Abstract
Cells are often considered input-output devices that maximize the transmission of information by converting extracellular stimuli (input) via signaling pathways (communication channel) to cell behavior (output). However, in biological systems outputs might feed back into inputs due to cell motility, and the biological channel can change by mutations during evolution. Here, we show that the conventional channel capacity obtained by optimizing the input distribution for a fixed channel may not reflect the global optimum. In a new approach we analytically identify both input distributions and input-output curves that optimally transmit information, given constraints from noise and the dynamic range of the channel. We find a universal optimal input distribution only depending on the input noise, and we generalize our formalism to multiple outputs (or inputs). Applying our formalism to Escherichia coli chemotaxis, we find that its pathway is compatible with optimal information transmission despite the ultrasensitive rotary motors.
Similar content being viewed by others
Introduction
Biological cells continuously process environmental cues, allowing them to make critical decisions quickly, e.g. whether to move or stay, whether to express a certain protein, or whether to divide1. These decisions are generally made based on the level of one or more key proteins, which are the internal representation of the extracellular stimulus. The higher the amount of environmental information encoded in the intracellular representation, the more reliable the response. In contrast, cell-external and internal noise may reduce the reliability. Hence, a biological system under evolutionary pressure is expected to evolve to optimally transmit information under biologically relevant constraints2, at least when information is a limiting factor3,4,5.
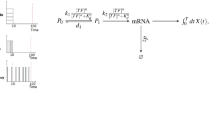
To formalize this optimization problem, cells can be considered input-output devices, where stimuli of extracellular concentrations are the input, receptors (or the entire pathway) are the communication channel, and the intracellular concentration of a key protein (or the final behavior of the cell) is the output. In contrast to engineered physical systems, the distinction between input, channel, and output is not always clear in biological systems with feedback. Take for instance Escherichia coli chemotaxis, a well-characterized pathway allowing bacteria to sense chemicals and to swim towards nutrients6,7. The intracellular level of the phosphorylated protein (CheYp) represents the extracellular concentration of a chemical and regulates the motors (clockwise or counterclockwise rotation) and hence motility (‘run’ or ‘tumble’) (Fig. 1). The swimming behavior clearly affects the input as cells change their location, making information flow a circular problem. Hence, the question emerges how to tackle such problems.
Connection between environmental cues and chemotactic response. Chemotactic bacteria live in complex microenvironments in which input distributions of chemical concentrations are shaped by the swimming behavior (top left), chemical sources and sinks (top middle), and competition with other bacteria (top right). Inputs are processed by the cell-internal chemotaxis pathway, which can be viewed as an input-output device (bottom). Specifically, input-output curves are measured in experiments by dose-response curves with noise. The resulting final behavior feeds back into the environment. Evolution is assumed to select the best input-output curves for maximizing fitness. The chemotactic pathway is a two-component system, and for modeling purposes, is divided into two information-transmission channels: receptors sense external concentration of stimuli and their activity regulates the protein CheYp (receptor channel, bottom left). CheYp is the internal representation of the external stimulus and regulates motor switching (clockwise or counterclockwise rotation) and thus bacterial motility (straight swimming via a ‘run’ or random reorientation via a ‘tumble’; motility channel, bottom right). Note that there is additional adaptation both at the receptors75 and the motors52.
Shannon’s mutual information is generally used to quantify information transmission, capturing the statistical (linear and nonlinear) dependencies between inputs and outputs8. Specifically, the mutual information describes the ability on average to reconstruct the input distribution after repeatedly measuring the output8,9. Often maximal mutual information is assumed, either to reflect biological function or because the mutual information cannot be calculated otherwise10,11. How should mutual information be maximized? Maximizing with respect to the (generally unknown) distribution of inputs leads to the channel capacity. Such an approach was, e.g., used to study transcriptional regulation in the developing fruit-fly embryo12,13. While in this case, the mother organism may be able to tune its maternal factors to the optimal input distribution to match the ‘expectation’ of the embryo, the channel capacity may not generally be a valid approach. Alternatively, it is possible to assume a fixed input distribution and to maximize the mutual information with respect to the input-output curve14,15. The underlying idea is that the input-output curve is adjusted by evolution to match the distribution of inputs. This approach might apply to organisms confined to certain environments, e.g. bacteria living in specific niches15. In addition to these two optimization procedures (and some attempts to combine them for specific types of input-output curves16,17,18), the Fisher information from estimation theory may also be used to predict the input distribution19,20. Hence, due to multiple available approaches there is considerable uncertainty to what optimization procedure to use.
Even the principle of maximal information transmission may be questioned. Recently, maximizing information transmission at the E. coli receptors led to maximal drift of cells swimming up a chemical gradient and hence optimal chemotactic behavior15. Although a reasonable result, there are two potential problems when considering the whole pathway: Firstly, the dose-response curves of the motors are measured to have Hill coefficients up to 2021. Hence, such switch-like motors may only transmit about one bit of information, distinguishing only low and high levels of CheYp. Secondly, the CheYp level, which maximizes the drift is not in the sensitive region of the motor22,23,24. Both issues may lead one to suggest that high information transmission at the receptors is wasted downstream at the motors, and that an entirely different principle may guide cell behavior25,26.
Here, we address two key questions: (1) How should the mutual information be calculated in a biological context, and (2) does the bacterial chemotaxis pathway maximize information transmission? Specifically, we reconcile the various optimization procedures, leading to a new way of maximizing the mutual information, particularly useful for biological systems. Assuming general external and internal noise, and a fixed range of sensitivity (as any biological or physical system is necessarily limited), we analytically derive both the optimal input distribution and input-output curve. Unlike previous approaches16,17,18, this general solution does not assume specific input-output curves, such as Hill functions. Surprisingly, we find an universal optimal input distribution, only dependent on the input noise. Furthermore, numerically and with the help of simulations, we were able to extend our formalism to multiple outputs (or inputs), greatly extending the applicability of our formalism to biological systems. As an illustrative example, we focus on the E. coli chemotaxis pathway using previously estimated noise and measured dose-response curves at the receptors and the motors. By deriving analytical results for multiple output motors, we show that although the optimal response is not a Hill function, the measured Hill coefficients naturally emerge from our optimal prediction. Overall our results confirm the idea of maximal information transmission in E. coli chemotaxis, and prove that maximal information transmission at the receptors is critical for the whole pathway despite the ultrasteep motor dose-response curves.
Results
Maximizing mutual information: comparison of different approaches
Information transmission between an input, X, and an output, Y, is often quantified by the mutual information, which is a measure of statistical dependency and reflects the average ability for inferring the input after measurements of the output (8,9,10,11,27,28,29 for extended reviews). For continuous random variables, X and Y, the mutual information is defined by
where p(y|x) is the conditional probability of observing Y = y at given X = x, encoding the input-output curve and noise. Quantities p(x) and p(y) represent the input and output distributions, respectively, which are mathematically connected by the conservation of probability p(x)dx = p(y)dy, valid in the small-noise limit. Considering small Gaussian noise with conditional probability, \(p(y|x)=\exp [-{(y-\bar{y})}^{2}\mathrm{/(2}{\sigma }_{{\rm{T}}}^{2})]/\sqrt{2\pi {\sigma }_{{\rm{T}}}^{2}}\), with mean \(\bar{y}\)(x) and standard deviation σT(x), Eq. (1) becomes
where xon and xoff set the sensitive region, i.e. the dynamic range of inputs14,15 (similar equations appear in12,13,20). In addition to p(x), Eq. (2) depends on the gain \(\bar{y}^{\prime} \)(x), i.e. the first derivative of the input-output curve \(\bar{y}\)(x), and total noise σT(x).
To understand if biological systems maximize information flow, we need to maximise the mutual information and derive general principles or compare with data. To maximise the mutual information, the channel capacity is often considered, i.e. the mutual information maximized with respect to the input distribution12,13. Alternatively, the mutual information can be maximized with respect to the input-output curve assuming a fixed input distribution14,15. The former method is an attempt to deal with the often unknown input distribution, while the latter is based on the idea that the biological channel can be modified by evolution. Additionally, the two approaches were combined for specific input-output Hill and Hill-like functions16,17,18. However, is there a general way to unify the different methods, without making assumptions about functional form of the input-output functions?
Formally, we maximize the mutual information, Eq. (2), with respect to p(x) and \(\bar{y}\)x) by writing
where the right-hand side of Eqs. (3) and (4) are Euler-Lagrange equations from calculus of variations with Lagrangian \( {\mathcal L} (x,p,\bar{y},\bar{y}^{\prime} )=p\,{\log }_{2}[\frac{\sqrt{2\pi e}{\sigma }_{{\rm{T}}}}{\bar{y}^{\prime} }p]\), i.e. the integrand of Eq. (2). Equation (3) represents the channel capacity applied to a Gaussian channel (i.e. Gaussian conditional probability p(y|x), see12,13 for examples in gene regulation). In contrast, Eq. (4) is used to obtain the optimal input-output curve for a given input distribution (see14,15 for examples in sensory systems). For completeness, we provide the solutions of the individual maximizations of Eqs. (3) and (4) in SI Text Sec. 1.1–1.2, with a discussion of the sensitive region in Sec. 1.8.
When the noise is uniform (σT constant) Eqs (3) and (4) coincide. Specifically, \(\frac{\partial {\mathcal L} }{\partial \bar{y}}=0\) in Eq. (4) so that \(\bar{y}\) is a cyclic variable. As a result, \(\frac{{\rm{d}}}{{\rm{d}}x}\frac{\partial p}{\partial \bar{y}^{\prime} }=0\) so that p/\(\bar{y}^{\prime} \) = const and hence is conserved (not in time but in input space), following the Emmy Noether theorem. In this case, maximizing the mutual information leads to a simple matching relationship (\(\bar{y}^{\prime} \) ∝ p), so that the input-output curve is the cumulative integral of the input distribution (see S1 text, Sec. 1.1 and Fig. S1)30. However, in general when both input and output noise matter the noise is a function of the input and input-output curve, given by σT = σT(x, \(\bar{y}\), \(\bar{y}^{\prime} \)). Assuming independent cell-external and internal noise, we consider
which follows from error propagation. Specifically, σx is the input noise depending on x only, amplified by the gain \(\bar{y}^{\prime} \)(x), and σy is the output noise depending on \(\bar{y}\)(x) only. In case of negligible input (σx ≈ 0) or output (σy ≈ 0) noise, Eq. (3) again converges to Eq. (4) and the system can be solved for any input-output curve \(\bar{y}\)(x) (see S1 text, Sec. 1.2–1.3). As a result, the predicted input and output distributions from the two optimization approaches become identical (Fig. 2A). However, in general the two equations differ and the resulting optimal input and output distributions are very different in the two approaches (Fig. 2B). In particular, the output distributions can be uni- or bimodal with details described in SI Text, Sec. 1.3.
Conventional ways of maximizing mutual information. Comparison of the channel capacity, i.e. solving the maximization problem Eq. (3) for p(x) using a fixed \(\bar{y}\)(x) and noise (red), and maximization with respect to input-output curve, i.e. solving the maximization problem Eq. (4) for \(\bar{y}\)(x) using a fixed p(x) and noise (blue). (For specific examples of the solutions of Eqs (3) and (4) and a discussion of the bimodality of the output distribution, see SI Text Sec. 1). In both cases, the noise is provided as a function of the input distribution and input-output curve (top row), with the input-output curve \(\bar{y}\)(x) = [1 + (x/kd)nH]−1 assumed a Hill function for simplicity, with Hill coefficient nH and threshold kd (inset). (A) For small input noise, the two approaches converge, i.e. the red and the blue input (middle left) and output (bottom left) distributions match. (B) For large input noise, the two approaches predict different input (middle right) and output (bottom right) distributions. Using Eq. (5) for the noise, the input noise is σx2 = α1x, while the output noise σy2 = α2\(\bar{y}\)(1 − \(\bar{y}\)) + α3\(\bar{y}\) + α4 has three different contributions with \(\bar{y}\) the input-output curve. The parameters are chosen to provide an overall similar level of noise, given by α1 = 10−8, α2 = 2 · 10−6, α3 = 10−7, α4 = 10−8 (panel A) and α1 = 10−7, α2 = 10−8, α3 = 10−7, α4 = 10−8 (panel B). For the red model, nH = 5 and kd = 0.1. For the blue model, the input distribution is fixed by normalizing the derivative of a Hill function with nH = 5 and kd = 0.1, with the input-output curve free to change according to the maximization. The sensitive region is set by xon = 0 and xoff = +∞.
It is worth noting that, in Bayesian statistics the Fisher information is linked to the channel capacity19,20. A key problem in Bayesian statistics is choosing a prior distribution for a given stochastic process (i.e. p(y|x))31. The idea of having a prior which does not affect the posterior distribution (i.e. p(x|y)) is linked to maximal mutual information, given by the average Kullback-Leibler divergence between prior and posterior distributions. This prior distribution is called the reference prior19, given by \(p(x)\propto \sqrt{ {\mathcal F} (x)}\) with Fisher information \( {\mathcal F} (x)=\int \,dy\,p(y|x){(\partial \log (p(y|x))/\partial x)}^{2}\). As shown in ref. 20. the Fisher information is the result of maximizing the equivalent of Eq. (2) for a general (not necessarily Gaussian) conditional probability distribution (see S1 Text, Sec. 2.). Hence, the channel capacity and the approach based on the Fisher information are equivalent.
Maximizing mutual information: a new approach
The difference between Eqs. (3) and (4) is that Eq. (3) assumes a fixed input-output curve and a variable input distribution, while Eq. (4) assumes a fixed input distribution and a variable input-output curve. There might be situations in which one approach is more appropriate than the other but in a general biological context the two are intrinsically connected (Fig. 1). From a mathematical point of view, Eqs (3) and (4) can be combined and solved together, i.e. \( {\mathcal I} [x,y]\) maximized with respect to both p(x) and \(\bar{y}\)(x). Similar numerical double optimizations are common in rate distortion theory using, e.g., the Blahut algorithm4,32.
In what follows, we provide the analytical solution for p(x) and \(\bar{y}\)(x) by solving Eqs. (3) and (4) together. We assume a fixed dynamical range of inputs set by xon and xoff (given by the receptor sensitivity), leading in return to a fixed dynamical range of outputs from y(xon) = 1 to y(xoff) = 0. We consider Eq. (2) with noise given by Eq. (5). After a first integration, we obtain the formal solution
where Z and Q are two constants set by normalization and boundary conditions, respectively (see Materials and Methods and S1 Text, Sec. 1.7).
Equation (6a) for the input distribution extends the matching relationship found in30 to nonuniform noise. In the latter case the optimal input distribution weighs certain inputs more than uncertain inputs13,20,33. Equation (6b) determines the input-output curve which maximizes the mutual information given the noise. A solution of the system of equations exists if the transmitted input noise can be expressed in terms of the output noise or vice versa (see S1 Text, Sec. 1.7). While such a solution may seem very specific, it is certainly plausible, given enough time, that evolution eventually finds it.
How may evolution find the solution? To mimic evolution, we envision an adaptive algorithm, allowing the pathway to iteratively reach optimal information transmission. Given an environment and hence a distribution of inputs, p1(x) (Fig. 3A), evolution selects the optimal internal input-output curve, y1(x) (Fig. 3B). However, the distribution of inputs is susceptible to changes, which might be caused by a change of the organism’s behavior, even in the same environment. The new input distribution, p2(x), may again lead to an increase in information transmission at fixed input-output curve, y1(x) (Fig. 3C). Subsequently, evolution will select a new input-output curve, y2(x), which enhances information transmission at a fixed input distribution, p2(x) (Fig. 3D). This cycle is repeated many times. If the optimal configuration is achievable and information transmission is a proxy for fitness, we expect that the solution of Eqs. (6a) and (6b) naturally emerges in the pathway. This is indeed the case for the examples studied here (see Fig. 3E,F).
Adaptive evolutionary algorithm approaches analytical result. (A) Bacteria are assumed to live in a given environment and experience a given distribution of inputs. (B) Assuming that maximal information transmission enhances the chance to survive, evolution selects the mutations and hence the phenotype with optimal input-output curve. (C) At given input-output curve, the optimal input distribution generally (dark blue) differs from the initial input distribution (light blue) in (A). A change in behavior (and hence a change in the inputs) may provide an increase in information transmission (see Discussion section for more details). (D) The input-output curve changes again to maximise information transmission for the new set of stimuli. (E,F) This iterative cycle continues and eventually converges to the solution given by Eqs. (6a) and (6b) (dashed red line). Parameters: α1−3 = 10−7, α4 = 10−8, xon = 0.115, xoff = 0.323.
Information transmission at E. coli chemoreceptors
To apply our new approach, we use the chemotaxis pathway of E. coli as an explicit example, since it is relatively simple and well characterized in its molecular components6. Briefly, chemoattractant (ligand) binding turns receptors off, inhibits the kinase CheA, and hence reduces the phospho-transfer from CheAp to CheY. This leads to ‘runs’ as only CheYp can bind the 6–8 motors to introduce ‘tumbling’. There is also an adaptation mechanism, where addition of methyl groups to receptors compensates for increased attractant concentration by increasing the receptor activity and hence the CheYp level to induce cell tumbling. Removal of methyl group has the opposite effect34,35. In order to study signaling in fixed adaptational states, the adaptation enzymes can be removed from the chromosome and the receptor expressed with specific, genetically engineered, receptor modification levels to mimic receptor methylation (see S1 Text, Sec. 4.3)34,35.
Specifically, we consider the instantaneous information transmission between the chemoattractant methylaspartate (MeAsp) as the input and the response regulator CheYp as the output. Hence, we consider the information transmitted by the initial (fast) response for a given adaptational state (which only changes slowly). (At a later time this response is removed by adaptation and hence is transient only). Note, unlike ref.36 we do not assume small Gaussian inputs but natural stimuli drawn from broad, potentially asymmetric input distributions p(x). When the input distribution of cells simulated in gradients of different strength match the optimal information-theoretical input distribution for the same receptor modification levels, the drift velocity up the gradient is maximized and, hence, this leads to optimal chemotaxis15. As this matching of input distributions occurs anywhere in the gradient, these initial responses describe chemotaxis in the whole gradient, and so implicitly include adaptation. Indeed, the predicted distributions of inputs are scale invariant (when normalized by the adapted concentration) and reproduce Weber’s law and logarithmic sensing15. The latter can also be captured by the predictive mutual information37.
The functional form of the noise is assumed to be known and derived from microscopic theory as in38 (see S1 Text, Secs. 3 and 4.2 for noise estimation and sensitivity to noise parameters, respectively). In short, the input variance is considered to be proportional to the input strength, σx2 = α1x, with x in units of the ligand concentration the cell is adapted to. Furthermore, α1 ∝ (DNτ)−1 is given by the Berg-and-Purcell limit39, where D is the diffusion constant of the ligand molecules, N is the number of receptors acting cooperatively in a cluster, and τ is the averaging time, assuming a spherical cell39. The output noise has three contributions: signaling noise, switching noise due to on/off changes of the receptor state, and a constant background noise, leading to σy2 = α2\(\bar{y}\)(1 − \(\bar{y}\)) + α3\(\bar{y}\) + α4 with phosphorylated \(\bar{y}\) in units of the total CheY level, YT, an intrinsic dependence on the (unknown) input-output curve, and α2−4 additional parameters defined in S1 Text, Sec. 3. Note these effective noise terms are time-averaged due to their dependence on chemical reactions based on finite rate constants38. Despite σy2 being specific, this noise should apply to many receptor-signaling pathways, including other two-component pathways40.
Using this noise, the explicit solution of Eq. (6b) is
where C and Q are constants set by imposing fixed boundary conditions, y(xoff) = 1 and y(xon) = 0 (Fig. 4A). Note that x appears with the prefactor Q/α1. Thus, α1 is set by the boundary conditions and it can be seen as the units of x. We consider two special cases: by simplifying the output noise for α2 = 0, we obtain
where the solution is again independent of α1 after imposing the boundary conditions. For α2,3 = 0, we obtain
which does not depend on both α1 and α4 once Q and C′′ are set by the boundary conditions. The optimal input distribution obtained by inserting Eq. (6b) into Eq. (6a) is \(p={(Z\sqrt{\mathrm{(1}+Q)/Q}{\sigma }_{x})}^{-1}\). In particular, for our choice of external noise and fixed sensitivity range, the input distribution converges to \(p(x)={[\mathrm{2(}\sqrt{{x}_{{\rm{on}}}}-\sqrt{{x}_{{\rm{off}}}})\sqrt{x}]}^{-1} \sim 1/\sqrt{x}\) independently of α1−4 (see S1 Text, Sec. 1.4.6 and Fig. S3). Importantly, this is a general result for the Berg-and-Purcell input noise, and hence should be valid for many signaling pathways. This result was previously found numerically16, and such an input distribution of glutamine was suggested to optimize nitrogen sensing33.
Robustness of the adaptive algorithm. Comparison between our analytical result (blue lines) and the corresponding result when the input-output curve is constrained to a Hill function with adjustable Hill coefficient nH (red lines). (A,B) Input distributions (A) and the input-output curves (B). (C) Comparison between the adaptive algorithm for the unconstrained (blue lines) and Hill-function constrained (red solid line) optimizations. The adaptive algorithm moves the input-output curves to the optimal value nH ≈ 5, steadily increasing the mutual information. The convergence towards the analytical solution is robust to different initial Hill coefficients (dashed blue lines). The optimal Hill coefficient is compatible with the corresponding experimental curve from35 (see also Fig. S9). When constraining the input-output curve to a Hill function, the highest mutual information at a given Hill coefficient is shown by the red dashed line. Noise parameters: α1−3 = 10−4, α4 = 10−5, xon = 0.115 and xoff = 0.323 (see S1 Text, Sec. 3 for details of the noise).
Extracting the sensitive regime of E. coli receptors for a fixed modification level (resembles receptor methylation level, see S1 Text, Sec. 4.3)35,41, we test the convergence of the adaptive algorithm to the solution in Eq. (7). After a few iterative cycles the system indeed converges (Fig. 4A), increasing the mutual information at each step (Fig. 4C, blue line). This convergence to the analytical solution occurs when starting at different initial conditions, showing robustness of our algorithm. Note that in Fig. 4C the solution \(\bar{y}\)(x) is fitted to Hill functions for convenience of presentation, allowing the mutual information to be plotted as a function of a single parameter (i.e. the Hill coefficient n). The optimal curve selects a Hill coefficient compatible to the experimental measurements from FRET data, at least for larger receptor modification levels (Fig. S9)35. Applying the adaptive algorithm instead to Hill-function constrained input-output curves produces the same optimal Hill coefficient n albeit with a smaller mutual information (Fig. 4C, red solid line, with Fig. 4B comparing the corresponding optimal input distributions and optimal input-output curves). Note that for a fixed Hill equation the optimal mutual information is calculated directly using the input distribution from Eq. (6a), resulting in
as shown in Fig. 4C (red dashed line).
However, unlike the sine function in Eq. (7), experimental dose-response curves of CheYp are thought to be well approximated by Hill functions rather than Eq. (7)34. There are several possible reasons for this discrepancy. For instance, in our model receptors are either fully sensitive or fully insensitive, and the solution given by Eq. (7) is only valid in the sensitive region and constant otherwise. In reality, receptors have a smooth sensitivity curve spanning the ligand-dissociation constant of the off and on states (such as dF/dlog(x) in41, where F is the receptor free-energy difference between on and off states). This may lead to a smooth Hill-function-like response. One way of imposing smooth input-output curves is to introduce the additional constraint of zero first derivatives at the boundary. In this case, however, we only obtain sigmoidal input-output curves without internal switching noise (α3 = 0) (see S1 Text, Sec. 1.4.4 and Fig. S3E). Moreover, very asymmetric (or even bimodal) input distributions might be uncommon in natural environments15, potentially favoring log-normal input distributions and hence Hill-function-like responses42. Finally, E. coli needs to account for many other constraints and the pathway performs other tasks at the same time, such as sensing temperature and pH43,44,45. Hence, the E. coli sensory system might be in a suboptimal configuration for transmitting information about chemicals in order to account for all the other tasks. In the S1 Text, Sec. 1.4.5 we also solve the inverse problem and derive the optimal noise, which leads to an exact Hill function (see Fig. S4). In this case, the predicted input and output noises are not independent anymore. In summary, the mismatch between the experimental Hill functions and the solution in Eq. (7) is not an artifact of our assumptions on the sources of noise; it emerges when considering independent input and output noise, and when maximizing the mutual information with respect to both the input distribution and the input-output curve.
Information transmission along the E. coli chemotaxis pathway
Now that we understand the optimization of the mutual information better, we can tackle the second problem: Does the chemotaxis pathway maximise information transmission? Previous work suggests that the higher the information transmission at the receptors, the higher the drift velocity in the direction of the gradient15. Is this finding compatible with the recent observation of the ultrasensitive response of the motor to changes in internal CheYp21, or does such a steep response prevent the cell from high information transmission? To answer this question we extend our analysis to the whole chemotaxis pathway.
We consider a minimal model of two channels: a receptor channel for sensing by the chemoreceptors and a motor channel for the flagellar motors. For the receptor channel, the external chemical concentration x is the input and the internal CheYp concentration, y, is the output. For the motor channel, y is the input and the motor clockwise (CW) bias z (for tumble) is the output (Fig. 5A). To simplify the problem and to closely resemble the experimental dose-response curves, we now restrict the curves to Hill functions, with Hill coefficients n and m for receptors and motors, respectively. The noise expressions for the receptor and motor channels are given by \({\sigma }_{{\rm{yT}}}=\sqrt{{\alpha }_{1}x{G}_{y}^{2}+{\alpha }_{2}\bar{y}\mathrm{(1}-\bar{y})+{\alpha }_{3}\bar{y}+{\alpha }_{4}}\) and \({\sigma }_{{\rm{zT}}}=\sqrt{{\sigma }_{{\rm{yT}}}^{2}{G}_{z}^{2}+{\beta }_{2}\bar{z}\mathrm{(1}-\bar{z})+{\beta }_{3}\bar{z}+{\beta }_{4}}\), respectively, where Gy and Gz are the gains of the receptor and motor channels. Parameters β2−4 represent the noise of the motors and are kept generic due to lack of characterization, but may reflect analogous biological processes including adaptation of the motors21,46,47 (see S1 Text, Sec. 3 for further details and robustness of results to changes in β values). Due to the immense gain at the motors21, we generally have higher noise at the motor then at the receptor (see S1 Text Sec. 4.5 for the discussion of the two limits). Here only n and m are considered adjustable parameters in our model.
Optimal information transmission in the E. coli chemotaxis pathway. (A) Minimal model of the E. coli chemotaxis pathway, using two concatenated channels. The extracellular concentration of stimulus x is the input of the receptor channel and CheYp, y, concentration is the output. For the motor channel, CheYp is the input and the tumble bias z the output. Hill functions with adjustable Hill coefficients n and m represent the input-output curves of the receptor and motor channels, respectively. The dissociation constants for the receptor and motor channels are kdn = 0.189/(1 μM) and kdm = 0.39 [YT], respectively. (B) Heat maps of the separately calculated mutual information of the receptor, \( {\mathcal I} [x,y]\), (left) and of the motor, I[y,z], (right) channels for a single motor as a function of Hill coefficients n and m. Vertical dashed lines correspond to maximal value of \( {\mathcal I} [x,y]\) (circles corresponds to arrows in C). (C) The mutual information as function of Hill coefficient m at n = 5 (dashed black lines in panel B). While for a single motor the optimal mutual information of the receptor channel (opt rec, blue solid line) is higher than the optimal mutual information at the motor (opt mot, red solid line), increasing the number of motors (see legend) enhances the optimal mutual information of the motor channel (opt 1 mot, dashed red line, for two motors). Arrows point to the predicted m values. The purple arrow indicates the optimal m value for a single motor (corresponding to purple circle in B), the green and orange arrows point to the optimal m values for two motors (corresponding to green and orange circles in B). The mutual information is further increased when the optimal mutual information for two motors is calculated (opt 2 mot, orange dashed line, see S1 Text, Sec. 4). Parameters: α1−3 = 10−4, α4 = 10−5, β2 = 7 · 10−4, β3 = 7 · 10−4, and β4 = 1 · 10−4.
The data processing inequality, which characterizes the flow of information in a Markov chain, states that, at any additional processing step, information can only be lost, never gained48. For instantaneous information transmission this means that the mutual information between the external concentration and the motor bias cannot be higher than the minimum of the mutual informations of the receptor and the motor channels, i.e. \( {\mathcal I} [x,z]\le \,{\rm{\min }}\{ {\mathcal I} [x,y],\, {\mathcal I} [y,z]\}\). A strategy to possibly increase the mutual information is then to maximise the limiting mutual information.
We start by considering a single motor, represented by a single output z. We calculate the maximal mutual information at the receptor (Fig. 5B, left) and motor (Fig. 5B, right) channels, dealing with the optimization of the two channels separately. The mutual information at the motor, \( {\mathcal I} [y,z]\), always limits the whole information transmission for any Hill coefficient n of the receptors and m of the motor. Hence, single-motor cells should optimize the motor rather than the receptor channel. This result is not unexpected for the chemotaxis pathways since the ultrasensitive motor enhances the downstream noise, which is generally larger than the upstream noise (here the total CheYp noise is the input noise for the motor channel). The resulting optimal information transmission corresponds to relatively low n and m (≈6; red area in right panel of Fig. 5B). Experimentally, the Hill coefficient of the receptor channel agrees with our prediction, ranging from 6–12 in Tar-only cells35. In contrast, the ultrasteep motor response curve with m ≈ 20 is in stark contradiction to our single-motor dose-response model. Hence, the single-motor model indicates higher information transmission at the receptors (see Figs. 5, S6, S7, and S1 Text, Secs. 3 and 4.2 for dependence on noise). However, E. coli has multiple motors, which might effect information transmission.
We now extend our model to multiple (K) motors, allowing the cell to make multiple measurements of the internal CheYp concentration. There are now a single input, y, and multiple outputs z1, ..., zK, of the CW biases. The chain rule for the mutual information allows us to calculate \( {\mathcal I} [y;{z}_{1},\,\mathrm{..}{z}_{K}]\). In particular, for the extreme case of fully coupled motors the mutual information does not increase with the growing motor number, \( {\mathcal I} [y;{z}_{1},\,\mathrm{..}{z}_{K}]= {\mathcal I} [y;{z}_{1}]\). In contrast, for completely uncoupled motors (i.e. for motors which are simultaneously independent and conditionally independent given y) the mutual information increases with the number of motors, \( {\mathcal I} [y;{z}_{1},\,\mathrm{..}{z}_{K}]=K {\mathcal I} [y;{z}_{1}]\) (see S1 Text, Sec. 4.5). Real motors show evidence of partial coupling49, and thus we assume conditional indepen-dent motors, i.e. p(z1, ..zK|y) = ΠiKp(zi|y). This means that all motors depend on the common y level but can independently select their CW bias. For two motors, the mutual information becomes \( {\mathcal I} [y;{z}_{1},{z}_{2}]= {\mathcal I} [{z}_{1};y]+ {\mathcal I} [{z}_{2};y|{z}_{1}]= {\mathcal I} [{z}_{1};y]-H({z}_{2}|y)+H({z}_{2}|{z}_{1})\). For K motors this is generalized to
Using the small noise Gaussian approximation for p(zi|y), the conditional entropy is given by \(H({z}_{1}|y)=\int {\rm{d}}y\,p(y)\) · \(\log (\sqrt{2\pi {\rm{e}}}{\sigma }_{T}(y))\), and H(z2|z1) ≈ 0 (Fig. 5C, see also S1 Text, Sec. 4.5). Thus, Eq. (11) becomes
We numerically tested that the conditional independence of the motors holds for two motors, despite the fact that motors compete for the binding of internal CheYp molecules, which can introduce negative correlations (see S1 Text, Sec. 4.6 and Fig. S10). Therefore, \( {\mathcal I} [y;{z}_{1},{z}_{2}] > {\mathcal I} [y;{z}_{1}]\), and more generally \( {\mathcal I} [y;{z}_{1},\,\mathrm{..}{z}_{K}] > {\mathcal I} [y;{z}_{1},\,\mathrm{..}{z}_{K-1}]\) for K conditionally independent motors50.
Hence, for conditionally independent motors, the mutual information at the motors will eventually overtake the mutual information at the receptors when the number of motors increases. Consequently, the mutual information at the receptors becomes the limiting factor for information transmission (Fig. 5C, see50 for the case of Gaussian input distributions). In other words, for a small number of motors the cell has high information transmission at the receptors, which will be wasted at the motors (cf. red and blue solid lines). In contrast, for a large number of motors the information transmission at the motors exceeds the information transmission at the receptors without overall improvement. However, in the intermediate case both receptors and motors equally limit the transmission of information (cf. red dashed and blue solid lines in Fig. 5C). E. coli chemotaxis seems to avoid bottlenecks and to optimally allocate resources the latter case is the most advantageous51. Hence, the ultrasteep Hill function of the motor (m ≈ 20) can be explained by this matching of the information transmission at the receptors and motors (orange arrow in Fig. 5C). Note that in addition to the high Hill coefficient m of the motors there is also a corresponding low m solution (green arrow in Fig. 5C). However, the latter is not robust to changes in m, i.e. a small change in m can lead to a drastic reduction of information transmission, which can emerge from varying the number of FliM molecules of the motor52. In addition, note that the mutual information shown in Fig. 5C with a red dashed line is calculated assuming that the two motors are optimized separately. However, the mutual information is further increased by maximizing the two motors simultaneously (dashed orange line in Fig. 5C). Our overall result that a high mutual information can be achieved with a high Hill coefficient of the motors remains valid (see S1 Text, Sec. 4). In between m ≈ 1 and ≈20, the information transmission of the motor is wasted as receptors are information-flow limiting. In conclusion, multiple ultrasensitive motors are only useful when motors are sufficiently independent. Any residual coupling among motors may be the result of close motor proximity or mechanical coupling of the flagella.
Discussion
This study presents a new approach to maximise the mutual information, particularly suitable for evolving biological systems subject to random mutations and selection. Previously, the channel capacity, i.e. the mutual information maximized with respect to the input distribution was widely used for electronic and biological communication channels12,13,20,33. However, this method fails to capture possible changes of the internal input-output curve (e.g. by mutations). Furthermore, the mutual information maximized with respect to the input-output curve neglects the biological relevant feedback of the output on the input14,15. Here, we reconciled these two approaches by maximizing the mutual information with respect to both the input distribution and the input-output curve for Gaussian channels with small noise. Only when the total noise is uniform, or when the input or the output noise is negligible, the two approaches are identical. Unlike previous joint optimizations16,17,18, our input-output curves are not restricted to Hill or Hill-like functions. Our adaptive algorithm demonstrates how evolution might implement this iteratively.
Our analytical solution of the joint optimization provides a number of new insights into optimal information transmission. First, the optimal input distribution is universal, depending only on the input noise. For Berg-and-Purcell type input noise, we specifically obtain \(p(x) \sim {\sqrt{x}}^{-1}\). Hence, organisms are optimized for environments in which low intensity stimuli occur with high frequency. This is sensible as their high frequency would compensate for their large relative noise levels. Second, the optimal input-output curve is invariant to up or down scaling of the input noise (parameter α1), which sets the units of the input. Hence, only the shape of the input noise (i.e. its functional dependence on input x) affects the input-output curve. Third, our optimal input-output curve is rather linear (Figs. 3D and 4A). While this does not match the sigmoidal Hill functions as suggested by models of E. coli chemotaxis34,53, a near linear input-output curve makes best use of a given dynamic range. Furthermore, enforcing either zero slope of the input-output curve at the boundaries of the sensitive region or Hill functions as input-output curves leads to assumptions on the noise which are hard to justify biologically. Hence, Hill functions are incompatible with independent input and output noise (see Supplementary Information Sec. 1.7 for details).
How can cells actively influence and optimize their distribution of sensory input? Genetic changes in the downstream pathway and motor can clearly change chemotactic behavior and hence the experienced input stimuli. For instance, increases in the motor speed lead to larger changes in stimulus and hence broader distributions of inputs. Similarly, faster adaptation leads to narrower distributions. However, the notion that cells influence their microenvironments is most supported by the important role of niches in stem cell differentiation, cancer development, gut microbiota, and host-pathogen interactions54,55,56,57,58. Once inside the gut, E. coli related pathogen C. rodentium in mice (and similarly EPEC/EHEC in humans) injects effector proteins into the epithelial host cells. In response, these cells secrete increased levels of oxygen, allowing in return the pathogen to perform aerobic metabolism59. Hence, its aerotaxis ability, inherited from E. coli based on Aer and Tsr receptors, experiences an increased frequency of oxygen stimuli, which the pathogen actively stimulated. If we take the assumption of maximal information transmission seriously, then cells do not only actively influence but also optimize their environment.
To apply our information-theoretical approach, we analytically showed that the entire E. coli chemotaxis pathway can maximize the instantaneous mutual information between chemical concentration and motor bias despite the ultrasteep dose-response curve of the motors. Briefly, ultrasensitive motors do not restrict information transmission, since a collection of motors boosts information transmission, in addition to providing other chemotactic advantages in the soil or animal intestine60. In particular, our model identifies the number of motors and their conditional independence as key quantities to transmit large amounts of information in peritrichous bacteria.
What is the additional information at the motors used for if the ultimate behavioral output is just binary runs and tumbles? We speculate that the tumble angle, torque, and filament handedness could be regulated61,62. Indeed, real-time imaging of E.coli with fluorescent flagella showed that the tumble angles increased with the number of clockwise-turning motors, allowing for differential cell responses61. Having non-identical motors with different Hill coefficients and thresholds may further increase the information transmission (e.g. as produced by different number of FliM in the motor ring)16 but this may not be feasible in the bacterial chemotaxis pathway, as the adapted activity set the operating point of the motors. For instance, different threshold values for the motor would lead to some motor always rotating clockwise and counter-clockwise. Our model also makes the prediction that chemotactic bacteria with a single motor should prefer a relatively low Hill coefficient at the motor or multiple response regulators feeding into a motor with a high Hill coefficient to highly transmit information. This prediction could be tested with the uni-flagellated bacterial species, such as Rhodobacter sphaeroides, Pseudomonas aerugiuosa or monotrichous marine bacteria60,63. In support of our theory, R. sphaeroides is known to have multiple CheY’s64,65.
While applicable to many biological systems, our model makes a number of simplifications (in addition to assumptions on noise and receptor sensitivity). Our results are based on the independent maximizations of the receptor and motor channels. However in SI text, Sec. 4.5, we discuss the general case, providing estimes of the mutual information \( {\mathcal I} [x;z]\), between ligand input and final motor output. Our analysis suggests, once again, that high Hill coefficient for multiple motors can support high information transmission. In particular, we analytically identify two expected limits, (i) when the receptor noise is much smaller than the motor noise, we obtain \( {\mathcal I} [x;z]\approx {\mathcal I} [y;z]\), and (ii) when the motor noise is smaller than the receptor noise, we have \( {\mathcal I} [x;z]\approx {\mathcal I} [x;y]\). Our analysis over the chemotactic pathway primarily focuses on the Hill coefficient. However, the dissociation constant kdm of the motor response is known to be larger than the adapted CheYp concentration. In SI text, Sec. 4.5.4, we explicitly study the role of the dissociation constant kdm, and found a relative weak dependence of the mutual information on it. We also found that after fixing the Hill coefficient m and using the optimized output distribution of the receptor channel, the kdm that maximizes the mutual information at the motor matches the experimentally measured value (which is larger than the adapted CheYp level, see Fig. S8).
Another simplification is that our model deals with instantaneous information transmission, and hence does not explicitly include any history dependence36,37,66,67. Hence, our approach should be highly suitable for the slow genetic response in quorum sensing68,69. In this system, the input-output relation has been measured but input distributions were simply guessed, and not predicted. Another area of application is eukaryotic chemotaxis, where cells move slowly while actively shaping their chemical gradient by ligand secretion70 and degradation71. In all these examples, the input distributions and cell behaviors need to match the input-output relations to allow for optimal information gathering. Nevertheless, our model is valid for information transmission by initial transient chemotactic responses, and as this applies anywhere in the gradient, our model describes chemotaxis even including adaptation15. We expect that our model even works in relatively steep gradients, where, in addition to adaptation, long-history effects are important, such as caused by receptor saturation and rotational diffusion24. The main assumption in15 is that gradients can be linearized over the range of input distributions. However, we do not assume small Gaussian-distributed inputs. A drawback of our model is that we neglect any cell-to-cell variability, which can be substantial72,73, so that in effect our theory focuses on a certain subpopulation of cells. This cell-to-cell variability may lead to advantages in terms of bet-hedging strategies not directly related to information processing74.
In conclusion, we provided a biologically inspired adaptive algorithm with analytical solution for complex problems in information transmission in sensory systems. Future studies may need to account for time-dependencies explicitly, including adaptation both at the receptors and motors. This may be achieved by considering trajectories of molecular concentrations and/or cell behavior, which may also help establish a link between chemotactic performance (e.g. drift velocity), information transmission, and energetic cost of chemotaxis. This link may show interesting tradeoffs and new design principles29. A methodological contribution might be necessary as calculation of the mutual information based on trajectories are hampered by the high-dimensional phase space of all possible trajectories.
Materials and Methods
Maximization of mutual information with respect to input distribution and input-output curve
To maximize Eq. (2) assuming the noise in Eq. (5), we focus on the integrand \( {\mathcal L} (x,p,\bar{y},\bar{y}^{\prime} )=p\,{\log }_{2}[\frac{\sqrt{2\pi e}{\sigma }_{{\rm{T}}}}{\bar{y}^{\prime} }p]\) and use the well-known Lagrange formalism from calculus of variations, where x is the independent variable while p = p(x), \(\bar{y}\) = \(\bar{y}\)(x) and \(\bar{y}^{\prime} \) = \(\bar{y}^{\prime} \)(x) are the dependent variables. Note that p′(x) is not appearing in the Lagrangian \( {\mathcal L} \). To find the maximum of I with respect to p and \(\bar{y}\), we need to solve Eqs. (3) and (4) together.
Equation (6a) is the solution of Eq. (3), which can be rewritten through differentiation as
To derive Eq. (6b), we evaluate the following derivatives
Using Eqs (13)–(16), Eq. (4) becomes
Now assuming independent cell-external and internal noises as in Eq. (5), Eq. (17) becomes Eq. (6b).
References
Perkins, T. J. & Swain, P. S. Strategies for cellular decision-making. Mol Syst Biol 5, 326–326 (2009).
Vergassola, M., Villermaux, E. & Shraiman, B. I. ‘infotaxis’ as a strategy for searching without gradients. Nature 445, 406 (2007).
Lander, A. D. How cells know where they are. Science 339, 923–927 (2013).
Taylor, S. F., Tishby, N. & Bialek, W. Information and fitness. arXiv preprint 0712.4382 (2007).
Rivoire, O. & Leibler, S. The value of information for populations in varying environments. J Stat Phys 142, 1124–1166 (2011).
Berg, H. C. Motile behavior of bacteria. Phys Today 53, 24–29 (2000).
Endres, R. Physical Principles in Sensing and Signaling: With an Introduction to Modeling in Biology (Oxford University Press, 2013).
Shannon, C. E. A mathematical theory of communication. Bell Syst Tech J 27, 379–423 (1948).
Bowsher, C. G. & Swain, P. S. Environmental sensing, information transfer, and cellular decision-making. Curr Opin Biotech 28, 149–155 (2014).
Tkačik, G. & Walczak, A. M. Information transmission in genetic regulatory networks: a review. Journal of Physics: Condensed Matter 23, 153102 (2011).
Tkačik, G. & Bialek, W. Information processing in living systems. Annual Review of Condensed Matter Physics 7, 89–117 (2016).
Tkačik, G., Callan, C. G. Jr & Bialek, W. Information flow and optimization in transcriptional regulation. Proc Natl Acad Sci USA 105, 12265–12270 (2008).
Tkačik, G., Callan, C. G. Jr & Bialek, W. Information capacity of genetic regulatory elements. Phys Rev E 78, 011910 (2008).
Detwiler, P. B., Ramanathan, S., Sengupta, A. & Shraiman, B. I. Engineering aspects of enzymatic signal transduction: photoreceptors in the retina. Biophys J 79, 2801–2817 (2000).
Clausznitzer, D., Micali, G., Neumann, S., Sourjik, V. & Endres, R. G. Predicting chemical environments of bacteria from receptor signaling. PLoS Comput Biol 10, e1003870 (2014).
Tkačik, G., Walczak, A. M. & Bialek, W. Optimizing information flow in small genetic networks. Physical Review E 80, 031920 (2009).
Walczak, A. M., Tkačik, G. & Bialek, W. Optimizing information flow in small genetic networks. ii. feed-forward interactions. Physical Review E 81, 041905 (2010).
Tkačik, G., Walczak, A. M. & Bialek, W. Optimizing information flow in small genetic networks. iii. a self-interacting gene. Physical Review E 85, 041903 (2012).
Bernardo, J. M. Reference posterior distributions for Bayesian inference. J R Stat Soc Ser B Stat Methodol 41, 113–147 (1979).
Brunel, N. & Nadal, J. P. Mutual information, Fisher information, and population coding. Neural Comput 10, 1731–1757 (1998).
Yuan, J. & Berg, H. C. Ultrasensitivity of an adaptive bacterial motor. J Mol Biol 425, 1760–1764 (2013).
Dufour, Y. S., Fu, X., Hernandez-Nunez, L. & Emonet, T. Limits of feedback control in bacterial chemotaxis. PLoS Comput Biol 10, e1003694 (2014).
Wong-Ng, J., Melbinger, A., Celani, A. & Vergassola, M. The role of adaptation in bacterial speed races. PLoS Comput Biol 12, e1004974 (2016).
Micali, G., Colin, R., Sourjik, V. & Endres, R. G. Drift and behavior of e. coli cells. Biophysical Journal 113, 2321–2325 (2017).
Skoge, M., Meir, Y. & Wingreen, N. S. Dynamics of cooperativity in chemical sensing among cell-surface receptors. Phys Rev Lett 107, 178101–178101 (2011).
Thomas, P. J. & Eckford, A. W. Capacity of a simple intercellular signal transduction channel. IEEE Transactions on Information Theory 62, 7358–7382 (2016).
Levchenko, A. & Nemenman, I. Cellular noise and information transmission. Curr Opin Biotechnol 28, 156–164 (2014).
Mc Mahon, S. S. et al. Information theory and signal transduction systems: From molecular information processing to network inference. Semin Cell Dev Biol 35C, 98–108 (2014).
Micali, G. & Endres, R. G. Bacterial chemotaxis: information processing, thermodynamics, and behavior. Curr Opin Microbiol 30, 8–15 (2016).
Laughlin, S. A simple coding procedure enhances a neuron’s information capacity. Z Naturforsch C 36, 910–912 (1981).
Jeffreys, H. An invariant form for the prior probability in estimation problems. P Roy Soc Lond A Mat 186, 453–461 (1946).
Blahut, R. Computation of channel capacity and rate-distortion functions. IEEE transactions on Information Theory 18, 460–473 (1972).
Komorowski, M. et al. Analog nitrogen sensing in escherichia coli enables high fidelity information processing. bioRxiv 015792 (2015).
Keymer, J. E., Endres, R. G., Skoge, M., Meir, Y. & Wingreen, N. S. Chemosensing in escherichia coli: two regimes of two-state receptors. Proc Natl Acad Sci USA 103, 1786–1791 (2006).
Endres, R. G. et al. Variable sizes of Escherichia coli chemoreceptor signaling teams. Mol Syst Biol 4, 211 (2008).
Tostevin, F. & ten Wolde, P. R. Mutual information between input and output trajectories of biochemical networks. Phys Rev Lett 102, 218101–218101 (2009).
Becker, N. B., Mugler, A. & ten Wolde, P. R. Optimal prediction by cellular signaling networks. Phys Rev Lett 115, 258103 (2015).
Clausznitzer, D. & Endres, R. G. Noise characteristics of the Escherichia coli rotary motor. BMC Syst Biol 5, 151–151 (2011).
Berg, H. C. & Purcell, E. M. Physics of chemoreception. Biophys J 20, 193–219 (1977).
Salazar, M. E. & Laub, M. T. Temporal and evolutionary dynamics of two-component signaling pathways. Curr Opin Microbiol 24, 7–14 (2015).
Endres, R. G. & Wingreen, N. S. Precise adaptation in bacterial chemotaxis through “assistance neighborhoods”. Proc Natl Acad Sci USA 103, 13040–13044 (2006).
Frank, S. A. Input-output relations in biological systems: measurement, information and the Hill equation. Biol Direct 8, 31–31 (2013).
Oleksiuk, O. et al. Thermal robustness of signaling in bacterial chemotaxis. Cell 145, 312–321 (2011).
Yang, Y. & Sourjik, V. Opposite responses by different chemoreceptors set a tunable preference point in Escherichia coli pH taxis. Mol Microbiol 86, 1482–1489 (2012).
Hu, B. & Tu, Y. Behaviors and strategies of bacterial navigation in chemical and nonchemical gradients. PLoS Comput Biol 10, e1003672 (2014).
Tu, Y. & Berg, H. C. Tandem adaptation with a common design in escherichia coli chemotaxis. Journal of molecular biology 423, 782–788 (2012).
Zhang, C., He, R., Zhang, R. & Yuan, J. Motor adaptive remodeling speeds up bacterial chemotactic adaptation. Biophys J 114, 1225–1231 (2018).
Cover, T. M. & Thomas, J. A. Elements of Information Theory (Wiley Series in Telecommunications and Signal Processing), 99 edn (Wiley-Interscience, 1991).
Mears, P. J., Koirala, S., Rao, C. V., Golding, I. & Chemla, Y. R. Escherichia coli swimming is robust against variations in flagellar number. eLife 3, e01916 (2014).
Cheong, R., Rhee, A., Wang, C. J., Nemenman, I. & Levchenko, A. Information transduction capacity of noisy biochemical signaling networks. Science 334, 354–358 (2011).
Govern, C. C. & ten Wolde, P. R. Optimal resource allocation in cellular sensing systems. Proc Natl Acad Sci USA 111, 17486–17491 (2014).
Yuan, J., Branch, R. W., Hosu, B. G. & Berg, H. C. Adaptation at the output of the chemotaxis signalling pathway. Nature 484, 233–236 (2012).
Sourjik, V. & Berg, H. C. Binding of the Escherichia coli response regulator CheY to its target measured in vivo by fluorescence resonance energy transfer. Proc Natl Acad Sci USA 99, 12669–12674 (2002).
Hawkins, E. D. et al. T-cell acute leukaemia exhibits dynamic interactions with bone marrow microenvironments. Nature 538, 518 (2016).
Tanaka, M. et al. Identification of anti-cancer chemical compounds using xenopus embryos. Cancer science 107, 803–811 (2016).
Cao, Z. et al. Angiocrine factors deployed by tumor vascular niche induce b cell lymphoma invasiveness and chemoresistance. Cancer cell 25, 350–365 (2014).
Plaks, V., Kong, N. & Werb, Z. The cancer stem cell niche: how essential is the niche in regulating stemness of tumor cells? Cell stem cell 16, 225–238 (2015).
Messer, J. S., Liechty, E. R., Vogel, O. A. & Chang, E. B. Evolutionary and ecological forces that shape the bacterial communities of the human gut. Mucosal immunology 10, 567 (2017).
Berger, C. N. et al. Citrobacter rodentium subverts atp flux and cholesterol homeostasis in intestinal epithelial cells in vivo. Cell metabolism 26, 738–752 (2017).
Stocker, R. Reverse and flick: Hybrid locomotion in bacteria. Proc Natl Acad Sci USA 108, 2635–2636 (2011).
Turner, L., Ryu, W. S. & Berg, H. C. Real-time imaging of fluorescent flagellar filaments. Journal of bacteriology 182, 2793–2801 (2000).
Darnton, N. C., Turner, L., Rojevsky, S. & Berg, H. C. On torque and tumbling in swimming escherichia coli. J Bacteriol 189, 1756–1764 (2007).
Sourjik, V. & Armitage, J. P. Spatial organization in bacterial chemotaxis. EMBO J 29, 2724–2733 (2010).
Porter, S. L., Wadhams, G. H. & Armitage, J. P. Rhodobacter sphaeroides: complexity in chemotactic signalling. Trends in Microbiology 16, 251–260 (2008).
Porter, S. L., Wadhams, G. H. & Armitage, J. P. Signal processing in complex chemotaxis pathways. Nature Reviews Microbiology 9, 153–165 (2011).
Tu, Y., Shimizu, T. S. & Berg, H. C. Modeling the chemotactic response of Escherichia coli to time-varying stimuli. Proc Natl Acad Sci USA 105, 14855–14860 (2008).
Shimizu, T. S., Tu, Y. & Berg, H. C. A modular gradient-sensing network for chemotaxis in Escherichia coli revealed by responses to time-varying stimuli. Mol Syst Biol 6, 382 (2010).
Mehta, P., Goyal, S., Long, T., Bassler, B. L. & Wingreen, N. S. Information processing and signal integration in bacterial quorum sensing. Mol Syst Biol 5, 325 (2009).
Taillefumier, T. & Wingreen, N. S. Optimal census by quorum sensing. PLoS computational biology 11, e1004238 (2015).
De Palo, G., Yi, D. & Endres, R. G. A critical-like collective state leads to long-range cell communication in Dictyostelium discoideum aggregation. PLoS Biol 15, e1002602 (2017).
Tweedy, L., Knecht, D. A., Mackay, G. M. & Insall, R. H. Self-generated chemoattractant gradients: attractant depletion extends the range and robustness of chemotaxis. PLoS Biol 14, e1002404 (2016).
Colin, R., Rosazza, C., Vaknin, A. & Sourjik, V. Multiple sources of slow activity fluctuations in a bacterial chemosensory network. eLife 6, e26796 (2017).
Keegstra, J. M. et al. Phenotypic diversity and temporal variability in a bacterial signaling network revealed by single-cell fret. eLife 6, e27455 (2017).
Frankel, N. W. et al. Adaptability of non-genetic diversity in bacterial chemotaxis. eLife 3, e03526 (2014).
Sourjik, V. & Berg, H. C. Receptor sensitivity in bacterial chemotaxis. Proc Natl Acad Sci USA 99, 123–127 (2002).
Acknowledgements
We thank Chiu Fan Lee, Thomas Ouldridge, Michael Stumpf and Peter Swain for valuable comments on the manuscript, Judith Armitage for valuable discussions about Rhodobactersphaeroides, and Victor Sourjik for collaborating with us on related projects. This work was supported by European Research Council Starting-Grant N. 280492-PPHPI. Additional support came from the Biotechnological and Biological Sciences Research Council grant BB/G000131/1 (RGE) and the Swiss National Science Foundation grant nr. 31003A_169978 to Martin Ackermann (GM).
Author information
Authors and Affiliations
Contributions
Both authors (G.M. and R.G.E.) designed the study. G.M. performed the research. Both analyzed results and data, and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Micali, G., Endres, R.G. Maximal information transmission is compatible with ultrasensitive biological pathways. Sci Rep 9, 16898 (2019). https://doi.org/10.1038/s41598-019-53273-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-53273-4
This article is cited by
-
Escherichia coli chemotaxis is information limited
Nature Physics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.