Abstract
To find the risk of time thresholds of PROM for infectious diseases of term neonates. A multi-center prospective cohort study including pregnancies with PROM at term with a single fetus were conducted. Time thresholds of the duration from PROM to delivery were examined in 2-h increments to assess the rates of infectious neonatal diseases. 7019 pregnancies were included in the study. Neonatal pneumonia and sepsis were most frequent infectious diseases in neonates born from mother with PROM at term. Rates of early-onset pneumonia varied significantly when comparing length of time of PROM greater than 16 h vs. less than 16 h (for EOP in 3 days of life, adjusted OR 1.864, 95% CI 1.159 ~ 2.997, p = 0.010; for EOP in 7 days of life, adjusted OR 1.704, 95% CI 1.104 ~ 2.628, p = 0.016). Neonates born from mother of whom the length of time from PROM to delivery ≥ 16 h were at a higher risk of acquiring EOP.
Similar content being viewed by others
Introduction
Prelabor rupture of membranes (PROM), previously known as premature rupture of membranes1, occurs in approximately 8% ~ 18% pregnancies2,3. Pregnancies with PROM were at higher risk of intrauterine infection2, their neonates were at higher risk of infectious diseases3.
To identify those neonates who are at risk of infection was essential. The signs of neonatal infection can be very subtle and difficult to differentiate from other conditions, especially during the early stages, and clinical deterioration can occur very rapidly4. Delay in initiating antibiotic treatment when it is needed may significantly increase neonatal diseases and mortality4. To determine the risk accurately is also important because prophylactic use of antibiotics might result in antibiotic treatment of many such infants who are not infected, and may lead to complications of antibiotic therapy (e.g. childhood asthma, allergy and obesity, and infant gut microbiota aberrancies)5,6,7,8,9. The current status of antibiotic usage is highly variable and often dependent on hospital preferences and personal experience even in hospitals of developed countries10.
It is controversial whether prophylactic antibiotics should be used in term neonates of mothers with PROM. Prolonged rupture of membranes is reported as a risk factor for perinatally acquired bacterial infection in term neonate4,11. Some clinicians start the usage of prophylactic antibiotics if rupture of membranes for ≥ 24 h12,13. Later, one study by Linder et al. suggested that it may be unnecessary to administer prophylactic antibiotics to term14, and this inference is only based on the result of early-onset sepsis (EOS). However, in NICE clinical guideline on antibiotics for the prevention and treatment of early-onset neonatal infection, PROM was classified as “non-red flag” as clinical indicators and risk factors for recognizing the septic neonate, and antibiotic treatment in the neonate was suggested if one “red flag” or more than one “non-red flag” risk factor or clinical indicator is present15. Although it is well known that risk of neonatal infectious diseases increases with increasing length of time of ruptured membranes16,17, time length from PROM to delivery was not considered as a factor for decision of antibiotic treatment.
However, most of the studies focused on EOS in preterm neonates or maternal infectious diseases and few studies aimed other infectious diseases in term neonates17,18,19. In Seaward’s study16, neonatal infection was assessed and time intervals were set to 24 h. In Andreas’s study, EOS was assessed and time intervals were set to 6 h. However, these studies may not accurately reflect the a priori risk of developing infectious diseases related to duration of membrane rupture until delivery.
In order to find out a more accurate time point of the increased risk of infectious neonatal diseases due to prolonged PROM, we did a secondary analysis of the cohort (MCPPNC, Multi-center Cohort of Pregnancies with PROM and their Neonates in China) and we would assess the rates of infectious neonatal diseases by bivariate and multivariable analysis using dichotomized time thresholds of length of PROM before delivery in 2-h increments.
Methods
This is a secondary analysis of the previous cohort (MCPPNC, Multi-center Cohort of Pregnancies with PROM and their Neonates in China), a prospective, multi-center cohort study aimed to describe the epidemiology of PROM and assess the influence of the implementation of the guideline3.
The definition of PROM is rupture of membranes before the on-set of labor20. PROM was confirmed by pooling and positive PROM test (PH test or insulin-like growth factor binding protein 1 detection test). Briefly, participants were recruited from patients admitted the three participating medical centers in China with a diagnosis of PROM between August 1, 2017, to March 31, 2018. All of the women with PROM were included in the study and participants at an estimated gestational age (GA) of < 24 weeks and ≥ 42 weeks were excluded. Pregnancies without PROM were eligible for the inclusion of unexposed group (non-PROM Group) if they satisfied the following conditions: the same gestational week, admission date ± 3 days and age ± 5 years compared with recruited PROM pregnancies. Maternal and neonatal data were collected until 7 days (death or hospital discharge if hospitalized for no more than 7 days). Clinical data including demographic, pregnancy history, obstetric and neonatal treatment regiments, laboratory test results and diagnosis were collected. This study was approved by the Ethical Committee of PLA Army General Hospital, China (2017-42) and assigned on the Protocol Registration and Results System of ClinicalTrials.gov (NCT03251898).
In the present study, we included pregnancies with PROM at term (estimated GA ≥ 37 weeks from MCPPNC) with a single fetus.
The rates of common neonatal infectious diseases including neonatal pneumonia, neonatal sepsis, omphalitis of newborn, neonatal urinary tract infection, congenital syphilis, neonatal conjunctivitis or dacryocystitis, necrotising enterocolitis of newborn, pyogenic abscess of the skin, congenital cytomegalovirus infection, bacterial meningitis, fungal infection of fetus or newborn, Rotavirus infection of the neonates were calculated in our study. The definition of each above diseases and fetal distress was according to the ICD 11th revision.
As early-onset pneumonia which develops within the first week of life and early-onset sepsis (develops in the first 3 days) were supposed to be result from perinatal factors. The primary outcomes of our study were set to be early-onset pneumonia (EOP) in 3 days of life, early-onset pneumonia in 7 days of life and neonatal early-onset sepsis (EOS, neonatal sepsis at < 3 days of age).
EOS was defined by the presence of clinical symptoms and a positive culture from blood or cerebrospinal fluid samples drawn within 7 days of birth21,22. Neonatal pneumonia was confirmed if meet the criteria in all three categories: (1) If there is underlying pulmonary or cardiac disease, two serial X-rays demonstrating at least one of the following: New or progressive infiltrate, consolidation, cavitation pneumoatocele. If there is no underlying pulmonary or cardiac disease, one definitive imaging test result is acceptable; (2) Worsening gas exchange. Any of the following: O2 desaturation, increased oxygen requirement, increased ventilator demand; (3) Clinical/laboratory evidence. Must have at least three of the following: Temperature instability; Leukopenia (≤ 4000 WBC/mm3) or leukocytosis (≥ 15,000 WBC/mm3) and left shift (≥ 10% band forms); New onset of purulent sputum or change in character of sputum, or increased respiratory secretions or increased suctioning requirements; Apnea, tachypnea, nasal flaring with retractions of the chest wall or nasal flaring with grunting; Wheezing, rales, or rhonchi; Cough; Bradycardia (< 100 beats/min) or tachycardia (> 170 beats/min). Early-onset pneumonia was defined as neonatal pneumonia develops within the first 7 days of life23.
The primary predictors were the length of time from PROM to birth, were examined before and after various dichotomized time thresholds by using 2-h time increments as a predictor of the 2 outcomes. For example, women delivering with a total duration of ruptured membranes of 8 h or less were compared with all those delivering with duration of ruptured membranes greater than 8 h. Similar comparisons were made for 8-, 10-, 12-, 14-, 16-, 18-, 20-, 22- and 24-h or more time thresholds to determine the threshold at which rates of each of the outcomes of interest achieved statistical significance (p < 0.05).
Education level of the pregnancies were defined as four levels: (1) Master's degree or above; (2) Bachelor’s degree; (3) Associate’s degree; (4) High school diploma or less.
If there was no indications for cesarean section, the following treatments were recommended by the guideline released in 2015 in China which was mainly referred to guidelines of the USA and Europe, and latest evidence-based medical evidences24. The GBS (Group B Streptococci) examination was by culture from vaginal or rectum swabs. For antibiotic usage according to the guideline, term pregnancies with clinical chorioamnionitis or a GBS (Group B Streptococci) positive result (no matter before or after admitted to the hospital) should receive antibiotics. If there was no GBS result or the GBS result is negative, those who had a fever of ≥ 38·0 °C or whose interval from PROM to delivery were ≥ 18 h should receive antibiotics. We defined the treatment follow the above procedure to be “using antibiotics according to the guideline” (Antibiotic). Induction of labor within 2 ~ 12 h after PROM is suggested for term pregnancies. During induction of labor with oxytocin, a sufficient period of adequate contractions (at least 12 ~ 18 h) should be allowed for the latent phase of labor to progress before diagnosing failed induction and moving to caesarean delivery. We defined the treatment follow the above procedure to be “induction of labor according to the guideline” (IL).
For umbilical care of the newborns, sterilize with 75% alcohol and keep dry for 2 times per day. For omphalitis care, the umbilical region should be cleaned with 3% hydrogen peroxide, disinfected with 95% alcohol and kept dry. For eye care, use 0.9% sodium chloride dipped in cotton swabs to wipe eye secretions for 2 times per day.
Power analysis were done using PASS 11. Data were analyzed by SAS (version 9.4). We provided descriptive statistics of obstetric and neonatal information. Continuous variables were summarized as mean (SD) or median (Q1 ~ Q3), and categorical variables were summarized as frequencies and proportions. Fisher's exact probability test and χ2 were used when appropriate for categorical variables. Multiple logistic models were conducted to assess the length of time from PROM to birth with EOP in 3 days of life, EOP in 7 days of life and EOS. We add the following potential confounding variables: the city where the hospital locates (sorted by latitude from low to high), the mother’s age, education level, induction of labor, prenatal antibiotic treatment, mode of delivery (the final way of delivery which means caesarean section or vaginal delivery), meconium-stained amniotic fluid (MSAF), the neonates’ sex, Apgar Score at 1 min (≤ 7 vs. ≥ 8).
Statement of ethics
This study was approved by the Ethical Committee of PLA Army General Hospital, China (2017-42) and assigned on the Protocol Registration and Results System of ClinicalTrials.gov (NCT03251898). All participants provided written informed consent.
Results
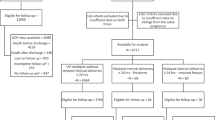
There were 7019 women who met inclusion criteria (Fig. 1). Demographic data and perinatal outcomes were showed in Table 1. The mean age of the pregnancies was 30.11 ± 4.00 and the mean gestational age was 38.81 ± 1.07 weeks. 29.25% (2053) of them were multiparous. 1634 pregnancies (23.28%) received induction of labor. The mean length of time from rupture of membrane to delivery were 20.52 ± 18.01 h. 272 (272/7019, 3.88%) fetuses were combined with fetal distress. Four fetuses (0.06%) died before birth and 7015 neonates were born. (Table 1).
Totally 4169 (59.40%) pregnancies recieved antibiotics before birth. The reasons of mothers with PROM given prenatal antibiotics including: term pregnancies with clinical chorioamnionitis or a GBS (Group B Streptococci) positive result (no matter before or after admitted to the hospital) should receive antibiotics; if there was no GBS result or the GBS result is negative, those who had a fever of ≥ 38·0 °C or whose interval from PROM to delivery were ≥ 18 h should receive antibiotics; pregnancies who received cesarean section.
The rates of common neonatal infectious diseases were calculated. Generally, neonatal pneumonia was accounted for 1.71% (120/7019) and neonatal sepsis was accounted for 0.21% (15/7019). One of the neonates suffered from omphalitis and 2 of the neonates got urinary tract infection. Three of the neonates got conjunctivitis or dacryocystitis and 1 of the neonates was infected by Rotavirus. No other infections were found in our study.
The overall incidence of EOP in 3 days of life, EOP in 7 days of life and EOS was 1.45% (102/7019), 1.71% (120/7019) and 0.19% (13/7019), respectively. The coexistence of EOS and EOP in 3 days of life was 0.11% (8/7019). For EOS and EOP in 7 days of life, the coexistense was 0.13% (9/7019). The culture results of the EOS neonates were: Staphylococcus hominis (1), Staphylococcus hominis (1), Listeria monocytogenes (2), Enterococcus faecalis (1), Klebsiella pneumonia (1), Escherichia coli (2), Staphylococcus epidermidis (4), Candida albicans (1).
Figure 2 shows the neonatal infection rates by time of PROM. Rates of the outcomes were not noted to increase with increasing of time of ruptured membranes before delivery. There was a fluctuation of infectious neonatal relevant outcomes in a long (0–24 h) interval of PROM. The rates fluctuated over time (Fig. 2).
Various dichotomized time thresholds of total length of PROM as a predictor for EOP in 3 days of life, EOP in 7 days of life and EOS were examined in the multivariable model (Tables 2, 3, 4). After adjusted with the city where the hospital locates (sorted by latitude from low to high), the mother’s age, education level, induction of labor, prenatal antibiotic treatment, mode of delivery (the final way of delivery which means caesarean section or vaginal delivery), meconium-stained amniotic fluid (MSAF), the results showed that the rates of EOP in 3 days differed significantly at a time threshold of greater than 16 h vs. less than 16 h of PROM (adjusted OR 1.864, 95% CI 1.159 ~ 2.997, p = 0.0102), and this finding continued for subsequent 2-h increment thresholds examined until 20 h. For EOP in 7 days of life, the rates was also differed significantly since the time threshold of greater than 16 hous vs. less than 16 h of PROM (adjusted OR 1.704, 95% CI 1.104 ~ 2.628, p = 0.016). The multivariable analyses showed that there was no significant difference of early-onset sepsis when comparing length of PROM at any thresholds. At the conventional 2-tailed significance level of P = 0.05, and based on the sample size, rate at different time point, the power to show the OR quantifying the association between latency period from RPOM to birth and EOP and EOS are in Tables 2, 3, and 4.
Tables 5 and 6 shows the adjusted OR of other factors at time thresholds with significant differences of the three outcomes according to length of time of PROM. For the outcome of EOP in 3 days and 7 days, the city where the hospitals locate showed great effect. Neonates whose Apgar score ≤ 7 showed to be at great risk of early onset Pneumonia.
Discussion
Our study was a real-world study on the relationship between infectious neonatal morbidity and mortality and latency period from PROM to delivery. We examined the relationship between the infectious diseases of neonates in NICU and the length of time from PROM to delivery by dichotomizing time intervals. We found the risk for EOP in 3 days of life and EOP in 7 days of life increased with increasing time of ruptured membranes since 16- hours.
There were few results on the relationship between infectious neonatal diseases and the length of time from PROM to delivery. The large International Multicenter Cohort Term PROM Study by Seaward et al.16 reported that ≥ 24 h was a risk factor of neonatal infection. A large cohort enrolled 113,568 singleton infants born at term in 2007 by Andreas et al.17 found that the risk of neonatal sepsis increased independently and nearly linearly with duration of membrane rupture up to 36 h, with an odds ratio of 1.29 for each 6-h increase in membrane rupture duration. A latest study by Shruti et al.25. reported that the rate of neonatal sepsis increases dramatically beyond 37 h of latency in term or near-term neonates (34 weeks ≤ GA ≤ 40 weeks) but the sample size was only 200. There is limited evidence of a more accurate time point of the increased risk of infectious neonatal diseases due to prolonged PROM. No evidence for EOP was reported.
In our study which enrolled pregnancies with PROM at term (estimated GA ≥ 37 weeks from MCPPNC) with a single fetus, the overall incidences of EOP and EOS in neonates werefound as 1.71% (120/7019) and 0.19% (13/7019). Within neonates admitted to NICU, early onset pneumonia in 3 days was 8/59(13.5%), early onset pneumonia in 7 days was 9/59(15.3%). It is reported acquired pneumonia was 21.3% (94/441) in neonates admitted NICU and 54.2% (totally 10.7%, 47/441) of them were early-onset pneumonia26. While in our study, within neonates admitted to NICU, early onset pneumonia in 3 days was 8/59(13.5%). The incidence of culture-proven early-onset neonatal sepsis in the United States is estimated to be 0.77 to 1 per 1,000 live births27. Among infants born at 37 weeks’ gestation or more in the United States, the rate of all-cause EOS is ~ 1.1 per 1000 LB (live birth) in black infants and ~ 0.4 per 1000 LB in nonblack infants28. In our previous study3, the rates of EOP and EOS in term neonates in PROM was significantly higher than those of non-PROM Group was significantly different. The result hints that for neonates born from mother with PROM at term, neonatal pneumonia and sepsis were main infectious diseases that need to be prevented and deal with.
For EOP in 3 days of life and EOP in 7 days of life, the city where the hospitals locate showed great effect. As in our study, different city means different hospitals, the difference may be related to the treatment strategies of different hospitals. Although there was recommendations by the Society of Obstetrics and Gynecology, Chinese Medical Association24, not all of the pregnancies were treated strictly according to the guidelines3. The difference of treatments might exist in different hospitals from different areas of our study. Other factors, such as mother’s age and education level, meconium-stained amniotic fluid, and neonate’s sex were not significant. The protective effect of induced labor is very significant. Curiously, use of antibiotics before delivery was a risk factor of EOP in 3 days of life and EOP in 7 days of life and this may because that mother who use antibiotics themselves were at higher risk of infection.
1-min Apgar Score (≤ 7) appeared to be a risk factor of early-onset pneumonia consistent with previous results. Elisha Ernest et al. reported that newborns, of any gestational age, with low 5 min Apgar scores appear to be at an increased risk for pediatric respiratory morbidity29. Sandra Costa et al. found that low Apgar score at one and five minutes was associated with neonates’ transient tachypnea, pneumonia30. Although apgar score cannot be used to decide whether resuscitation is needed, how to resuscitate and how to resuscitate, it has certain guiding significance for whether resuscitation should be continued. According to Guidelines for Neonatal Resuscitation in China (2016)31, the way of neonatal resuscitation including keeping warm, maintaining body position, clearing the airway, positive pressure ventilation and even endotracheal intubation et al. Neonatal tracheal lumen stenosis, immaturity of lung elastic fibers and other reasons make it easy for bacteria to invade the alveoli, trachea, bronchi and other parts. In addition, the introduction of exogenous microorganisms and mucosal damage during the process of clearing the airway and endotracheal intubation during resuscitation will also increase the possibility of infectious pneumonia.
Previous studies have evaluated 617, 1225, or 2416 h cutoffs of latency period from PROM to delivery for term neonates. Most of the studies focused on neonatal sepsis. In addition, those methodology may not accurately reflect the priori risk of developing infectious diseases related to duration of PROM until delivery. There were limited evidences of a more accurate time point of the increased risk of infectious neonatal diseases due to prolonged PROM. No evidence for EOP was reported.
The limitation of our study was that we did not exclude neonates with critical congenital heart disease for that underlying cardiac disease might have effects on pneumonia. Critical congenital heart disease should be taken into account in the future study.
Our data was from real-world observations. Our study was strengthened by the sample size (7019) and being conducted at 3 centers from 3 different provinces of China and “city” as a confounder was also included in multivariable analysis. All pregnancies with PROM at term from MCPPNC which included all PROM pregnancies (24 weeks ≤ estimated gestational age (GA) ≤ 42 weeks) with a single fetus to avoid selective bias. Despite the considerable sample size of our study, one of the limitations was that the number of EOS cases were not sufficient to assess the influence of length of time from PROM to birth and chorioamnionitis.
Conclusion
According to the data, the length of time from PROM to delivery ≥ 16 h is associated with an increased risk of EOP in 3 days of life and EOP in 7 days of life. The results could be a reference for antibiotic use of neonates born from mother with PROM.
Data availability
After publication, the data will be made available to others on reasonable requests to the corresponding author. A proposal with detailed description of study objectives and statistical analysis plan will be needed for evaluation of the reasonability of requests. Additional materials might also be required during the process of evaluation. Deidentified participant data will be provided after approval from the corresponding author and Seventh Medical Centre, PLA general hospital.
Abbreviations
- PROM:
-
Prelabor rupture of membranes
- EOS:
-
Early-onset sepsis
- MCPPNC:
-
Multi-center cohort of pregnancies with PROM and their neonates in China
- GA:
-
Gestational age
- C-section:
-
Caesarean section
- GBS:
-
Group B Streptococci
- IL:
-
Induction of labor according to the guideline
- MSAF:
-
Meconium-stained amniotic fluid
- BD:
-
Bachelor’s degree
- AD:
-
Associate’s degree
- HL:
-
High school or less
References
American College of O Gynecologists’ Committee on Practice B-O. Practice bulletin No. 172: Premature rupture of membranes. Obstet. Gynecol. 128, e165–e177 (2016).
Siegler, Y., Weiner, Z. & Solt, I. ACOG practice bulletin no. 217: Prelabor rupture of membranes. Obstet. Gynecol. 136, 1061 (2020).
Zhuang, L. et al. The correlation between prelabour rupture of the membranes and neonatal infectious diseases, and the evaluation of guideline implementation in China: A multi-centre prospective cohort study. Lancet Reg. Health West. Pac. 3, 100029 (2020).
Ungerer, R. L., Lincetto, O., McGuire, W., Saloojee, H. & Gulmezoglu, A. M. Prophylactic versus selective antibiotics for term newborn infants of mothers with risk factors for neonatal infection. Cochrane Database Syst. Rev. 2004, 003957 (2004).
Uzan-Yulzari, A. et al. Neonatal antibiotic exposure impairs child growth during the first six years of life by perturbing intestinal microbial colonization. Nat. Commun. 12, 443 (2021).
Penders, J., Kummeling, I. & Thijs, C. Infant antibiotic use and wheeze and asthma risk: A systematic review and meta-analysis. Eur. Respir. J. 38, 295–302 (2011).
Penders, J. et al. Gut microbiota composition and development of atopic manifestations in infancy: The KOALA Birth Cohort Study. Gut 56, 661–667 (2007).
Polin, R. A. Management of neonates with suspected or proven early-onset bacterial sepsis. Pediatrics 129, 1006–1015 (2012).
Salvatore, S. et al. Neonatal antibiotics and prematurity are associated with an increased risk of functional gastrointestinal disorders in the first year of life. J. Pediatr. 212, 44–51 (2019).
Villanueva, P., Freyne, B., Hickey, L., Carr, J. & Bryant, P. A. Impact of an antimicrobial stewardship intervention in neonatal intensive care: Recommendations and implementation. J. Paediatr. Child Health. 57, 1208 (2021).
Sorsa, A. Epidemiology of neonatal sepsis and associated factors implicated: Observational study at neonatal intensive care unit of Arsi University Teaching and Referral Hospital, South East Ethiopia. Ethiop. J. Health Sci. 29, 333–342 (2019).
Wolf, R. L. & Olinsky, A. Prolonged rupture of fetal membranes and neonatal infections. S. Afr. Med. J. 50, 574–576 (1976).
St Geme, J. W. Jr. et al. Perinatal bacterial infection after prolonged rupture of amniotic membranes: An analysis of risk and management. J. Pediatr. 104, 608–613 (1984).
Linder, N. et al. Neonatal sepsis after prolonged premature rupture of membranes. J. Perinatol. 15, 36–38 (1995).
Caffrey Osvald, E. & Prentice, P. NICE clinical guideline: Antibiotics for the prevention and treatment of early-onset neonatal infection. Arch. Dis. Child Educ. Pract. Ed. 99, 98–100 (2014).
Seaward, P. G. et al. International multicenter term PROM study: Evaluation of predictors of neonatal infection in infants born to patients with premature rupture of membranes at term Premature Rupture of the Membranes. Am. J. Obstet. Gynecol. 179, 635–639 (1998).
Herbst, A. & Kallen, K. Time between membrane rupture and delivery and septicemia in term neonates. Obstet. Gynecol. 110, 612–618 (2007).
Aziz, N., Cheng, Y. W. & Caughey, A. B. Factors and outcomes associated with longer latency in preterm premature rupture of membranes. J. Matern. Fetal Neonatal. Med. 21, 821–825 (2008).
Drassinower, D., Friedman, A. M., Obican, S. G., Levin, H. & Gyamfi-Bannerman, C. Prolonged latency of preterm premature rupture of membranes and risk of neonatal sepsis. Am. J. Obstet. Gynecol. 214(743), e1-6 (2016).
Committee on Practice B-O. ACOG Practice bulletin No. 188: Prelabor rupture of membranes. Obstet. Gynecol. 131, e1–e14 (2018).
Giannoni, E. et al. Neonatal sepsis of early onset, and hospital-acquired and community-acquired late onset: A prospective population-based cohort study. J. Pediatr. 201(106–14), e4 (2018).
Jiang, S. et al. Early-onset sepsis among preterm neonates in China, 2015 to 2018. Pediatr. Infect. Dis. J. 38, 1236–1241 (2019).
Hooven, T. A. & Polin, R. A. Pneumonia. Semin. Fetal Neonatal Med. 22, 206–213 (2017).
The Society of Obstetrics and Gynecology CMA. Guidelines for diagnosis and management of premature rupture of membranes. Chin. J. Obstet. Gynecol. 50, 3–7 (2015).
Gupta, S., Malik, S. & Gupta, S. Neonatal complications in women with premature rupture of membranes (PROM) at term and near term and its correlation with time lapsed since PROM to delivery. Trop. Doct. 50, 8–11 (2020).
Langer, M., Cigada, M., Mandelli, M., Mosconi, P. & Tognoni, G. Early onset pneumonia: A multicenter study in intensive care units. Intensive Care Med. 13, 342–346 (1987).
Simonsen, K. A., Anderson-Berry, A. L., Delair, S. F. & Davies, H. D. Early-onset neonatal sepsis. Clin. Microbiol. Rev. 27, 21–47 (2014).
Schrag, S. J. et al. Epidemiology of invasive early-onset neonatal sepsis 2005 to 2014. Pediatrics 138, 1–10 (2016).
Ernest, E. et al. Apgar score and long-term respiratory morbidity of the offspring: A population-based cohort study with up to 18 years of follow-up. Eur. J. Pediatr. 178, 403–411 (2019).
Costa, S., Rocha, G., Leitao, A. & Guimaraes, H. Transient tachypnea of the newborn and congenital pneumonia: A comparative study. J. Matern Fetal Neonatal Med. 25, 992–994 (2012).
Group CNRPE. Guidelines for neonatal resuscitation in China. Chin. J. Perinatal Med. 19, 481–486 (2016).
Funding
This study was founded by National Natural Science Foundation of China (81170602) and Capital Medical Development Research Fund of Beijing (20053044).
Author information
Authors and Affiliations
Contributions
F.-Z.C., X.L., S.-D.H. and L.Z. designed the study. L.Z., Z.-K.L., Y.-F.Z. and R.J. wrote the manuscript. Z.-K.L., Y.-F.Z., R.J., C.-Z.Y., Y.-P.Z., L.L., T.L., Y.Y., W.Z., J.C., X.-Y.C., J.-Y.P. recruited and followed up the participants and collected the data. L.Z., X.-Y.C. and J.-Y.P. did the statistical analyses. All authors contributed to review and revision, and have seen and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhuang, L., Li, ZK., Zhu, YF. et al. Latency period of PROM at term and the risk of neonatal infectious diseases. Sci Rep 12, 12275 (2022). https://doi.org/10.1038/s41598-022-16593-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-16593-6
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.