Abstract
The DISC1 gene is one of the most relevant susceptibility genes for psychosis. However, the complex genetic landscape of this locus, which includes protective and risk variants in interaction, may have hindered consistent conclusions on how DISC1 contributes to schizophrenia (SZ) liability. Analysis from haplotype approaches and brain-based phenotypes can contribute to understanding DISC1 role in the neurobiology of this disorder. We assessed the brain correlates of DISC1 haplotypes associated with SZ through a functional neuroimaging genetics approach. First, we tested the association of two DISC1 haplotypes, the HEP1 (rs6675281-1000731-rs999710) and the HEP3 (rs151229-rs3738401), with the risk for SZ in a sample of 138 healthy subjects (HS) and 238 patients. This approach allowed the identification of three haplotypes associated with SZ (HEP1-CTG, HEP3-GA and HEP3-AA). Second, we explored whether these haplotypes exerted differential effects on n-back associated brain activity in a subsample of 70 HS compared to 70 patients (diagnosis × haplotype interaction effect). These analyses evidenced that HEP3-GA and HEP3-AA modulated working memory functional response conditional to the health/disease status in the cuneus, precuneus, middle cingulate cortex and the ventrolateral and dorsolateral prefrontal cortices. Our results are the first to show a diagnosis-based effect of DISC1 haplotypes on working memory-related brain activity, emphasising its role in SZ.
Similar content being viewed by others
Introduction
The Disrupted in Schizophrenia 1 gene (DISC1) was first recognised in the context of psychiatric illness when a balanced chromosomal translocation (1;11)(q42.1;q14.3) was found to segregate with major mental disorders, including schizophrenia (SZ)1. Since then, molecular investigations have highlighted that the liability of the DISC1 gene towards psychosis is mediated by the protein role in processes associated with the pathophysiology of SZ, such as neurodevelopment and neurosignalling2,3. In neurodevelopment, the DISC1 protein acts as a central coordinator of neuronal trafficking, enabling the proper delivery of a range of neuronal cargoes with spatial and temporal precision, thereby ensuring normal neuronal development and functional homeostasis4. More specifically, DISC1 is involved in many stages of neurogenesis, such as neural precursor proliferation, neuronal migration, and neuronal integration/maturation5. Also, the synaptic location of DISC1 in adult dendritic spines and its enrichment in the post-synaptic density have suggested a role in the functional regulation of synaptic plasticity4, which is supported by several studies that show synaptic plasticity impairments in a variety of different DISC1 mouse mutant models6. Finally, it is worth mentioning that DISC1 protein interacts directly with the dopamine D2 receptor7, the main target of antipsychotic medications, suggesting that functional changes in the DISC1 sequence could interfere with dopamine signalling and antipsychotic drug response. Overall, these data indicate that any factor that compromises normal DISC1 function will likely impact brain development and create neurosignalling deficits5.
After identifying the DISC1 translocation, numerous genetic association studies and meta-analyses have also provided support for the role of Single Nucleotide Polymorphisms (SNPs) and mutations at this gene in the risk for SZ8,9,10,11, as well as in other mental disorders and psychosis-related traits12,13,14. Additionally, recent data has shown lower DISC1 expression levels in patients with SZ, which, in turn, were associated with increased severity of symptoms15. Therefore, the DISC1 gene is currently considered one of the most relevant susceptibility genes for psychosis16. However, while many genes identified through Genome-Wide Association Studies (GWAS) in SZ form part of the DISC1 regulome and interactome17,18, this gene has never been identified through genome-wide approaches by itself19,20,21. This lack of direct GWAS significance may be due to the DISC1 genetic structure, which is complex and includes protective and risk single-SNP variants11,14. Efforts to characterise such complexity have identified epistatic effects among DISC1 polymorphisms on the susceptibility towards SZ14,22, bipolar disorder, psychosis-related traits, and emotional liability12,14,22,23. As well, from haplotype-based approaches, the combined effect of different alleles has been related to risk and protective effects towards SZ and schizoaffective disorders13,24,25,26,27,28, confirming the interest in analysing the DISC1 variability considering its haplotypic structure. In this sense, among the haplotypes more robustly associated with psychosis are the so-called HEP3 haplotype (spanning at intron 1/exon 2), which includes the rs751229 and the rs3738401, and the HEP1 haplotype (spanning at exon 9/intron 9), which includes SNPs such as the rs6675281, the rs1000731 and the rs99971013,26,27,28.
To better comprehend this complex genetic landscape and how DISC1 contributes to SZ, strategies based on quantifiable brain-derived phenotypes have been proposed29,30. On the one hand, many studies have reported associations between different DISC1 SNPs and brain structural variations in adults and neonates31,32, in line with the pivotal role of DISC1 in neurodevelopment. On the other hand, DISC1 variability has been found to affect cognitive performance in domains such as attention and working memory33,34,35, and the brain functional response to executive function and memory tasks36,37,38,39,40. From functional magnetic resonance imaging (fMRI) studies, among the regions where DISC1 functional effects have been typically described, we can highlight the hippocampal region39,40 and the prefrontal cortex38,41,42,43. These areas, indeed, have been critically involved in both the functional response to memory and attention tasks44,45, and SZ itself46,47,48. Considering all the above mentioned together with GWAS data showing that several genomic regions associated with SZ have been related to working memory18, this cognitive domain is a recognised intermediate phenotype to study SZ's neurobiological basis. Accordingly, studying the DISC1 correlates of working memory at a brain level through fMRI might shed light on the disorder's brain functional changes.
Compared with the amount of research based on single polymorphic DISC1 variants (reviewed Johnstone et al. 2011 and Duff et al. 201332,49), the research based on DISC1 haplotypic variability is scarce. To the best of our knowledge, only two studies have related DISC1 haplotypes to cortical grey matter reductions in healthy subjects and patients with SZ27,43. Similarly, its haplotypic variability was associated with short- and long-term memory impairments27. Nonetheless, there is no data on the role of DISC1 haplotypes on fMRI brain phenotypes which may help bridge the gap between the previously detected effects at the brain structural and cognitive level and the altered neurobiological basis in patients.
Therefore, in this study, we aimed to investigate the brain activity correlates of DISC1 haplotypic variants associated with SZ through a neuroimaging (fMRI) genetics study. First, we conducted a case–control approach to identify the haplotypes associated with SZ in our sample. Then, we explored whether these haplotypes exerted their effect by differentially modulating working memory cognitive processes during the performance of the n-back task depending on health/disease status.
Results
Genetic association study
First, in our sample of 138 healthy subjects (HS) and 238 subjects with a diagnosis of SZ, our analysis at DISC1 haplotypic level revealed three haplotypes associated with the risk for the disorder. The haplotypes HEP1-CTG and HEP3-GA were more frequent in HS than in patients, while the HEP3-AA haplotype was significantly overrepresented in patients (Table 1).
Neuroimaging association study
Based on the DISC1 haplotypes associated with SZ, we performed the neuroimaging analysis with the haplotypes HEP1-CTG, HEP3-GA and HEP3-AA in a subsample of 70 HS and 70 patients (groups matched for age, sex and estimated IQ). The haplotypes were dichotomised, and each subject was defined as a carrier of 0 or 1/2 copies of the protective/risk haplotypes. Subsequently, the diagnosis x haplotype interactions were tested on n-back functional response and behavioural performance.
N-back functional response
While the haplotype × diagnosis status interactions were assessed in all the n-back contrasts (1-back vs baseline, 2-back vs baseline and 2-back vs 1-back), we focused on the 2-back vs baseline and 2-back vs 1-back findings because these contrasts are the ones better depicting working memory networks50.
The HEP1-CTG × diagnosis interaction revealed no significant results. Concerning the HEP3, we found that both haplotypic combinations interacted with diagnosis and modulated n-back functional response. In the case of the HEP3-GA haplotype, the interaction was significant in the 1-back vs baseline (fully described in Supplementary Information and Supplementary Fig. S1) and the 2-back vs baseline contrasts.
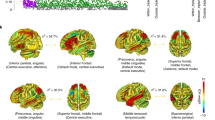
As regards the 2-back vs baseline contrast, one significant cluster of interaction was seen, involving the cuneus and precuneus medially and the right middle cingulate cortex and the superior parietal cortex (735 voxels, peak activation at Montreal Neurological Institute coordinates system (MNI) [-4,-66,72], Z = 3.2, p = 0.0182). For interpretation of the direction of the interaction results, the mean activation scores were estimated from the areas where significance was detected, and the mean values were plotted. The mean activations of the region of interest (ROI) indicated that the patients with SZ carrying no copies of the protective HEP3-GA exhibited higher activation scores than those with 1or 2 copies. In contrast, the HS showed the opposite pattern (Fig. 1).
(a) Brain regions showing the axial view of the cluster with significant diagnosis × HEP3-GA interaction in 2-back vs baseline contrast. The right side of the image represents the right side of the brain. The MNI coordinates are given for each slice. Units of the bar are the corresponding β values from the regression standardised to Z-scores. (b) Plot with the corresponding estimated marginal mean activity scores and ± 2 standard error (SE) for HEP3-GA haplotype in healthy subjects (42.90% with 0 copies and 57.10% with 1 or 2 (1/2) copies) and patients with SZ (51.40% with 0 copies and 48.60% with 1or 2 (1/2) copies).
When the HEP3-AA haplotype was assessed, we found a significant interaction with the diagnosis in all the analysed contrasts (for 1-back vs baseline contrast results, see Supplementary Information and Supplementary Fig. S2). In the 2-back vs baseline, the diagnosis and HEP3-AA interaction was significant in three clusters: cluster (1) was in the left middle and posterior cingulate cortex, extending to the cuneus, precuneus, the thalamus and the paracentral lobule (850 voxels, peak activation at MNI [-22,-40,28], Z = 4.10, p = 0.008); cluster (2) was in the right hemisphere including the postcentral and supramarginal gyrus, the middle cingulate cortex, the paracentral lobule and also reaching, the hippocampus (930 voxels, peak activation at MNI [38,-4,28], Z = 3.70, p = 0.00464); and, cluster (3) involved regions of the lingual and fusiform gyri on the left, the calcarine sulcus and the cerebellum (1348 voxels, peak activation at MNI [-28,-72,10], Z = 4.06, p = 0.000333). In this contrast, ROI analysis revealed that for the three clusters, the HS and the patients with SZ showed similar activity profiles when they had no copies of the HEP3-AA risk haplotype. Conversely, among individuals with 1 or 2 copies of the risk haplotype, HS showed increased activation than patients with SZ, who deactivated these regions. The mean activation scores for cluster 2 are shown in Fig. 2.
(a) Brain regions showing the axial view of the clusters with significant diagnosis × HEP3-AA interaction in 2-back vs baseline contrast. Cluster one is shown in red, cluster two in blue and cluster three in yellow. The right side of the image represents the right side of the brain. The MNI coordinates are given for each slice. Units of the bar are the corresponding β values from the regression standardised to Z-scores. (b) Plot corresponding to the 2nd cluster's estimated marginal mean activity scores ± 2 standard error (SE) for the HEP3-AA haplotype in healthy subjects (92.9% with 0 copies and 7.10% with 1 or 2 (1/2) copies) and patients with SZ (74.30% with 0 copies and 25.70% with 1 or 2 (1/2) copies).
In the 2-back vs 1-back contrast, a significant interaction emerged in one cluster located in the right superior and middle frontal cortex, the middle and inferior orbitofrontal cortex and the dorsolateral and ventrolateral prefrontal cortices (585 voxels, peak activation at MNI [48,44,-16], Z = 3.83, p = 0.0441). Within individuals with no copies of the risk HEP3-AA haplotype, there were barely any differences between the HS and the SZ patients. However, among individuals with 1 or 2 copies of the HEP3-AA risk haplotype, the response was in opposite directions depending on the diagnosis: HS with 1 or 2 risk copies responded with an activity increase, whereas patients showed minimal changes (Fig. 3).
(a) Brain regions showing the axial view of the cluster with significant diagnosis × HEP3-AA interaction in 2-back vs 1-back contrast. The right side of the image represents the right side of the brain. The MNI coordinates are given for each slice. Units of the bar are the corresponding β values from the regression standardised to Z-scores. (b) Plot corresponding to the estimated marginal mean activity scores ± 2 standard error (SE) for the HEP3-AA haplotype copies in healthy subjects (92.9% with 0 copies and 7.10% with 1 or 2 (1/2) copies) and patients with SZ (74.30% with 0 copies and 25.70% with 1 or 2 (1/2) copies).
N-back behavioural performance
The signal detection theory index of sensitivity (d') was the behavioural measure used (d'1 for 1-back and d'2 for 2-back). Higher values of d' indicate a better execution of the task. The comparison between HS and patients with SZ revealed significant differences at both n-back difficulty levels (d'1: F = 12.85, p < 0.001, d'2: F = 37.31, p < 0.001). Patients' performance was significantly worse than the HS' one, and the differences were more pronounced for d'2 scores (d'1 estimated marginal mean (SE) for HS: 4.24 (0.11) and for patients: 3.71 (0.11); d'2 estimated marginal mean (SE) in HS: 3.30 (0.09) and patients: 2.48 (0.09)). The interaction between the associated haplotypes and diagnosis revealed no significant results.
Discussion
This study explored whether DISC1 haplotypic variability exerted differential effects on working memory-related brain activity. We evidenced the association of three DISC1 haplotypes with SZ (HEP1-CTG, HEP3-GA and HEP3-AA) and subsequently the modulating role of HEP3-GA and HEP3-AA on brain activations during the performance of the n-back task depending on the health/disease status.
Our genetic association results add to previous research on the involvement of DISC1 haplotypic variability in the risk for SZ and other psychotic disorders13,25,27,28,51. On the one hand, our data revealed that the HEP1-CTG (rs6675281, rs1000731 and rs999710) was associated with a protective effect (i.e., less frequent in patients than in HS). In line with these data, a HEP1 haplotype containing rs6675281-C and rs1000731-T alleles was identified to be underrepresented in patients with a schizoaffective disorder through a case–control study28. Contrary, from a family-based approach, the opposite combination (rs6675281-T allele and rs1000731-C allele) was over-transmitted to the patients with SZ27. On the other hand, our findings also indicated the protective effect of the HEP3-GA (rs751229, rs3738401) and the risk effect of the HEP3-AA (i.e., more frequent in patients as compared to HS). In this view, previous studies have likewise reported HEP3-AA to be more frequent in patients with a psychotic disorder than in their relatives13,27. Nonetheless, it is of note that the allelic variants conforming HEP1 and HEP3 and the relative frequencies observed in patients and HS are not always in consensus across studies25,51. Such divergencies could be due to the sample origin (closely related to the haplotypic structure), the association designs, and the differences in the diagnostic criteria at inclusion. Still, what became evident from a study aiming to retrieve consistent results on how DISC1 variability contributes to SZ's liability was that the locus contains both risk and protective SNPs and haplotypes14.
Our genetic association analyses identified the haplotypic combinations related to SZ in our sample, leading to the assessment of their influence on brain functional differences in HS and patients with SZ. Through the fMRI analyses, we found no significant interaction between diagnosis and HEP1-CTG on n-back brain function. Given the scarce previous studies, a direct comparison of our results with others is not possible. However, it can be noted that one study reported changes in cortical thickness in the left supramarginal gyrus associated with a rare DISC1 haplotype containing the rs6675281-C allele43.
With reference to HEP3, the two haplotypic combinations revealed significant interactions with the diagnosis on n-back brain response. The interaction between the HEP3-GA and diagnosis in both 1-back vs baseline and 2-back vs baseline implicated the cuneus, the precuneus and the middle cingulate cortex. We observed that HS deactivated such regions and that patients with no copies of the protective GA haplotype had a less marked deactivation or even failed to deactivate in the most difficult level (2-back vs baseline). Considering that our association findings related the GA haplotype to a protective effect towards the disorder, this neuroimaging result seems to be in the same direction. The precuneus forms part of the so-called default mode network, a network of regions that HS deactivate during the performance of a wide range of cognitive tasks52 and its failure of deactivation during the performance of the n-back and other tasks has also been reported in several studies in SZ53. Additionally, changes in the precuneus structure and functional connectivity in SZ have been previously related to DISC1 genetic variability54.
As regards the HEP3-AA, the interaction has been observed in all the n-back contrasts analysed. This suggests that the HEP3-AA haplotype modulates the different cognitive requirements engaged during the n-back50. Concerning the 2-back vs baseline contrast, the interaction was found in regions related to the previously described HEP3-GA interaction, comprising the precuneus, the posterior and middle cingulate cortex and the cuneus. This suggests that, regardless of the haplotypic combination, the HEP3 haplotype may be involved in the functional response of these brain regions. In detail, we observed that among patients with SZ, those without the protective GA haplotype and those with the risk AA haplotype were the ones presenting activation patterns in opposite directions compared to the rest of the individuals. Since the haplotypes were dichotomised, eight individuals had 1 copy of each of the two haplotypes (1 of the protective HEP3-GA and the other of the risk HEP3-AA). To overcome this haplotypic overlap, we retested the interactions with the estimated mean activity scores once these subjects were removed from the analyses, and the results remained unchanged.
In the 2-back vs 1-back contrast, the regions with significant HEP3-AA interaction with diagnosis included the right ventrolateral and the dorsolateral prefrontal cortices. Previously, DISC1 variability has been found to modulate the dorsolateral prefrontal cortex activation in response to working memory in healthy subjects55. Likewise, a functional neuroimaging meta-analysis of different executive and working memory tasks found that the dorsolateral prefrontal cortex bilaterally and the right ventrolateral and premotor cortex were involved in these cognitive demanding tasks and also that their activation was reduced in SZ48. Considering the HEP3-AA neuroimaging results together, the most distinctive pattern occurred within HS carrying 1 or 2 copies of this risk haplotype compared to the others (HS without it and all the patients). This pattern arises from the larger absolute degree of brain activity change observed between HS carriers and non-carriers of the risk haplotype, compared to the degree of change detected within patients. Such differential effect of diagnosis has already been highlighted by Crespi & Badcock56 when reviewing the complex relationship between genetic factors and SZ intermediate phenotypes.
About the n-back behavioural analyses, we did not detect significant interaction effects between the diagnosis and either of the haplotypes. In this sense, the comparability of the results is hampered because previous studies assessing DISC1 variability on working memory do not report DISC1 behavioural analyses evaluated during fMRI protocols37,38. However, one fMRI study is partially in line with our data, as they did not detect an effect of the DISC1 on behavioural performance when analysing one SNP at HEP1 (rs6675281) and a different working memory task43. Beyond functional studies, neurocognitive evidence has associated a rare 4-SNP haplotype (including the HEP3) with visuospatial working memory27. Then, the results in our sample could be interpreted from the perspective that the genetic variability effect at the behavioural level is less penetrant than at the brain activity level57, and further analyses in larger samples will be needed to furtherly explore the relationship between fMRI and behavioural data.
Regarding the effects of HEP3 haplotype on gene expression, it has been highlighted that the regions covered by this haplotype are highly conserved after human and mouse divergence, and the fact that these noncoding regions have such evolutionary conservation may be indicative of some functional significance and/or a potential regulatory role25. Furthermore, the rs3738401-G/A polymorphism, located in exon 2, is a missense variant that causes an Arg264Gln aminoacidic substitution. It has been reported that this polymorphism has a biological impact on Wnt signalling transduction pathways affecting neurogenesis58, suggesting a putative mechanism for its role in decisive neurodevelopmental processes leading to psychiatric disorders. So, our results on the modulation effect that DISC1 haplotypic variability has on brain function would link the evidence highlighting the role of DISC1 in neurogenesis with the pathophysiological mechanisms underlying SZ.
Finally, some limitations of the current study need to be considered. First, for the genetic association analysis, our sample could be regarded as quite small; nonetheless, the fact that we inspected the haplotypic instead of single SNP variability adds power to our approach. Also, with 70 patients and 70 controls, our sample is large for functional imaging standards considering that most of the previous studies are focused exclusively on HS37,38,39,40,41 or include a reduced group of patients39,59. On the other hand, the fact that the neuroimaging analyses were based on our haplotypic association results represents a strength of the study. Notwithstanding, future studies performed in larger samples and higher resolution scanners would be desirable. Finally, we must consider that variables related exclusively to the illness status could not be included in the interaction analysis. With this in mind, we checked within patients the possible impact of PANSS score or medication on the mean activity and the d' scores through regressions, with none of them reaching significance.
In conclusion, our data add to previous findings of an association of the HEP1-CTG, HEP3-GA and HEP3-AA haplotypes with SZ susceptibility. Additionally, this study shows, for the first time, evidence of the effect of DISC1 haplotypic variability on brain functional differences between patients affected by SZ and HS. Although further studies are needed, our data suggest a putative role of the DISC1 gene in the altered functional and behavioural substrates of SZ associated with n-back task performance. This might, in turn, contribute to closing the gap between the role of this gene in neurodevelopment and the pathophysiological underpinnings of schizophrenia.
Methods
Sample
The genetic association analysis to identify DISC1 haplotypes related to SZ was conducted in a sample of 138 healthy subjects (HS) and 238 subjects with a DMS-IV-TR diagnosis of SZ (based on interviews by two psychiatrists). All participants were of European ancestry, between 19 and 65 years old. There were group differences regarding sex (χ2 = 15.85 p < 0.001, 72% males within patients with SZ and 51% within HS) and age (t = − 2.65 p = 0.008, mean age (SD) for patients with SZ = 41.98 (11.81) and for HS = 38.65 (11.64)). The HS had no personal or family history of psychiatric disorders or treatment. All participants met the same exclusion criteria: co-existent neurological disorder or medical illness affecting brain function, history of head trauma with loss of consciousness and history of drug abuse or dependence.
The neuroimaging analyses were performed in a subsample of 70 HS and 70 patients matched for age, sex, and estimated IQ (premorbid IQ in the patients), as assessed using the Word Accentuation Test (Test de acentuación de palabras, TAP60) (Table 2). In addition to the previous inclusion criteria, all participants in this part of the study were right-handed and had an estimated IQ ≥ 70. Symptoms were evaluated with the Positive and Negative Symptoms Scale (PANSS61,62).
Ethical approval was obtained from the Germanes Hospitalàries Research Ethics Committee, and all participants provided written informed consent about the study procedures and implications. All procedures were carried out according to the Declaration of Helsinki.
Genotyping and haplotype estimation
Genomic DNA was extracted for all individuals either from buccal mucosa through cotton swabs using ATP Genomic Mini Kit Tissue (Taknokroma Analitica, S.A., Sant Cugat del Vallès, Span) or peripheral blood cells using Realpure SSS kit (Durviz, S.L.U., Valencia, Spain). The set of SNPs was selected according to previous studies in which DISC1 haplotypes associated with SZ were described25,28. Two SNPs within the HEP3 haplotype (rs751229 and rs3738401) and three SNPs within the HEP1 haplotype (rs6675281, rs1000731 and rs999710) were genotyped (Table 3). The allelic discrimination was performed using a fluorescence-based procedure (Applied Biosystems Taqman 5 ‘-exonuclease assays) using standard conditions, and the polymerase chain reaction plates were read on ABI PRISM 7900HT instrument with SDS v2.1 software (Applied Biosystems). The genotyping call rate was > 0.97, and the method's accuracy was retested by running in duplicate 10% of the samples and confirming all the repeated genotypes. All SNPs were in Hardy–Weinberg equilibrium in both diagnostic groups. The minor allele frequencies in our sample were similar to that described for the European population in the 1000 Genomes Project. There were no differences between the SNPs/haplotype frequencies from the whole sample and the neuroimaging subsample. For the neuroimaging approach, the estimation and tabulation of the individual haplotype phases were performed using PLINK 1.0763.
N-back task description and behavioural response
Functional magnetic resonance images (fMRI) were obtained while participants performed a sequential-letter version of the n-back task64. This functional paradigm engages storage and executive processes related to attention and memory65. The task had two levels of memory load (the 1-back and the 2-back), and as the difficulty load increases, higher-order executive functions like working memory become more relevant66,67. Since working memory is a cognitive dimension where patients affected by SZ exhibit affectations48,68,69,70,71, we focused on the contrasts better characterising the working memory network, which, according to recent independent component analysis, are the 2-back vs baseline and the 2-back vs 1-back contrasts50.
The two memory load levels were presented in a blocked design manner. Each block consisted of 24 letters that were shown every 2 seconds (1 second on, 1 second off). All blocks contained five repetitions (one letter beforehand in the 1-back version and two letters beforehand in the 2-back version) located randomly within the blocks. Individuals had to indicate repetitions by pressing a button. Four 1-back and four 2-back blocks were presented in an interleaved way, and between them, a baseline stimulus (an asterisk flashing with the same frequency as the letters) was presented for 16 seconds. Characters were shown in green and red for 1-back and 2-back, respectively, to identify which task had to be performed. The same day, before the scanning session, all participants underwent a training session outside the scanner.
The behavioural measure used was the signal detection theory index of sensitivity, d'72. Higher values of d' indicate a better ability to discriminate between targets and distractors, while negative values indicate that subjects are not performing the task. All the individuals included in the analyses had positive d' values (d'1 for 1-back and d'2 for 2-back).
Neuroimaging data acquisition
In each scanning session, 266 volumes were acquired from a GE Sigma 1.5-T scanner (General Electric Medical Systems, Milwaukee, Wisconsin, USA). A gradient echo-planar imaging sequence depicted the blood oxygen level-dependent signal. Each volume contained 16 axial planes acquired with the following parameters: repetition time = 2000 ms., echo time = 20 ms., flip angle = 70°, section thickness = 7 mm, section skip = 0.7 mm, in-plane resolution = 3 × 3 mm. To avoid T1 saturation effects, the first 10 volumes were discarded.
Statistical analyses
Genetic association study
We tested all the possible allelic combinations for the two haplotypes assessed (HEP1 and HEP3) for association with SZ through a logistic regression model, including sex as a covariate (PLINK). The given p values are those obtained after 10,000 permutations procedure. Only those haplotypes significantly associated with the disorder were furtherly examined in the neuroimaging association study.
Neuroimaging association study
Based on our genetic association results, we performed the neuroimaging analysis with the HEP1-CTG, the HEP3-GA and the HEP3-AA in the matched subsample of 70 HS and 70 patients. Because of the haplotypic frequencies in our sample, the analyses were conducted considering all haplotypes as dichotomous variables and each subject was defined as a carrier of 0 or 1/2 copies of the protective/risk haplotypes.
The fMRI analyses were performed with the FEAT tool from FSL software (FMRIB Software, University of Oxford, Oxford, UK72). Images were corrected for movement and co-registered to a common stereotactic space (the Montreal Neurological Institute (MNI) template). Subjects with an estimated maximum absolute movement > 3.0 mm or an average absolute movement > 0.3 mm were a priori excluded from the study to minimise unwanted movement-related effects. Normalised volumes were spatially smoothed using a Gaussian filter of 5 mm full-width at half maximum, and general linear models were fitted to generate individual activation maps for three different contrasts: 1-back vs baseline, 2-back vs baseline, and 2-back vs 1-back. The movement variables were added to the model as nuisance variables to control for movement in the scanner. All statistical tests were performed at the cluster level with a corrected p value of 0.05 and an initial height threshold of 2.3 (equivalent to an uncorrected p value of 0.01, using the Standard Field Theory correction implemented in FSL73). Afterwards, the interaction effect on brain function between the diagnosis and the three haplotypes was tested using regression models (whole-brain corrected and controlled for age, sex and estimated IQ). For interpretation of the direction of the interaction results, the mean activation scores were estimated from the areas where significance was detected with the FSLSTATS tool in FSL, and the mean values were plotted using SPSS (IBM SPSS Statistics, version 23.0, released 2015, IBM Corporation, Armonk, New York). The mean activity scores obtained from the 2-back vs 1-back contrast do not represent the mean activation per se, but the mean activation change occurring from 1-back to 2-back levels.
Analyses of the behavioural data were carried out using SPSS. First, n-back task performance (d'1 and d'2) was compared between HS and patients using an ANOVA (controlling for age, sex and estimated IQ). Next, the interaction between diagnosis and the three haplotypes was tested through full-factorial ANOVAs (including the diagnosis and haplotype main effects and controlled for age, sex and estimated IQ). These analyses were corrected for multiple comparisons (Bonferroni).
References
St Clair, D. et al. Association within a family of a balanced autosomal translocation with major mental illness. Lancet 336, 13–16 (1990).
Howes, O. D., McCutcheon, R., Owen, M. J. & Murray, R. M. The role of genes, stress, and dopamine in the development of schizophrenia. Biol. Psychiatr. 81, 9–20 (2017).
Eltokhi, A., Santuy, A., Merchan-Perez, A. & Sprengel, R. Glutamatergic dysfunction and synaptic ultrastructural alterations in schizophrenia and autism spectrum disorder: Evidence from human and rodent studies. Int. J. Mol. Sci. 22, 1–26 (2021).
Tomoda, T., Hikida, T. & Sakurai, T. Role of DISC1 in neuronal trafficking and its implication in neuropsychiatric manifestation and neurotherapeutics. Neurotherapeutics 14, 623–629 (2017).
Thomson, P. A. et al. DISC1 genetics, biology and psychiatric illness. Front. Biol. 8, 1–31 (2013).
Tropea, D., Hardingham, N., Millar, K. & Fox, K. Mechanisms underlying the role of DISC1 in synaptic plasticity. J. Physiol. 596, 2747–2771 (2018).
Su, P. et al. A dopamine D2 receptor-DISC1 protein complex may contribute to antipsychotic-like effects. Neuron 84, 1302–1316 (2014).
Ma, J. H. et al. Association on DISC1 SNPs with schizophrenia risk: A meta-analysis. Psychiatry Res. 270, 306–309 (2018).
Song, W. et al. Identification of high risk DISC1 structural variants with a 2% attributable risk for schizophrenia. Biochem. Biophys. Res. Commun. 367, 700–706 (2008).
Sachs, N. A. et al. A frameshift mutation in Disrupted in Schizophrenia 1 in an American family with schizophrenia and schizoaffective disorder. Mol. Psychiatry 10, 758–764 (2005).
Wang, H. Y. et al. Gene polymorphisms of DISC1 is associated with schizophrenia: Evidence from a meta-analysis. Prog. Neuropsychopharmacol. Biol. Psychiatry 81, 64–73 (2018).
Tomppo, L. et al. Association of variants in DISC1 with psychosis-related traits in a large population cohort. Arch. Gen. Psychiatry 66, 134–141 (2009).
Palo, O. M. et al. Association of distinct allelic haplotypes of DISC1 with psychotic and bipolar spectrum disorders and with underlying cognitive impairments. Hum. Mol. Genet. 16, 2517–2528 (2007).
Hennah, W. et al. DISC1 association, heterogeneity and interplay in schizophrenia and bipolar disorder. Mol. Psychiatry 14, 865–873 (2009).
Chen, Y.-M., Lin, C.-H. & Lane, H.-Y. Distinctively lower DISC1 mRNA levels in patients with schizophrenia, especially in those with higher positive, negative, and depressive symptoms. Pharmacol. Biochem. Behav. 213, 173335 (2022).
Ayalew, M. et al. Convergent functional genomics of schizophrenia: From comprehensive understanding to genetic risk prediction. Mol. Psychiatry 17, 887–905 (2012).
Facal, F. & Costas, J. Evidence of association of the DISC1 interactome gene set with schizophrenia from GWAS. Prog. Neuropsychopharmacol. Biol. Psychiatry 20, 1–5 (2019).
Greenwood, T. A. et al. Genome-wide association of endophenotypes for schizophrenia from the consortium on the genetics of schizophrenia (COGS) study. JAMA Psychiatry 76, 1274–1284 (2019).
Ripke, S. et al. Biological insights from 108 schizophrenia-associated genetic loci. Nature 511, 421–427 (2014).
Holland, D. et al. Beyond SNP heritability: Polygenicity and discoverability of phenotypes estimated with a univariate Gaussian mixture model. PLoS Genet. 16, 1–30 (2020).
Pardiñas, A. F. et al. Common schizophrenia alleles are enriched in mutation-intolerant genes and in regions under strong background selection. Nat. Genet. 50, 381–389 (2018).
Ram Murthy, A. et al. Gender-specific association of TSNAX/DISC1 locus for schizophrenia and bipolar affective disorder in South Indian population. J. Hum. Genet. 57, 523–530 (2012).
Harris, S. E. et al. Variation in DISC1 is associated with anxiety, depression and emotional stability in elderly women. Mol. Psychiatry 15, 232–234 (2010).
Luo, X. et al. Association study of DISC1 genetic variants with the risk of schizophrenia. Psychiatr. Genet. 26, 132–135 (2016).
Hennah, W. et al. Haplotype transmission analysis provides evidence of association for DISC1 to schizophrenia and suggests sex-dependent effects. Hum. Mol. Genet. 12, 3151–3159 (2003).
Hennah, W. et al. A haplotype within the DISC1 gene is associated with visual memory functions in families with a high density of schizophrenia. Mol. Psychiatry 10, 1097–1103 (2005).
Cannon, T. D. et al. Association of DISC1/TRAX haplotypes with schizophrenia, reduced prefrontal gray matter, and impaired short-and long-term memory. Arch. Gen. Psychiatry 62, 1205–1213 (2005).
Hodgkinson, C. A. et al. Disrupted in schizophrenia 1 (DISC1): Association with schizophrenia, schizoaffective disorder, and bipolar disorder. Am. J. Hum. Genet. 75, 862–872 (2004).
Walton, E., Turner, J. A. & Ehrlich, S. Neuroimaging as a potential biomarker to optimise psychiatric research and treatment. Int. Rev. Psychiatry 25, 619–631 (2013).
Braff, D. L. The importance of endophenotypes in schizophrenia research. Schizophr. Res. 163, 1–8 (2015).
Harari, J. H. et al. The association between gene variants and longitudinal structural brain changes in psychosis: A systematic review of longitudinal neuroimaging genetics studies. NPJ Schizophrenia 3, 1–12 (2017).
Duff, B. J., Macritchie, K. A. N., Moorhead, T. W. J., Lawrie, S. M. & Blackwood, D. H. R. Human brain imaging studies of DISC1 in schizophrenia, bipolar disorder and depression: A systematic review. Schizophr. Res. 147, 1–13 (2013).
Carless, M. A. et al. Impact of DISC1 variation on neuroanatomical and neurocognitive phenotypes. Mol. Psychiatry 16, 1096–1104 (2011).
Teng, S. et al. Rare disruptive variants in the DISC1 Interactome and Regulome: Association with cognitive ability and schizophrenia. Mol. Psychiatry 23, 1270–1277 (2018).
Vázquez-Bourgon, J. et al. Effect of DISC1 polymorphisms on the long-term course of neurocognitive deficits in non-affective psychosis. Eur. Psychiatry 30, 861–867 (2015).
Chakirova, G. et al. The effects of DISC1 risk variants on brain activation in controls, patients with bipolar disorder and patients with schizophrenia. Psychiatry Res. Neuroimaging 192, 20–28 (2011).
Callicott, J. H. et al. Variation in DISC1 affects hippocampal structure and function and increases risk for schizophrenia. Proc. Natl. Acad. Sci. U S A. 102, 8627–8632 (2005).
Nicodemus, K. K. et al. Evidence of statistical epistasis between DISC1, CIT and NDEL1 impacting risk for schizophrenia: Biological validation with functional neuroimaging. Hum. Genet. 127, 441–452 (2010).
Callicott, J. H. et al. DISC1 and SLC12A2 interaction affects human hippocampal function and connectivity. J. Clin. Investig. 123, 2961–2964 (2013).
DiGiorgio, A. et al. Association of the Ser704Cys DISC1 polymorphism with human hippocampal formation gray matter and function during memory encoding. Eur. J. Neurosci. 28, 2129–2136 (2008).
Prata, D. P. et al. Effect of disrupted-in-schizophrenia-1 on pre-frontal cortical function. Mol. Psychiatry 13, 915–917 (2008).
Prata, D. P. et al. No association of Disrupted-in-Schizophrenia-1 variation with prefrontal function in patients with schizophrenia and bipolar disorder. Genes Brain Behav. 10, 276–285 (2011).
Brauns, S. et al. DISC1 is associated with cortical thickness and neural efficiency. Neuroimage 57, 1591–1600 (2011).
Kahn, R. S. & Keefe, R. S. E. Schizophrenia is a cognitive illness: Time for a change in focus. JAMA Psychiatry 70, 1107–1112 (2013).
Huang, A. S. et al. Brain function during stages of working memory in schizophrenia and psychotic bipolar disorder. Neuropsychopharmacology 44, 2136–2142 (2019).
Bähner, F. & Meyer-Lindenberg, A. Hippocampal–prefrontal connectivity as a translational phenotype for schizophrenia. Eur. Neuropsychopharmacol. 27, 93–106 (2017).
Guo, J. Y., Ragland, J. D. & Carter, C. S. Memory and cognition in schizophrenia. Mol. Psychiatry 24, 633–642 (2019).
Minzenberg, M. J., Laird, A. R., Thelen, S., Carter, C. S. & Glahn, D. C. Meta-analysis of 41 functional neuroimaging studies of executive function in schizophrenia. Arch. Gen. Psychiatry 66, 811–822 (2009).
Johnstone, M. et al. DISC1 in schizophrenia: Genetic mouse models and human genomic imaging. Schizophr. Bull. 37, 14–20 (2011).
Egli, T. et al. Identification of two distinct working memory-related brain networks in healthy young adults. eNeuro 5, 1–19 (2018).
Zhang, F. et al. Genetic association between schizophrenia and the DISC1 gene in the Scottish population. Am. J. Med. Genet. B Neuropsychiatr. Genet. 141B, 155–159 (2006).
Buckner, R. L. & DiNicola, L. M. The brain’s default network: updated anatomy, physiology and evolving insights. Nat. Rev. Neurosci. 20, 593–608 (2019).
Hu, M.-L. et al. A review of the functional and anatomical default mode network in schizophrenia. Neurosci. Bull. 33, 73 (2017).
Gong, X. et al. A brain-wide association study of DISC1 genetic variants reveals a relationship with the structure and functional connectivity of the precuneus in schizophrenia. Hum. Brain Mapp. 35, 5414–5430 (2014).
Opmeer, E. M. et al. DISC1 gene and affective psychopathology: A combined structural and functional MRI study. J. Psychiatr. Res. 61, 150–157 (2015).
Crespi, B. & Badcock, C. Psychosis and autism as diametrical disorders of the social brain. Behav. Brain Sci. 31, 241–320 (2008).
Rose, E. J. & Donohoe, G. Brain vs behavior: An effect size comparison of neuroimaging and cognitive studies of genetic risk for schizophrenia. Schizophr. Bull. 39, 518–526 (2013).
Singh, K. K. et al. Common DISC1 polymorphisms disrupt Wnt/GSK3β signaling and brain development. Neuron 72, 545–558 (2011).
Gou, N. et al. Effects of DISC1 polymorphisms on resting-state spontaneous neuronal activity in the early-stage of schizophrenia. Front. Psychiatry 9, 1–11 (2018).
Gomar, J. J. et al. Validation of the word accentuation test (TAP) as a means of estimating premorbid IQ in Spanish speakers. Schizophr. Res. 128, 175–176 (2011).
Kay, S. R., Fiszbein, A. & Opler, L. A. The positive and negative syndrome scale (PANSS) for schizophrenia. Schizophr. Bull. 13, 261–276 (1987).
Peralta, V. & Cuesta, M. J. Psychometric properties of the Positive and Negative Syndrome Scale (PANSS) in schizophrenia. Psychiatry Res. 53, 31–40 (1994).
Purcell, S. et al. PLINK: A tool set for whole-genome association and population-based linkage analyses. Am. J. Hum. Genet. 81, 559–575 (2007).
Gevins, A. & Cutillo, B. Spatiotemporal dynamics of component processes in human working memory. Electroencephalogr. Clin. Neurophysiol. 87, 128–143 (1993).
Cremaschi, L. et al. Assessing working memory via n-back task in euthymic bipolar i disorder patients: A review of functional magnetic resonance imaging studies. Neuropsychobiology 68, 63–70 (2013).
Nikolin, S. et al. An investigation of working memory deficits in depression using the n-back task: A systematic review and meta-analysis. J. Affect. Disord. 284, 1–8 (2021).
Zhao, J., Wang, J., Huang, C. & Liang, P. Involvement of the dorsal and ventral attention networks in visual attention span. Hum. Brain Mapp 43, 1941-1954 (2022).
Pomarol-Clotet, E. et al. Failure to deactivate in the prefrontal cortex in schizophrenia: Dysfunction of the default mode network?. Psychol. Med. 38, 1185–1193 (2008).
Zanello, A., Curtis, L., Badan Bâ, M. & Merlo, M. C. G. Working memory impairments in first-episode psychosis and chronic schizophrenia. Psychiatry Res. 165, 10–18 (2009).
Forbes, N. F., Carrick, L. A., McIntosh, A. M. & Lawrie, S. M. Working memory in schizophrenia: A meta-analysis. Psychol. Med. 39, 889–905 (2009).
van Snellenberg, J. X. et al. Mechanisms of working memory impairment in schizophrenia. Biol. Psychiatry 80, 617–626 (2016).
Smith, S. M. et al. Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23(Suppl 1), S208–S219 (2004).
Worsley, K. J. Statistical analysis of activation images. In Functional Magnetic Resonance Imaging 251–270 (Oxford University Press, 2001).
Acknowledgements
This study received funding provided by: (i) the Spanish Ministry of Science and Innovation, Instituto de Salud Carlos III through the projects PI15/01420 and PI18/01535 (co-funded by European Regional Development Fund (ERDF)/European Social Fund "Investing in your future"), and also through PFIS predoctoral contracts to M Guardiola-Ripoll and N Hostalet (FI19/0352 and FI21/00093) and a Miguel Servet contract to M Fatjó-Vilas (CP20/00072); (ii) The Health Department from the Generalitat de Catalunya with a PIF-Salut contract to AS-M (SLT017/20/000233), iii) the Comissionat per a Universitats i Recerca del DIUE of the Generalitat de Catalunya (Agència de Gestió d'Ajuts Universitaris i de Recerca (AGAUR), 2017SGR1271 and 2017SGR1577).
Author information
Authors and Affiliations
Contributions
M.G.-R., P.J.M., E.P.-C. and M.F.-V. conceived the study. M.G.-R., A.S.-M., C.A.-P., B.A. and M.F.-V. conducted the genetic markers selection, the DNA extraction and the genotyping. A.G.-P., N.R., J.O.-G., M.M., and J.S.-V. conducted the recruitment and/or the clinical evaluations. R.S., P.J.M. and E.P.-C. designed the MRI protocol and supervised the fMRI analyses. M.G.-R., A.S.-M. and M.F.-V. performed the data curation and the statistical analyses. M.G.-R., A.S.-M., C.A.-P. and M.F.-V. wrote the first draft and subsequent drafts of the paper. M.G.-R., A.S.-M., C.A.-P., N.H., R.S., P.J.M., E.P.-C. and M.F.-V. interpreted the results and revised the manuscript. M.M., E.P.-C. and M.F.-V. obtained the funding and resources. All the authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guardiola-Ripoll, M., Sotero-Moreno, A., Almodóvar-Payá, C. et al. Combining fMRI and DISC1 gene haplotypes to understand working memory-related brain activity in schizophrenia. Sci Rep 12, 7351 (2022). https://doi.org/10.1038/s41598-022-10660-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-10660-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.