4.31, p = 0.05), though Tukey post-hoc tests did not reveal significant differences among seasons (all p > 0.1; Fig. 1). CTmin did not appear to be affected by which trial was experienced first (F1, 9 = 3.85, p = 0.08). We did not find an interaction between chameleon temperature rate change and mass (F1, 9 = 0.80 p = 0.39), and neither mass nor temperature rate change alone influenced CTmin (F1, 9 = 3.06, p = 0.11; F1, 9 = 0.52, p = 0.49, respectively).
Thermal metrics and associated standard errors from chameleons collected from the northernmost established population of Furcifer pardalis in Florida, USA after exposure to seasonal fluctuations in temperature at USDA Wildlife Services National Wildlife Research Center in Gainesville, Florida, from October 2020 to August 2021. CTmin refers to the critical thermal minimum, the temperature at loss of righting response; CTgape refers to the gaping threshold, the temperature at which chameleons gape to thermoregulate; Tbreadth refers to the difference between the CTgape and CTmin; Tpref refers the preferred body temperature, as the average body temperature selected in a thermal gradient.
CTgape averaged 39.2 °C + /− 1.4 (SD), with an average rate of chameleon temperature change of 0.6 °C + /− 0.2 (SD) per minute. We did not find an effect of season on the high temperature at which gaping occurred (CTgape; F2, 9 = 2.18, p = 0.17; Fig. 1). CTgape was not affected by which trial was experienced first (F1,9 = 0.08, p = 0.79). We did not observe an interaction between chameleon temperature rate change and mass (F1, 9 = 0.56, p = 0.48), nor did we observe an effect of mass (F1, 9 = 0.35, p = 0.57). Rate of chameleon temperature change was negatively correlated to CTgape, such that an increase of heating rate by 1 °C per minute led to lower CTgape by 6.6 °C (F1, 9 = 6.18, p = 0.04).
Average Tbreadth was 29.3 °C + /− 2.7 (SD). There was an effect of season on thermal breadth (Tbreadth; F2, 11 = 5.69, p = 0.02), although Tukey post-hoc tests did not distinguish significant differences in Tbreadth among the seasons (all p > 0.1; Fig. 1). Tbreadth was not affected by which trial was experienced first (F1, 11 = 2.84, p = 0.12), nor was it influenced by mass (F1, 11 = 1.81, p = 0.21).
Thermal preference
The maximum body temperature reached during a preference trial was 40.6 °C and the minimum was 21.9 °C. Average preferred body temperature was 32.8 °C + /− 2.9 and did not differ by season (F2, 12 = 1.23, p = 0.33; Fig. 1), and was not affected by mass (F1, 12 = 0.011, p = 0.919). The body temperature exhibited most often by each chameleon (mode) was 34.2 °C + /− 4.7, and also did not differ by season (F2, 12 = 2.78, p = 0.10) and was not affected by mass (F1, 12 = 0.32, p = 0.59). The standard deviation of each chameleon’s body temperature during a trial was 2.9 °C and also did not differ by season (F2, 12 = 2.78, p = 0.10) and was not affected by mass (F1, 12 = 0.32, p = 0.59).
Thermal performance
Thermal sensitivity of sprint performance (Q10) differed significantly among temperature intervals (F3, 80 = 4.71, p = 0.005). Tukey post hoc tests revealed that Q10 values for the 15–20 °C interval (x̅ = 5.08) differed significantly from the 25–30 °C interval (x̅ = 1.86, p = 0.01) and from the 30–35 °C interval (x̅ = 4.50, p = 0.008), but not the 20–25 °C interval (x̅ = 2.78, p = 0.43). Q10 values were not affected by SVL (F1, 80 = 0.008, p = 0.93). Additionally, season had no impact on thermal sensitivity of sprint performance (F2, 80 = 0.052, p = 0.95; Fig. 2).
Q10 values calculated from maximal sprint speed for four temperature intervals tested in individuals from the northernmost established population of Furcifer pardalis in Florida, USA after exposure to seasonal fluctuations in temperature at USDA Wildlife Services National Wildlife Research Center in Gainesville, Florida, from October 2020 to August 2021. These values represent the magnitude of increase in a rate with a 10 °C increase in temperature, such that a value of two represents a doubling the rate per 10 °C increase, values of one represent thermal independence, and values of 0.5 represent a halving of the rate per 10 °C increase.
Correlative distribution model
The top model (Supplementary Fig. S4) predicted no suitable habitat for F. pardalis in Florida, despite established populations existing there. The top model had a regularization multiplier of 2 and used feature classes of linear, quadratic, hinge, product, and threshold, with AICc = 3288.793 and AUC of 0.916 (see data repository for details). The final set of layers and their percent contribution consisted of: temperature annual range (bio7, 82.32%), precipitation of wettest month (bio13, 12.53%), maximum temperature of the warmest month (bio5, 2.04%), precipitation of the warmest quarter (bio18, 2.01%), and isothermality (bio3, 1.10%).
Integrative distribution models
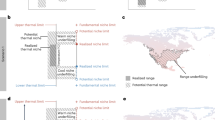
Summary data extracted from model projections at F. pardalis presences (N = 9 distinct presences within the 1 km resolution of PRISM data) are presented in Table 1. F. pardalis populations have established in areas where the winter temperatures fell below the lowest critical thermal minimum for 6 consecutive hours an average of fewer than 10 days per season (Fig. 3, Table 1). At the other two thresholds, populations have established in areas experiencing the temperature threshold for under one month each winter (9 °C, near average CTmin) and 5 days per season (3 °C, below CTmin and exposed temperatures; Table 1; see Supplementary Fig. S5 online). The average activity window estimated for areas F. pardalis have established falls between 2.97 and 6.28 h of 11 h of available daylight per day (Table 1), and this window decreases with increasing latitude (Fig. 4). At the highest latitude populations, there is an average of 3–4 h per day through the winter period where ambient air temperatures are within the preferred temperatures of F. pardalis (Fig. 4). The entire state of Florida falls within 80% of the average predicted performance for F. pardalis during the winter period (Fig. 5), and the predicted performance at areas F. pardalis have established exists in a relatively narrow range of 44.5–63.2% (Table 1). The normalized activity window and predicted performance were relatively similar in their predictions; however, the activity window was slightly more conservative in its output, placing established populations in a threshold 10% more restrictive than the performance model (see Supplementary Fig. S6 online).
Average number of days during each winter (Dec 15–Feb 15) in Florida, USA, where temperature fell below the lowest critical thermal minimum of Furcifer pardalis for 6 or more hours, from 2001 to 2021. Yellow points indicate locations where F. pardalis populations have established. White point indicates the location of the experimental exclosure at USDA Wildlife Services National Wildlife Research Center in Gainesville, Florida.
The average number of potential daylight hours Furcifer pardalis may be active in Florida, USA during the winter (Dec 15–Feb 15) from 2001 to 2021, based on thermal preference data for the northernmost population of this species in Florida. There are 11 possible total hours of daylight during this winter period. Yellow points indicate locations where F. pardalis populations have established. White point indicates the location of the experimental exclosure at USDA Wildlife Services National Wildlife Research Center in Gainesville, Florida.
The predicted performance of Furcifer pardalis in Florida, USA, expressed as a percentage of the maximum predicted performance value from a generalized additive model of thermal performance data from the northernmost population of this species in Florida (inset). Yellow points indicate locations where F. pardalis populations have established. White point indicates the location of the experimental exclosure at USDA Wildlife Services National Wildlife Research Center in Gainesville, Florida.
Discussion
We conducted the first tests of seasonal acclimation of thermal traits in Furcifer pardalis, and found that CTmin, CTmax, Tpref, and performance were not influenced by season. We anticipated seasonal acclimatization in thermal traits after exposure to low temperatures. For example, increased cold tolerance is reported after acclimation to cool temperatures in other arboreal lizards12,25; increases in thermal preference follow increases in ambient temperatures in newts26; and introduced geckos prefer lower temperatures in winter27. Our general lack of observed seasonal acclimation may be partly influenced by experimental choice; namely exposure of chameleons to natural temperature fluctuations rather than constant temperature treatments in each season, combined with natural variation in acclimation ability among individuals28, but see29. In other tropical species, extended constant exposure to low temperatures may be necessary to observe acclimation of thermal traits e.g.30. The influence of seasonal acclimatization on thermal traits varies widely31. Some species display acclimation of certain thermal traits, but not others. For example, introduced curly-tailed lizards show seasonal acclimation in CTmax but not CTmin32, and in the frog Pleurodema thaul thermal performance and preference were not influenced by acclimation temperature, but thermal tolerance was33. Other species may show inter-population variation in acclimation. In A. cristatellus, one population demonstrates ability to acclimate to cold temperatures, but another population of the same species does not12, while in the armadillo girdled lizard (Ouroborus cataphractus), two populations maintain similar thermal preference despite seasonal change34. Discrepancies and patterns in acclimation of thermal traits are well documented elsewhere e.g.31, and the underlying mechanisms for these deserve further study.
In the case of F. pardalis, a potential explanation for lack of observed acclimatization is that the study population may have adapted to local thermal conditions to express conserved thermal traits year-round. The fact that the individuals we tested are from the highest latitude documented for the species18, that F. pardalis have relatively fast generation times23, and that this species does not typically burrow or seek shelter to buffer from cold temperatures (Claunch pers obsv) lends credence to the possibility of local adaptation. Adaptation without acclimation is not unprecedented. Another tree-dwelling lizard, A. cristatellus, demonstrates differences in CTmin among introduced and native populations, without associated acclimation ability in one of the introduced populations25. Local adaptation of CTmax35; CTmin36,37 and thermal preference27 are documented among various lizard populations. It is important to note that our study is not equipped to test whether adaptation or acclimatization had occurred in the F. pardalis population prior to our testing. Unfortunately, our attempts to include chameleons from lower-latitude populations in a common-garden style comparison were thwarted due to collection-depletion and lack of public access at documented sites. The native-range origin of our study animals is also not known with certainty—coloration suggests they may be hybrids from multiple geographic origins18. Our preferred body temperature result is slightly higher than a previous study on F. pardalis—where Ferguson et al.20 report preferred temperatures from three individuals as 31 °C, we report slightly higher preferred temperatures approaching 33 °C. While gaping thresholds are not reported, Ferguson et al.20 report panting observed above 36 °C; this may represent the lowest observed panting threshold, as gaping typically precedes panting and we report gaping at temperatures averaging around 40 °C. Expanded sampling of multiple populations will be necessary to determine whether this population is representative of inherent thermal plasticity in F. pardalis or represents improved cold tolerance at the extremes of thermal plasticity in the species.
As anticipated, the correlative niche modeling approach, which did not integrate physiological data, under-predicted suitable habitat in the non-native range in Florida. This can largely be attributed to using native occurrences to model the invaded range. We chose this approach because it is the best practice approach in species distribution modeling of invasive species. Modeling invaded ranges using occurrences from within that range is problematic because of the lack of presumed equilibrium with the environment and often sparse occurrence data4. These issues especially limit the ability to effectively forecast distributions of nonnative species under climate changes7,38. While it is possible to combine native and nonnative populations into a joint model of F. pardalis distribution, the data density remains weighted strongly towards the native range and the end result is a model tuning exercise to determine how to balance omission and commission errors, without a clear external means to assess optimal model quality. Here, our native-range model is not useful for informing about potential distributions of F. pardalis in Florida, but may instead provide important baseline niche information, which can be used to compare the magnitude of potential niche shift.
Models directly integrating physiological information may be the most effective way to improve predictions of potential distribution of invaders8,39,40. Of the physiological traits we tested, cold tolerance is likely the most limiting factor for F. pardalis range expansion in Florida, given what we know about its native abiotic niche. Our lack of observed seasonal differences among thermal traits justify the use of average trait values, which simplifies modeling approaches and interpretation. Namely, our model predictions would remain largely unchanged if we had only used thermal trait data derived from a single sampling effort. Additionally, because we tested individuals from a population that is of higher latitude than its native range, data from this population may give a decent approximation of the limits of thermal flexibility of the species. At the very least, we demonstrate that physiological tolerance information even on a limited number of individuals can provide a better estimate of occurrence than comparatively abundant occurrence data from the native range. By integrating physiological data from populations at the edge of their niche into niche modeling frameworks, we can gain a more accurate picture of the niche limits of this species than is possible with occurrence data alone.
Our use of consecutive hours at each minimum temperature threshold is a conservative method to apply physiological data to understanding chameleon occurrence. Ectotherms can often survive brief exposure to temperatures below their CTmin, while longer exposure can lead to death, e.g.41. By considering length of exposure to cold thresholds, we may gain a more ecologically relevant insight into distribution limits and elucidate where population-limiting temperature thresholds occur. As expected, as temperature thresholds decrease, the average number of Florida-wide occurrences of each threshold also decreases. In areas where chameleons have established, there are fewer instances of potential exposure to cool temperatures. Chameleons are potentially exposed to 4 total weeks where temperatures are 1 °C below their average CTmin for at least 6 consecutive hours; whereas they potentially experience 2 weeks of temperatures at the lowest measured CTmin, and less than one week at the 3 °C threshold (Fig. 3; see Supplementary Fig S5 online). Existing populations showed less variation in 6-h exposures to the lowest two temperature thresholds than near the average CTmin. That the near-average CTmin threshold was not as useful as the lowest and below-CTmin thresholds suggests that the more extreme cold tolerance values more closely represent population-limiting temperature exposures, especially when considering the temperature values used in thresholds were derived from brief exposures to determine CTmin.
The use of average critical thermal limit thresholds in distribution models has been criticized when applied as a filter of single-value temperature occurrences in a landscape (i.e., when not accounting for exposure time;42), but also because CTmin is often several degrees above the lethal minimum temperature16. In some cases species may be immobilized by cold but are able to survive long bouts of cold exposure43. Lower lethal temperatures are unknown in F. pardalis. Ethical concerns aside, lethal temperature values may be too restrictive an approach to predicting suitable habitat. For example, a chameleon experiencing a chill coma (a temperature at or below CTmin) may not die directly from low temperatures but the restricted foraging efforts, increased vulnerability to predators, and dampened immune function at low temperatures may manifest as population-level effects that prevent sustained survival of populations at higher-than-lethal temperature thresholds. The range in intra-species thermal tolerance can serve as a starting point for developing informative temperature thresholds on activity restriction in a species. A more holistic approach integrates chill coma temperatures or CTmin as well as higher temperatures where activity and performance are reduced but may still limit population survival.
Activity window and thermal performance thresholds provide perspective on potential behavioral limitations using ambient temperature data. Calculation of activity windows from thermal trait data is not new; there are many frameworks used to estimate activity windows and activity budgets, ranging from models requiring many data inputs, (e.g.44,45,46), to simpler threshold-based inputs (e.g.47). Our approach differs slightly from others in the calculation of hourly trait values, and in our choice of thermal preference or selected body temperature data as the basis for an activity window. The range of body temperatures we recorded (22–40 °C) in the thermal preference trial encompasses the range of field body temperatures reported from F. pardalis in Madagascar exhibiting normal activity (24–36 °C20). This demonstrates that our thermal preference data accurately reflect an activity window for the species. Our normalized data comparisons confirm that our activity window data derived from thermal preference are more restrictive than performance data. This is expected, as our activity window is constrained to temperature values chameleons chose, while the performance data clearly demonstrate that chameleons are capable of activity at higher and lower temperatures when induced to move.
The activity window and thermal performance thresholds represent different constraints and should be interpreted in different ways. The activity window, derived from thermal preference data, more likely demonstrates propensity to forage or explore, whereas temperatures outside this window are more likely directed towards seeking thermal refugia such as spending time basking to raise temperatures to levels where foraging can occur. The performance threshold more likely represents the ability to respond when extremely motivated to move, such as during pursuit by a predator. However, performance data may not be as useful a threshold in F. pardalis as it is for some other ectotherms. First, no area in Florida fell below 20% of the maximum predicted performance for this species, and predicted performance was around 50% at established populations, so variation in this trait was not particularly informative when applied to Florida winters. Second, sprint data may not correlate to success of escape from predators, as chameleons are fairly slow reptiles even at their best performance. Thermal preference data thus may be more biologically relevant, because if chameleons choose not to forage outside of their preferred temperatures they may not meet energy requirements to survive the winter. It is important to note these models do not account for potential radiative heating of basking chameleons to combat low ambient temperatures48, and conversely do not account for convective or conductive cooling during wind or rainfall. Indeed, the combination of fine-scale behavior data with radiation, windspeed, and precipitation data may improve the resolution of predictions and has been proposed in more complex frameworks that also estimate activity budgets (e.g.44,45). We chose to use only ambient temperature data as this is most commonly available49 and may be the only climate data consistently available in regions of the world where ectotherm biodiversity is highest50,51,52,53. We argue the value of our simple framework is that it can be applied in data-limited contexts, especially as global, fine-scale daily temperature data are now available worldwide49.
Conclusions
We have shown that easy-to-collect physiological thermal trait and ambient temperature data can be used to accurately predict distributions of ectothermic organisms, in absence of native-range occurrence data. The thresholding approaches applied herein are relatively simple to execute, and limitations lie with computing power (which is typically not prohibitive), depending on the resolution and extent of predicted areas. While our case study focused on a non-native animal in an introduced range, the techniques herein are applicable beyond predicting distributions of invasive species. While mechanistic modeling approaches can have great utility for conservation with adequate inputs54, the majority of terrestrial ectothermic diversity and in turn the majority of data-deficient and at-risk ectotherms with limited occurrence data are unlikely to be candidates for more sophisticated mechanistic modeling approaches7. However, daily global temperature data are becoming available49, which creates opportunities for modeling approaches such as ours, which apply commonly available thermal trait data. These methods can thus be applied to improve understanding of distributions in data-deficient, rare, or threatened species using physiological data derived from a limited number of individuals. The methods could also be applied to simulate future distributions using predicted daily climate data. Finally, we provide predictions of potential occurrences of F. pardalis in Florida that may help focus management surveillance efforts. The limitations and accuracy of our approaches for this particular species will become clearer as additional populations are located.
Methods
Animal collection and housing
Ten chameleons were captured from an established non-native population in Central Florida from October 2019-February 2020 (N = 7 males, 2 females) and October 2020 (N = 1 male). Adult panther chameleons were housed individually in an experimental mesh exclosure at the US Department of Agriculture (USDA), Wildlife Services, National Wildlife Research Center, Florida Field Station, located approximately 115 km north of the collection site in Gainesville, Florida from September 30, 2020, to July 28, 2021. Conflicts with residents at the site of the established population prevented collection of more individuals24. Nine of the chameleons were housed by the authors prior to accessioning at USDA; the tenth was accessioned directly from the wild population to the exclosure in October 2020 (detailed in55). Animals were housed individually in screen enclosures with natural vegetation and exposure to natural sunlight and weather patterns in the greater exclosure (see Supplementary Fig. S1 online). An automated misting system provided dripping water for drinking four times daily. Chameleons were fed every other day with crickets dusted with calcium without D3 (Rep-Cal, Rep-Cal Research Labs, Los Gatos, California, USA) at every feeding, except when replaced with a multivitamin (Reptivite, Zoo Med, San Luis Obispo, California, USA) dusting once every other week. Chameleons received visual wellness checks once daily. Chameleons were exposed to natural thermal regimes, including low nighttime temperatures (12 °C) in winter (Fig. 1). On nights forecast below 12 °C, 250 W heat emitters were turned on over the cages and tarps placed to cover the sides and roof (see Supplementary Fig. S1 online). On nights forecast below 7 °C, chameleons were brought indoors (65–70 °C) and held in individual cloth bags. Thermal data loggers (iButton DS1922L, resolution 0.06 °C, accuracy 0.5 °C, Maxim Integrated, San Jose, California, USA) were placed within screen enclosures to collect data on variation in ambient temperatures (see Supplementary Fig. S2 online). All protocols were approved by USDA QA-3214 (Study Director, Bryan Kluever) and University of Florida IACUC 201,910,938.
Assessing thermal traits
Thermal limit thresholds, preferences, and performance of chameleons were evaluated in three seasons: November 2020 (Fall), February 2021 (Winter), and July 2021 (Summer; see Supplementary Fig. S2 online). At each season, we collected mass using spring scales (Pesola Präzisionswaagen AG Schindellegi, Switzerland 0100 and 40,300 1 g and 2 g resolution, respectively) and SVL using a measuring tape (1 mm resolution). Animals were not evaluated in Spring due to outbreak of fungal infection (see55). All animals were de-accessioned from the study as of 1 August 2021. Chameleons were assessed for critical thermal limit thresholds before preference or performance trails were conducted. We conducted linear mixed models using the lme function in package nlme56 in R57. The alpha level for all statistical tests was set at 0.05.
Critical thermal limit thresholds
Due to the proximity of critical thermal maximum to lethal maximum in some animals58,59 and the limited number of animals available for testing, we opted to measure the body temperature at which gaping occurred as a heat stress response (CTgape) as our upper thermal threshold. We used the body temperature at loss of righting response (CTmin) as our lower temperature threshold.
At each season, individuals were randomly assigned to be tested first for either CTgape or CTmin. After full recovery from each threshold, chameleons were returned to their cages and were tested the following day for the remaining threshold. Animals were acclimated to room temperature (26 °C) for at least one hour prior to conducting thermal limit threshold measurements. A thermal probe was inserted into the cloaca and secured with medical tape to allow continuous recording of animal body temperature every 10 s throughout the trial. For CTmin, animals were placed into a cooling incubator with windows (Benchmark Scientific, Sayreville, New Jersey, USA) set to 6 °C. When body temperatures were below 15 °C, we placed chameleons on their side with a gloved hand to test for righting response every 1.5 min and every 1 °C decrease in body temperature, whichever occurred first. When an animal was unable to right itself for 10 s after being flipped onto its side, we considered this CTmin, recorded the time, and removed the animal from the incubator to recover. For CTgape, animals were placed into a heated incubator with windows (Labnet International Inc, Woodbridge, New Jersey, USA) set at 45 °C. We considered the gaping threshold reached when an animal held its mouth open for at least 5 s. We then recorded the time and removed the animal from the incubator to recover. All animals recovered from thermal limit threshold testing without incident. We extracted body temperatures for each threshold at the time each threshold was reached from the thermal logger data. We calculated the thermal breadth (Tbreadth) for each season for each individual by subtracting the CTmin from the CTgape value.
We conducted linear mixed models with gaussian error distribution with fixed response variables CTmin, CTgape and Tbreadth. Because rates of temperature change can influence thermal limit thresholds12,42 we calculated the average rate of body temperature change per minute during the trial and included this as a covariate in CTmin and CTgape analyses. In the thermal threshold models we included the following variables: season, which trial was experienced first (i.e. CTmin or CTgape trial), and the interaction between rate of body temperature change and mass. In the Tbreadth model we included season, which trial was experienced first, and mass as covariates. To account for repeated measures, animal ID was included as a random intercept effect in all models. Animals that were moribund or in poor body condition were excluded from analyses (N = 1 female in winter, N = 1 male in summer; see Supplementary Fig. S2 online). Where a factor variable (e.g., season) indicated significance at a threshold of alpha = 0.05, we conducted a Tukey post-hoc test using package emmeans60 in an attempt to discriminate differences among factor levels.
Thermal preference
To assess thermal preference, a thermal gradient was created in a 1.25 m by 2 m arena divided in half to create two lanes. To facilitate use by chameleons, two wooden dowels were inserted into the center of each lane. At one end, two 250 W and two 150 W ceramic heat emitters were arranged to provide a hot environment to 51 °C ambient temperature. The other end was surrounded by ice packs and a bucket of salted ice with two small electronic fans (Shenzhen Glovion Technology Co., Shenzhen, China) to maintain cool air flow to 18 °C ambient temperature (see Supplementary Fig. S3 online). A curtain was drawn to prevent chameleons from reacting to researcher presence. Chameleons were monitored occasionally from behind the curtain to ensure they remained on the dowels. Chameleons had a thermocouple (0.076 mm diameter, Item 5SRTC-TT-K-40–72, Omega Engineering) inserted into the cloaca and secured with medical tape, attached by a 1.8 m lead to a datalogger (Item# OM-HL-EH-TC, Omega Engineering, resolution 0.1 °C, accuracy 0.8 °C) set to collect temperatures every 10 s for 65 min. The length of thermocouple leads and their small size and weight allowed for unrestrained movement throughout the area. Chameleons were initially placed in the middle of gradient. The first 5 min of data after animals were introduced to the arena were discarded to account for an acclimation period to the arena and after thermocouple insertion.
We extracted the average, mode, and standard deviation in body temperature from each chameleon’s thermal preference trial thermal logger data for analysis. Two chameleons’ thermocouples fell out within 15 min of the end of the trial (Fall season), and two chameleons exited the gradient mid-trial at which point they were replaced into the gradient and the portion of data where the chameleon was outside the gradient plus one minute after being replaced was discarded (Fall season). We used the remaining within-gradient trial data for analysis for these cases.
After confirming normality of data, we conducted a linear mixed model on the response variables of average, mode, and standard deviation of body temperature during the thermal preference trials. To account for repeated measures, we included chameleon ID as a random effect. We included mass and season as covariates. All chameleons walked to the hot end of the gradient after initial placement, thus starting choice was not included in analysis.
Thermal performance
To assess thermal performance we tested chameleon sprint speed at five different body temperatures. Chameleons were randomly assigned to be tested at either “warm” (30 and 35 °C) or “cool” (15, 20, and 25 °C) temperatures per day. To achieve the assigned body temperature, chameleons were placed into either a cooling or heating incubator (described above in Critical Thermal Limit Thresholds) and continually monitored until the target temperature was reached, indicated by thermocouple in the cloaca. After reaching the target temperature, thermocouples were removed, and we placed chameleons at one end of a rubber mat divided into six, 0.25 m segments, and encouraged them to sprint across the mat by simultaneously tapping their tail gently with a gloved hand and luring with a bamboo branch ahead of the chameleon as it moved to the other side. During this, an observer used a stopwatch to record the time for the tip of the chameleon’s snout to pass each 0.25 m segment (segment time). Upon completing the entire length of the mat (1.5 m), we immediately repeated the process at the starting point of the mat; thus every trial consisted of two “laps”. Chameleons were allowed a minimum of 30 min rest between trials and chameleons completed two trials at each temperature. If the chameleon refused to move or was uncooperative (i.e., clearly performed submaximally), we denoted trials as being unsuccessful and excluded these trials from further analyses. We calculated the velocity of each segment by dividing the distance by segment time. For each acclimation temperature and chameleon, we retained only the maximal sprint speed over a 0.25-m segment for further analysis.
To compare the thermal sensitivity of sprint performance among seasons, we calculated Q10 values for each temperature interval. These values represent the magnitude of increase in a rate with a 10 °C increase in temperature, such that a value of two represents a doubling the rate per 10 °C increase, values of one represent thermal independence, and values of 0.5 represent a halving of the rate per 10 °C increase. Q10 values were calculated using the following equation61:
where vi represents velocity and Ti the corresponding acclimation temperature. We calculated four Q10 values corresponding to intervals of 15–20, 20–25, 25–30, and 30–35 °C. Trials were discarded if they spanned larger intervals (i.e., if an individual was only tested at 15 and 25 °C). To determine if season affected thermal sensitivity, we performed a linear mixed model with Gaussian distribution on the Q10 values. We used temperature interval and SVL as covariates; SVL was log-transformed to account for allometric effects. To account for repeated measures, we included individual ID as a random factor. All Q10 values were natural-log-transformed to meet model assumptions of homoskedasticity.
Distribution modeling
Correlative niche model
We used Maxent to construct correlative niche models using native range occurrence and climate data to project onto Florida. Occurrence data for F. pardalis spanning from 1876 to 2022 were downloaded from various databases62,63,64 and were sorted to remove duplicate records, records without georeferences, and improbable georeferences based on locality descriptions. Only points from the native range (Madagascar) were considered; we did not include data from nearby introduced populations of F. pardalis on Réunion. We thinned the points at a 4 km threshold to a total of 149 remaining points.
We calibrated models using ENMeval65, using the ENMevaluate function with extrapolation and without clamping using default parameters and input of 19 bioclimatic layers at 1 km2 resolution (WorldClim266) in the native range region. This method evaluates models constructed with varying combinations of regularization multipliers (0.5, 1, 2, 3, 4) and feature classes (linear, linear—quadratic, linear-quadratic-hinge, linear-quadratic-hinge-product, and linear-quadratic-hinge-product-threshold). Before running models in ENMevaluate, and to avoid potentially problematic multicollinearity in our models, we calculated the variance inflation factors (VIF) of our initial model with all 19 bioclimatic variables. If any predictor variable had a VIF > 5, we removed the variable with the lowest permutation contribution to the model. ENMevaluate was used to select the top model based on the AICc value within 2 to the lowest AIC model. We then used the top model to project onto Florida using the Maxent graphical user interface67 with extrapolation and no clamping and 10,000 background points.
Thresholded winter temperatures: cold tolerance, activity window, predicted performance
Because cold tolerance is often the most limiting factor for ectotherms at higher latitudes68, and because F. pardalis CTgape exceeds typical annual maximum ambient temperatures in Florida, we chose to subset our distribution modeling to winter environmental data. To examine physiological data in the context of environmental temperatures in Florida, we created thresholds based on our thermal trait data. First, we assembled PRISM climate data for the extent of Florida for December 15 to February 15 from 2001 to 202169,70 at 800 m resolution and extracted daily minimum (Tmin) and maximum (Tmax) temperatures. These dates encompass the 7 years prior to the first reported population, to account for potential lag times in reporting or discovery of this and other populations71. While ambient air temperature data has been criticized for use in predictive modeling frameworks (reviewed in16), such data may provide a decent approximation of available temperatures in this arboreal species72.
We used package ChillR to calculate hourly temperatures for each 800 m pixel in Florida for each day over December 15th to February 15th time period for all years. ChillR takes as input Tmin and Tmax as well as sunrise and sunset times, and latitude73. It then applies an algorithm from74 which models hourly temperatures by fitting a sine curve for daytime temperatures, and a logarithmic decay function for nighttime temperatures. To assess how cold tolerance (CTmin) may influence F. pardalis distribution in Florida, we flagged the hourly temperature data where the temperature was below three different thresholds (see below) for six or more consecutive hours. The six-hour exposure time threshold accounts for time to overcome thermal inertia associated with differences in cooling rates of ambient temperature and body temperature16, as well as removes potential noise in the data associated with rapid but unsustained changes in temperature (e.g., a passing storm front). We counted the number of days that met this condition for each threshold for each yearly period, and then averaged the number of occurrences across those winter periods for reporting. The thresholds were: 9 °C, representing ~ 1 °C below the average measured CTmin in this study; 6 °C, representing the lowest individual CTmin recorded in this study and the lowest temperature experienced in the exclosure; and 3 °C, below temperatures experienced by chameleons in this study and below recorded CTmin for the species.
To examine the remaining thermal traits, we subset the hourly temperature data to daylight hours, as F. pardalis is diurnal and reliably falls asleep upon darkness (Claunch and Goodman pers obs). We calculated an activity window for F. pardalis by categorizing all temperatures during daylight hours that fell within the recorded body temperatures in the thermal preference gradient as active hours. We examined the activity window in two ways: first by averaging the number of active hours across all winters for each pixel in Florida, then by calculating the percent of active hours falling within available daylight hours across all winters for each pixel. We also calculated predicted performance during winter to serve as a proxy for the relative ability for F. pardalis to forage or engage in escape behaviors. To do this, we generated a performance curve using a generalized additive model of the maximum individual velocities per 25 cm segment at each test temperature bookended by velocities equaling 0 at CTmin and CTmax test temperatures, with k = 5 and smoothing parameter = 0.1 (Fig. 5). From this curve, we calculated the predicted performance for each daylight hour’s temperature at each pixel. We assessed performance in two ways: (1) by averaging the predicted performance across all winters per pixel; (2) by thresholding performance values that fell within 80% of maximum performance in the GAM across all winters per pixel.
Assessing model predictions
We assessed model predictions in two ways. First, we overlaid known established or formerly established presences in Florida on the Florida projections from all models described above, then extracted the associated data at these localities for comparison and evaluation. Because verified established presences are limited in number for F. pardalis, we also compared state-wide model projections produced by each metric. Second, to compare the utility of activity versus performance models, we normalized model outputs by setting the lowest value to 0 and the maximum value to 1; this was achieved by subtracting the lowest output value from all records, then dividing all output records by the resultant maximum value for each model. Thus, the scale reflects relative predicted activity and performance such that a value closer to 0 reflects the minimum predicted activity or performance, while a value closer to 1 reflects the maximum predicted activity or performance.
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed. All protocols were approved by USDA QA-3214 (Study Director, Bryan Kluever) and University of Florida IACUC 201,910,938. The reporting in the manuscript follows the recommendations in the ARRIVE guidelines.
Data availability
Data for experimental determination of thermal traits are archived with USDA75. The data and code generated during the current study are available at GitHub [https://github.com/nmclaunch/F_pardalis_thermal].
References
Peterson, A. T., et al. Ecological niches and geographic distributions. MPB-49. (Princeton University Press, 2011)
Peterson, A. T. & Soberón, J. Species distribution modeling and ecological niche modeling: Getting the concepts right. Nat. Conserv. 10, 102–107 (2012).
Rodda, G. H., Jarnevich, C. S. & Reed, R. N. Challenges in identifying sites climatically matched to the native ranges of animal invaders. PLoS ONE 6, e14670 (2011).
Jarnevich, C. S., Stolhgren, T. J., Kumar, S., Morisette, J. T. & Holcombe, T. R. Caveats for correlative species distribution modeling. Ecol. Inform. 29, 6–15 (2015).
Wiens, J. J., Litvinenko, Y., Harris, L. & Jezkova, T. Rapid niche shifts in introduced species can be a million times faster than changes among native species and ten times faster than climate change. J. Biog. 46, 2115–2125 (2019).
Kearney, M. & Porter, W. P. Mapping the fundamental niche: Physiology, climate, and distribution of a nocturnal lizard. Ecol. 85, 3119–3131 (2004).
Tourinho, L. & Vale, M. M. Choosing among correlative, mechanistic, and hybrid models of species’ niche and distribution. Integr. Zool. 18, 93–109 (2021).
Kearney, M. et al. Modelling species distributions without using species distributions: the cane toad in Australia under current and future climates. Ecography 31, 423–434 (2008).
Rödder, D., Schmidtlein, S., Veith, M. & Lötter, S. Alien invasive slider turtle in unpredicted habitat: a matter of niche shift or of predictors studied?. PLoS ONE 4, e7843 (2009).
Saleeba, K., Phillips, B. L., O’Shea, M. & Kearney, M. P. Using biophysical models to improve survey efficiency for cryptic ectotherms. Wildl. Manag. 84, 1185–1195 (2020).
Boardman, L. et al. The future of invasion science needs physiology. Bioscience 72, 1204–1219 (2022).
Kolbe, J. J., VanMiddlesworth, P. S., Losin, N., Dappen, N. & Losos, J. B. Climatic niche shift predicts thermal trait response in one but not both introductions of the Puerto Rican lizard Anolis cristatellus to Miami, Florida. USA. Ecol. Evol. 2, 1503–1516 (2012).
Claunch, N. M., Goodman, C., Reed, R. N., Romagosa, C. M. & Taylor, E. N. Invaders from Islands: Thermal Matching, Potential, or Plasticity?. Biol. J. Linn. Soc. 134, 587–603 (2021).
Kearney, M. & Porter, W. Mechanistic niche modelling: Combining physiological and spatial data to predict species’ ranges. Ecol. Lett. 12, 334–350 (2009).
Peterson, A. T., Papeş, M. & Soberón, J. Mechanistic and correlative models of ecological niches. Eur. J. Ecol. 1, 28–38 (2015).
Taylor, E. N. et al. The thermal ecology and physiology of reptiles and amphibians: A user’s guide. J. Exp. Zool. A 335, 13–44 (2020).
Atkinson, D. & Sibly, R. M. Why are organisms usually bigger in colder environments? Making sense of a life history puzzle. Trends Ecol. Evol. 12, 235–239. https://doi.org/10.1016/S0169-5347(97)01058-6 (1997).
Fieldsend, T. W. et al. Extreme male color polymorphism supports the introduction of multiple native-range panther chameleon (Furcifer pardalis) lineages to Florida. USA. Reptil. Amphib. 28, 255–256 (2021).
Edwards, J. R., Rochford, M. R., Mazzotti, F. J. & Krysko, K. L. New county record for the veiled chameleon (Chamaeleo calyptratus Dumeril and Bibron 1851), in Broward County, Florida, with notes on intentional introductions of chameleons in southern Florida. Reptil. Amphib. 21, 83–85 (2014).
Ferguson, G. W., Murphy, J. B., Ramanamanjato, J.-B. & Raselimanana, A. P. The Panther Chameleon: Color Variation, Natural History, Conservation, and Captive Management (Krieger Publishing Company, 2004).
Andrews, R. M. Lizards in the slow lane: Thermal biology of chameleons. J. Therm. Biol. 33, 57–61 (2008).
Bourgat, R. M. Étude des variations annuelles de la densité de population de Chamaeleo pardalis Cuv., 1892, dans son biotope de l'ile de la Réunion. Vie et Milieu (Ser. C) 19, 199–220 (1968)
Ferguson, G. W. et al. Is the natural UV zone important for successful captive propagation of the Panther Chameleon (Furcifer pardalis); are different UVB irradiance exposures that generate a similar dose equally successful?. Zoo. Biol. 40, 150–159 (2021).
Goodman, C. M., Claunch, N. M., Steele, Z. T., Episcopio-Sturgeon, D. J. & Romagosa, C. M. Colorful lizards and the conflict of collection. Preprint At https://doi.org/10.1101/2023.08.10.552819 (2023).
Kolbe, J. J., Ehrenberger, J. C., Moniz, H. A. & Angilletta, M. J. Physiological variation among invasive populations of the brown anole (Anolis sagrei). Physiol. Biochem. Zool. 87, 92–104 (2014).
Hadamová, M. & Gvoždík, L. Seasonal acclimation of preferred body temperatures improves the opportunity for thermoregulation in newts. Physiol. Biochem. Zool. 84, 166–174 (2011).
Lapwong, Y., Dejtaradol, A. & Webb, J. K. Shifts in thermal preference of introduced Asian house geckos (Hemidactylus frenatus) in temperate regions of southeastern Australia. J. Therm. Biol. 91, 102625 (2020).
Huey, R. B. & Buckley, L. B. Designing a seasonal acclimation study presents challenges and opportunities. Integ. Org. Biol. 4, obac016 (2022).
Young, V. K. H. & Gifford, M. E. Limited capacity for acclimation of thermal physiology in a salamander. Desmognathus. Brimleyorum. J. Comp. Physiol. 183, 409–418 (2013).
Pintor, A. F. V., Schwarzkopf, L. & Krockenberger, A. K. Extensive acclimation in ectotherms conceals interspecific variation in thermal tolerance limits. PLoS ONE 11, e0150408 (2016).
Clusella-Trullas, S. & Chown, S. L. Lizard thermal trait variation at multiple scales: a review. J. Comp. Physiol. B 184, 5–21 (2014).
Neel, L. K., Curlis, J. D., Kinsey, C. K., Cox, C. L. & McBrayer, L. D. Acclimatization in the physiological performance of an introduced ectotherm. J. Exp. Biol. 223, 1–8 (2020).
Ruiz-Aravena, M. et al. Impact of global warming at the range margins: phenotypic plasticity and behavioral thermoregulation will buffer an endemic amphibian. Ecol. Evol. 4, 4467–4475 (2014).
Truter, J. C., van Wyk, J. H. & Moulton, P. F. N. An evaluation of daily, seasonal and population-level variation in the thermal preference of a group-living lizard, Ouroborus cataphractus (Sauria: Cordylidae). Amphib. Reptil. 35, 391–403 (2014).
Campbell-Staton, S. C. et al. Parallel selection on thermal physiology facilitates repeated adaptation of city lizards to urban heat islands. Nat. Ecol. Evol. 4, 652–658 (2020).
Leal, M. & Gunderson, A. R. Rapid change in the thermal tolerance of a tropical lizard. Am. Nat. 180, 815–822 (2012).
Litmer, A. R. & Murray, C. M. Critical thermal tolerance of invasion: Comparative niche breadth of two invasive lizards. J. Therm. Biol. 86, 102432 (2019).
Woodin, S. A., Hilbish, T. J., Helmuth, B., Jones, S. J. & Wethey, D. S. Climate change, species distribution models, and physiological performance metrics: predicting when biogeographic models are likely to fail. Ecol. Evol. 3, 3334–3346 (2013).
Evans, T. G., Diamond, S. E. & Kelly, M. W. Mechanistic species distribution modelling as a link between physiology and conservation. Conserv. Physiol. 3, cov056 (2015).
Enriquez-Urzelai, U., Kearney, M. R., Nicieza, A. G. & Tingley, R. Integrating mechanistic and correlative niche models to unravel range-limiting processes in a temperate amphibian. Glob. Chang. Biol. 25, 2633–2647 (2019).
Andersen, J. L. et al. How to assess Drosophila cold tolerance: chill coma temperature and lower lethal temperature are the best predictors of cold distribution limits. Funct. Ecol. 19, 55–65 (2015).
Rezende, E. L., Castañeda, L. E. & Santos, M. Tolerance landscapes in thermal ecology. Funct. Ecol. 28, 799–809 (2014).
Costanzo, J. P. & Lee, R. E. Jr. Avoidance and tolerance of freezing in ectothermic vertebrates. J. Exp. Biol. 216, 1961–1967 (2013).
Gunderson, A. R. & Leal, M. A conceptual framework for understanding thermal constraints on ectotherm activity with implications for predicting responses to global change. Ecol. Lett. 19, 111–120 (2016).
Kearney, M. R. & Porter, W. P. NicheMapR—an R package for biophysical modelling: The ectotherm and Dynamic Energy Budget models. Ecography 42, 1–12 (2019).
Caetano, G. H. O. et al. Time of activity is a better predictor of the distribution of a tropical lizard than pure environmental temperatures. Oikos 129, 953–963 (2020).
Sinervo, B. et al. Erosion of lizard diversity by climate change and altered thermal niches. Science 328, 894–899 (2010).
Ferguson, G. W. et al. Do Panther chameleons bask to regulate endogenous vitamin D3 production?. Physiol. Biochem. Zool. 76, 52–59 (2003).
Zhang, T. et al. A global dataset of daily near-surface air temperature at 1-km resolution (2003–2020). Earth Sys. Sci. Dat 14, 5637–5649 (2022).
Stuart, S. N. et al. Status and trends of amphibian declines and extinctions worldwide. Science 306, 1783–1786 (2004).
Buckley, L. B. & Jetz, W. Environmental and historical constraints on global patterns of amphibian richness. Proc. Royal Soc. B 274, 1167–1173 (2007).
Deutsch, C. A. et al. Impacts of climate warming on terrestrial ectotherms across latitude. Proc. Natl. Acad. Sci. 105, 6668–6672 (2008).
Roll, U. et al. The global distribution of tetrapods reveals a need for targeted reptile conservation. Nat. Ecol. Evol. 1, 1677–1682 (2017).
Mitchell, N. et al. Linking eco-energetics and eco-hydrology to select sites for the assisted colonization of Australia’s rarest reptile. Biology 2, 1–25 (2013).
Claunch, N. M. et al. Dermatomycosis caused by Paranannizziopsis australasiensis in non-native captive panther chameleons (Furcifer pardalis) captured in central Florida, USA. J. Wildl. Dis. In press https://doi.org/10.7589/JWD-D-22-00018 (2022).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D., R Core Team. nlme: Linear and nonlinear mixed effects models. R package version 3.1–152, https://CRAN.R-project.org/package=nlme (2021)
R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing. Vienna, Austria. https://www.R-project.org/ (2021)
Araújo, M. B. et al. Heat freezes niche evolution. Ecol. Lett. 16, 1206–1219 (2013).
Hoffmann, A. A., Chown, S. L. & Clusella-Trullas, S. Upper thermal limits in terrestrial ectotherms: How constrained are they?. Funct. Ecol. 27, 934–949 (2013).
Lenth, R. V. emmeans: Estimated marginal means, aka least-squares means. R package version 1.8.1–1. https://CRAN.R-project.org/package=emmeans (2022)
van’t Hoff, J. H. Etudes de dynamique chimique. 214pp (Muller, Amsterdam, Denmark, 1884)
GBIF.org (01 March 2022) GBIF Occurrence Download https://doi.org/10.15468/dl.vfcx7z
IDigBIO. http://www.idigbio.org/portal (2022), 173 records, accessed on 2022–03–18T14:39:44.552125, contributed by 15 Recordsets, Recordset identifiers: http://www.idigbio.org/portal/recordsets/7ce9b7d0-a8da-4528-bbe3-2c4f407f9cea (90 records) http://www.idigbio.org/portal/recordsets/a6eee223-cf3b-4079-8bb2-b77dad8cae9d (12 records) http://www.idigbio.org/portal/recordsets/51b958bb-9d5f-48d7-9a97-e372c0c747c3 (11 records) http://www.idigbio.org/portal/recordsets/fd9201e8-391d-4ab2-b7d4-b5dc39b0e995 (11 records) http://www.idigbio.org/portal/recordsets/cf60ed8a-2c79-4b85-a259-15a8e216dae4 (10 records) http://www.idigbio.org/portal/recordsets/bd7cfd55-bf55-46fc-878d-e6e11f574ccd (9 records) http://www.idigbio.org/portal/recordsets/b4bcc255-4acf-4966-b9b3-af9dd4e458d1 (6 records) http://www.idigbio.org/portal/recordsets/d0c73947-fce1-4914-abf0-280584f89510 (5 records) http://www.idigbio.org/portal/recordsets/029e1b92-bd6c-4037-9a0b-10136a879a74 (5 records) http://www.idigbio.org/portal/recordsets/80daac2f-e496-4c65-b196-6be7a9c4c98e (5 records) http://www.idigbio.org/portal/recordsets/5ab348ab-439a-4697-925c-d6abe0c09b92 (3 records) http://www.idigbio.org/portal/recordsets/58619649-7813-443d-9a99-3d4cfac8e0c4 (2 records) http://www.idigbio.org/portal/recordsets/2d86bdb0-a563-4a35-b990-469e9e896712 (2 records) http://www.idigbio.org/portal/recordsets/e3efdab9-2200-480c-8960-e163ee23dddf (1 records) http://www.idigbio.org/portal/recordsets/a2422e89-eee0-4bdc-a6e2-350d21e74534 (1 records)
iNaturalist community. Observations of Furcifer pardalis observed on/between 1 January 1800–29 March 2022. Exported from https://www.inaturalist.org on 29 March 2022.
Kass, J. M. et al. ENMeval 2.0: Redesigned for customizable and reproducible modeling of species’ niches and distributions. Methods Ecol. Evol. 12, 1602–1608 (2021).
Fick, S. E. & Hijmans, R. J. WorldClim 2: new 1km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 37, 4302–4315 (2017).
Phillips, S. J., Dudík, M., & Chapire, R. E. Maxent software for modeling species niches and distributions (Version 3.4.4). Available from url: http://biodiversityinformatics.amnh.org/open_source/maxent/ Accessed on 2023–1–18
Sunday, J. et al. Thermal tolerance patterns across latitude and elevation. Phil. Trans. R. Soc. B 374, 1778 (2019).
PRISM Climate Group, Oregon State University, https://prism.oregonstate.edu, accessed 17 May (2022)
Hart, E. M. and Bell, K. prism: Download data from the Oregon prism project. R package version 0.0.6. https://github.com/ropensci/prism (2015)
Hester, S. M. & Cacho, O. J. The contribution of passive surveillance to invasive species management. Biol. Invasions 19, 737–748 (2017).
Gehring, P.-S., Lutzmann, N., Furrer, S. & Sossinka, R. Habitat preferences and activity patterns of Furcifer pardalis (Cuvier, 1829) in the Masoala Rain Forest Hall of the Zurich Zoo. Salamandra 44, 129–140 (2008).
Luedeling, E. & Fernandez, E. chillR: Statistical methods for phenology analysis in temperate fruit trees. R package version 0.72.8. https://CRAN.R-project.org/package=chillR (2022).
Linvill, D. E. Calculating chilling hours and chill units from daily maximum and minimum temperature observations. Hort. Sci. 25, 14–16 (1990).
Kluever, B. M. Thermal tolerance of an invasive ectotherm occurring in Florida and Hawaii, the panther chameleon (Furcifer pardalis). Unpublished Final Report QA#3214. USDA-APHIS-WS National Wildlife Research, Fort Collins, CO (2022).
Acknowledgements
We would like to thank P. Miller, M. Sandfoss, K. Hengstebeck, D. Juárez-Sánchez, S. Nielson, S. Tillis, D. Catizone for their assistance in the field. We thank P. Miller for assistance building chameleon enclosures. We thank D. Sherman, P. Harrell, C. Hildenbrand, K. Beloyan, C. Lackey, M. Vilchez, M. Harman, S. Smit for assistance with chameleon husbandry. We thank J. Humphrey for retrieving local weather station data. Thanks to L. Baeten, M. McBride, R. Ossiboff, J.F.X. Wellehan for assistance coordinating veterinary care. The findings and conclusions in this publication have not been formally disseminated by the U.S. Department of Agriculture and should not be construed to represent any agency determination or policy.
Funding
This work was supported in part by the Department of Wildlife Ecology and Conservation, University of Florida (C.M.R.). This material is based upon work supported by the NSF Postdoctoral Research Fellowships in Biology Program under Grant No. 2109663. Salary for B. Kluever was provided by the USDA.
Author information
Authors and Affiliations
Contributions
All authors conceived the ideas and designed methodology; N.C. and C.G. collected the data; N.C., C.G., N.B. and R.G. analyzed the data; N.C. led the writing of the manuscript. All authors contributed critically to the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Claunch, N.M., Goodman, C.M., Kluever, B.M. et al. Commonly collected thermal performance data can inform species distributions in a data-limited invader. Sci Rep 13, 15880 (2023). https://doi.org/10.1038/s41598-023-43128-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-43128-4
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.