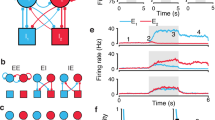
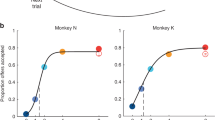
Abstract
Decisions are made with different degrees of consistency, and this consistency can be linked to the confidence that the best choice has been made. Theoretical work suggests that attractor dynamics in networks can account for choice consistency, but how this is implemented in the brain remains unclear. Here we provide evidence that the energy landscape around attractor basins in population neural activity in the prefrontal cortex reflects choice consistency. We trained two rhesus monkeys to make accept/reject decisions based on pretrained visual cues that signaled reward offers with different magnitudes and delays to reward. Monkeys made consistent decisions for very good and very bad offers, but decisions were less consistent for intermediate offers. Analysis of neural data showed that the attractor basins around patterns of activity reflecting decisions had steeper landscapes for offers that led to consistent decisions. Therefore, we provide neural evidence that energy landscapes predict decision consistency, which reflects decision confidence.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Data from the manuscript can be found in the following Figshare repository:
Wang et al. Data for ‘Attractor dynamics reflect decision confidence in macaque prefrontal cortex’. Figshare. Dataset: https://doi.org/10.6084/m9.figshare.21701282. Source data are provided with this paper.
Code availability
Custom spike sorting and data analysis codes were used. Codes for the custom spike sorter can be found at https://github.com/wangxsiyu/WangGit_Pilot_SpikeSorter.git. Custom MATLAB scripts used to perform all analysis and generate all figures can be found at https://github.com/wangxsiyu/Paper_NN-A81142A.git
References
Resulaj, A., Kiani, R., Wolpert, D. M. & Shadlen, M. N. Changes of mind in decision-making. Nature 461, 263–266 (2009).
Fleming, S. M. & Frith, C. D. The Cognitive Neuroscience of Metacognition (Springer, 2014).
Kepecs, A. & Mainen, Z. F. A computational framework for the study of confidence in humans and animals. Philos. Trans. R. Soc. Lond. B Biol. Sci. 367, 1322–1337 (2012).
Pouget, A., Drugowitsch, J. & Kepecs, A. Confidence and certainty: distinct probabilistic quantities for different goals. Nat. Neurosci. 19, 366–374 (2016).
Kiani, R. & Shadlen, M. N. Representation of confidence associated with a decision by neurons in the parietal cortex. Science 324, 759–764 (2009).
De Martino, B., Fleming, S. M., Garrett, N. & Dolan, R. J. Confidence in value-based choice. Nat. Neurosci. 16, 105–110 (2013).
Lebreton, M., Abitbol, R., Daunizeau, J. & Pessiglione, M. Automatic integration of confidence in the brain valuation signal. Nat. Neurosci. 18, 1159–1167 (2015).
Kepecs, A., Uchida, N., Zariwala, H. A. & Mainen, Z. F. Neural correlates, computation and behavioural impact of decision confidence. Nature 455, 227–231 (2008).
Prat-Ortega, G., Wimmer, K., Roxin, A. & de la Rocha, J. Flexible categorization in perceptual decision making. Nat. Commun. 12, 1283 (2021).
Machens, C. K., Romo, R. & Brody, C. D. Flexible control of mutual inhibition: a neural model of two-interval discrimination. Science 307, 1121–1124 (2005).
Wong, K. F., Huk, A. C., Shadlen, M. N. & Wang, X. J. Neural circuit dynamics underlying accumulation of time-varying evidence during perceptual decision making. Front. Comput. Neurosci. 1, 6 (2007).
Wang, X. J. Probabilistic decision making by slow reverberation in cortical circuits. Neuron 36, 955–968 (2002).
Deco, G., Rolls, E. T., Albantakis, L. & Romo, R. Brain mechanisms for perceptual and reward-related decision-making. Prog. Neurobiol. 103, 194–213 (2013).
Insabato, A., Pannunzi, M., Rolls, E. T. & Deco, G. Confidence-related decision making. J. Neurophysiol. 104, 539–547 (2010).
Wong, K. F. & Wang, X. J. A recurrent network mechanism of time integration in perceptual decisions. J. Neurosci. 26, 1314–1328 (2006).
Amit, D. J. Modeling Brain Function: The World of Attractor Neural Networks (Cambridge Univ. Press, 1989).
Inagaki, H. K., Fontolan, L., Romani, S. & Svoboda, K. Discrete attractor dynamics underlies persistent activity in the frontal cortex. Nature 566, 212–217 (2019).
Atiya, N. A. A., Rano, I., Prasad, G. & Wong-Lin, K. A neural circuit model of decision uncertainty and change-of-mind. Nat. Commun. 10, 2287 (2019).
Averbeck, B. B. Pruning recurrent neural networks replicates adolescent changes in working memory and reinforcement learning. Proc. Natl Acad. Sci. USA 119, e2121331119 (2022).
Atiya, N. A. A., Huys, Q. J. M., Dolan, R. J. & Fleming, S. M. Explaining distortions in metacognition with an attractor network model of decision uncertainty. PLoS Comput. Biol. 17, e1009201 (2021).
Marton, C. D., Schultz, S. R. & Averbeck, B. B. Learning to select actions shapes recurrent dynamics in the corticostriatal system. Neural Netw. 132, 375–393 (2020).
Finkelstein, A. et al. Attractor dynamics gate cortical information flow during decision-making. Nat. Neurosci. 24, 843–850 (2021).
Shenoy, K. V., Sahani, M. & Churchland, M. M. Cortical control of arm movements: a dynamical systems perspective. Annu. Rev. Neurosci. 36, 337–359 (2013).
Okazawa, G., Hatch, C. E., Mancoo, A., Machens, C. K. & Kiani, R. Representational geometry of perceptual decisions in the monkey parietal cortex. Cell 184, 3748–3761 (2021).
Mante, V., Sussillo, D., Shenoy, K. V. & Newsome, W. T. Context-dependent computation by recurrent dynamics in prefrontal cortex. Nature 503, 78–84 (2013).
Galgali, A. R., Sahani, M. & Mante, V. Residual dynamics resolves recurrent contributions to neural computation. Nat. Neurosci. 26, 326–338 (2023).
Chaudhuri, R., Gercek, B., Pandey, B., Peyrache, A. & Fiete, I. The intrinsic attractor manifold and population dynamics of a canonical cognitive circuit across waking and sleep. Nat. Neurosci. 22, 1512–1520 (2019).
Nair, A. et al. An approximate line attractor in the hypothalamus encodes an aggressive state. Cell 186, 178–193 (2023).
Gold, J. I. & Shadlen, M. N. Banburismus and the brain: decoding the relationship between sensory stimuli, decisions, and reward. Neuron 36, 299–308 (2002).
Drugowitsch, J., Moreno-Bote, R., Churchland, A. K., Shadlen, M. N. & Pouget, A. The cost of accumulating evidence in perceptual decision making. J. Neurosci. 32, 3612–3628 (2012).
Mitz, A. R. et al. High channel count single-unit recordings from nonhuman primate frontal cortex. J. Neurosci. Methods 289, 39–47 (2017).
Falcone, R. et al. Temporal coding of reward value in monkey ventral striatal tonically active neurons. J. Neurosci. 39, 7539–7550 (2019).
Abramson, N. Information Theory and Coding. First Edition (McGraw-Hill, 1963).
Strogatz, S. H. Nonlinear Dynamics and Chaos: With Applications to Physics, Biology, Chemistry, and Engineering (Addison-Wesley Publication, 1994).
Berlemont, K. & Nadal, J. P. Perceptual decision-making: biases in post-error reaction times explained by attractor network dynamics. J. Neurosci. 39, 833–853 (2019).
Minamimoto, T., La Camera, G. & Richmond, B. J. Measuring and modeling the interaction among reward size, delay to reward, and satiation level on motivation in monkeys. J. Neurophysiol. 101, 437–447 (2009).
Lee, E., Seo, M., Dal Monte, O. & Averbeck, B. B. Injection of a dopamine type 2 receptor antagonist into the dorsal striatum disrupts choices driven by previous outcomes, but not perceptual inference. J. Neurosci. 35, 6298–6306 (2015).
Drugowitsch, J., Mendonca, A. G., Mainen, Z. F. & Pouget, A. Learning optimal decisions with confidence. Proc. Natl Acad. Sci. USA 116, 24872–24880 (2019).
Khalvati, K., Kiani, R. & Rao, R. P. N. Bayesian inference with incomplete knowledge explains perceptual confidence and its deviations from accuracy. Nat. Commun. 12, 5704 (2021).
Lak, A. et al. Orbitofrontal cortex is required for optimal waiting based on decision confidence. Neuron 84, 190–201 (2014).
Charles, L. & Yeung, N. Dynamic sources of evidence supporting confidence judgments and error detection. J. Exp. Psychol. Hum. Percept. Perform. 45, 39–52 (2019).
Geurts, L. S., Cooke, J. R. H., van Bergen, R. S. & Jehee, J. F. M. Subjective confidence reflects representation of Bayesian probability in cortex. Nat. Hum. Behav. 6, 294–305 (2022).
Li, H. H. & Ma, W. J. Confidence reports in decision-making with multiple alternatives violate the Bayesian confidence hypothesis. Nat. Commun. 11, 2004 (2020).
Alder, W. T. & Ma, W. J. Comparing Bayesian and non-Bayesian accounts of human confidence reports. PLoS Comput. Biol. 14, e1006572 (2018).
Rolls, E. T., Grabenhorst, F. & Deco, G. Decision-making, errors, and confidence in the brain. J. Neurophysiol. 104, 2359–2374 (2010).
Seo, M., Lee, E. & Averbeck, B. B. Action selection and action value in frontal-striatal circuits. Neuron 74, 947–960 (2012).
Beck, J. M. et al. Probabilistic population codes for Bayesian decision making. Neuron 60, 1142–1152 (2008).
Zhang, K., Ginzburg, I., McNaughton, B. L. & Sejnowski, T. J. Interpreting neuronal population activity by reconstruction: unified framework with application to hippocampal place cells. J. Neurophysiol. 79, 1017–1044 (1998).
Haefner, R. M., Berkes, P. & Fiser, J. Perceptual decision-making as probabilistic inference by neural sampling. Neuron 90, 649–660 (2016).
Averbeck, B. B., Sohn, J. W. & Lee, D. Activity in prefrontal cortex during dynamic selection of action sequences. Nat. Neurosci. 9, 276–282 (2006).
Masset, P., Ott, T., Lak, A., Hirokawa, J. & Kepecs, A. Behavior- and modality-general representation of confidence in orbitofrontal cortex. Cell 182, 112–126 (2020).
Rich, E. L. & Wallis, J. D. Decoding subjective decisions from orbitofrontal cortex. Nat. Neurosci. 19, 973–980 (2016).
Enel, P., Wallis, J. D. & Rich, E. L. Stable and dynamic representations of value in the prefrontal cortex. eLife 9, e54313 (2020).
Sussillo, D. & Abbott, L. F. Generating coherent patterns of activity from chaotic neural networks. Neuron 63, 544–557 (2009).
Khona, M. & Fiete, I. R. Attractor and integrator networks in the brain. Nat. Rev. Neurosci. 23, 744–766 (2022).
Sejnowski, T. J. On the stochastic dynamics of neuronal interaction. Biol. Cybern. 22, 203–211 (1976).
Zhang, K. Representation of spatial orientation by the intrinsic dynamics of the head-direction cell ensemble: a theory. J. Neurosci. 16, 2112–2126 (1996).
Yoon, K. et al. Specific evidence of low-dimensional continuous attractor dynamics in grid cells. Nat. Neurosci. 16, 1077–1084 (2013).
Russo, A. A. et al. Motor cortex embeds muscle-like commands in an untangled population response. Neuron 97, 953–966 (2018).
Bartolo, R. & Averbeck, B. B. Prefrontal cortex predicts state switches during reversal learning. Neuron 106, 1044–1054 (2020).
Hays, A. V. Jr., Richmond, B. J. & Optican, L. M. A UNIX-based multiple process system for real-time data acquisition and control. In Proceedings of WESCON Conference 1–10 (1982); https://www.osti.gov/biblio/5213621
Costa, V. D., Kakalios, L. C. & Averbeck, B. B. Blocking serotonin but not dopamine reuptake alters neural processing during perceptual decision making. Behav. Neurosci. 130, 461–468 (2016).
Shinn, M., Lam, N. H. & Murray, J. D. A flexible framework for simulating and fitting generalized drift-diffusion models. eLife 9, e56938 (2020).
Acknowledgements
This work was supported by the intramural research program of NIMH (ZIA MH002928 to B.A., ZIA MH002619 to B.R.). The authors thank C. Robinson and Y. Wei for their assistance in spike sorting.
Author information
Authors and Affiliations
Contributions
R.F. and B.R. designed the behavioral task, R.F. collected the data, S.W. and B.A. developed the analytical approach, S.W. analyzed the data, and S.W. and B.A. wrote the manuscript, with input from R.F. and B.R.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Reconstruction of energy landscapes for intermediate cues, separated by choice.
The manifolds for reject (dotted lines) and accept (solid lines) choices for Monkey V (panel a) and Monkey W (panel b) for intermediate cues. Intermediate cues are defined as cues with p(accept) between 0.25 and 0.75. The lines are separated by the probability of accepting the offer. Positive values in the choice dimensions reflect accept choices. Data are presented as mean values ± SEM.
Extended Data Fig. 2 Position of mean activity in 1-D choice dimension for each cue.
a-b. Position in 1-D choice dimension vs. time for each cue for monkey V (panel a) and monkey W (panel b). Data are presented as mean values ± SEM. The colors reflect p(accept), red reflects cues signaling offers that monkeys mostly accept, green reflects cues signaling offers that monkeys mostly reject, and yellow reflects cues signaling offers that monkeys have intermediate probabilities of accepting. c-d. Correlation of position of mean trajectory at the mean reaction time and entropy for accept and reject decisions for monkey V (panel c) and monkey W (panel d). For monkey V, r = −0.72, p < 0.001; for monkey W, r = −0.62, p < 0.001. e-f. Significance of correlation of entropy and position of mean activity over time for monkey V (panel e) and for monkey W (panel f). A two-tailed one-sample t-test was performed at each time bin, uncorrected for multiple comparison. N = 8 for each animal. Data are presented as mean values ± SEM.
Extended Data Fig. 3 Evidence estimated from neural data vs behavior.
a. Evidence in favor of choice estimated from behavior (see Methods). N = 8 for each animal. Data are presented as mean values ± SEM. b. Correlation of the behaviorally extracted evidence \({z}_{i}\) and the evidence term fitted from the neural data \({h}_{i}\) over time. A two-tailed one-sample t-test was performed at each time bin, uncorrected for multiple comparison. N = 8 for each animal. Data are presented as mean values ± SEM. c and d. Correlations between \({z}_{i}\) and \({h}_{i}\) at the mean reaction time for monkey V and W respectively. For monkey V, r = 0.82, p < 0.001; for monkey W, r = 0.67, p < 0.001.
Extended Data Fig. 4 Parameters in the alternative evidence model.
Error bars represent standard error of the mean. For plots that show data separately for each monkey, N = 8 as SEM was computed across sessions for each animal. a. Choice-related activity, characterized as the undriven fixed points, \({x}_{0,\,j},\) over time. Note the darker color curve is accept decision and lighter color curve is reject decision, for each monkey. b. Strength of evidence-driven neural activity over time by offer cue. The colors reflect p(accept), red reflects cues signaling offers that monkeys mostly accept, green reflects cues signaling offers that monkeys mostly reject, and yellow reflects cues signaling offers that monkeys have intermediate probabilities of accepting. c. Retraction coefficients are higher for consistent choices. A one-tailed paired t-test was performed at each time bin between certain and uncertain conditions by first averaging between accept and reject conditions, uncorrected for multiple comparison. d. Retraction coefficient significantly correlates with choice entropy across the 9 cues. A two-tailed one-sample t-test was performed at each time bin, uncorrected for multiple comparison. e. Correlation between retraction coefficient and choice entropy at median reaction time for Monkey V (left) and Monkey W (right). For monkey V, r = −0.62, p < 0.001; for monkey W, r = −0.56, p < 0.001. * = 0.05, ** = 0.01, *** = 0.001.
Extended Data Fig. 5 Model coefficients estimated when activity projected on an alternative dimension.
Model coefficients estimated when activity projected on choice-dimension estimated by vector difference in means for accept vs. reject decisions. a-e. Model parameters for dynamics model after data projected onto the 1-D choice dimension defined by the difference in mean responses for accept and reject trials. N = 8 for each animal. Data are presented as mean values ± SEM. Significance values as in Fig. 5. e. For monkey V, r = −0.45, p < 0.001; for monkey W, r = −0.56, p < 0.001. f. Correlation between retraction coefficients defined in the 1-D choice dimension defined by either the SVM or the projection onto the line defined by the difference in mean responses for each condition. The retraction coefficients were strongly correlated (r = 0.78, p < 0.001).
Extended Data Fig. 6 Parameters from a 3-dimensional dynamical system model.
a-c. Correlation between the three eigenvalues of the retraction matrix for each cue, and the behavioral entropy of that cue. A two-tailed one-sample t-test was performed at each time bin, uncorrected for multiple comparison. d. Alignment of the vector connecting the 3D undriven fixed point and the 1-D choice dimension. N = 8 for each animal. Data are presented as mean values ± SEM.
Extended Data Fig. 7 Mean firing rate of individual neurons to value.
a and b. Mean firing rates of neurons in response to different offers as a function of value-coding, where 1 is preference for better options, and 0 is preference for worse options. The colors reflect p(accept), red reflects cues signaling offers that monkeys mostly accept, green reflects cues signaling offers that monkeys mostly reject, and yellow reflects cues signaling offers that monkeys have intermediate probabilities of accepting. c. Weight of each neuron on the 1-D choice dimension as a function of value coding. N = 8 for each animal. Data are presented as mean values ± SEM.
Extended Data Fig. 8 Dynamical system model fit to positive, negative and mixture neurons.
a-c. Correlation between the retraction coefficient for each cue, and the behavioral entropy of that cue. Data are presented as mean values ± SEM. A two-tailed one-sample t-test was performed at each time bin, uncorrected for multiple comparison. d-f. The retraction coefficients over time for certain vs uncertain cues. N = 8 for each animal. Data are presented as mean values ± SEM. A one-tailed paired t-test was performed at each time bin between certain and uncertain conditions by first averaging between accept and reject conditions, uncorrected for multiple comparison.
Extended Data Fig. 9 Retraction coefficients vs accept reaction time.
a. The correlations between retraction coefficients and reaction time for accept decisions over time. A two-tailed one-sample t-test was performed at each time bin, uncorrected for multiple comparison. N = 8 for each animal. Data are presented as mean values ± SEM. b. The correlations between retraction coefficients at average reaction time for reject decisions and reaction time for accept decisions (monkey V, r = −0.05, p = 0.753). c. Same as b for monkey W (monkey W, r = −0.38, p = 0.021).
Extended Data Fig. 10 Posterior predictive checks.
Energy landscape for simulated data from the linear dynamical system model. The colors reflect p(accept), red reflects cues signaling offers that monkeys mostly accept, green reflects cues signaling offers that monkeys mostly reject, and yellow reflects cues signaling offers that monkeys have intermediate probabilities of accepting. Data are presented as mean values ± SEM.
Supplementary information
Supplementary Information
Supplementary Figs. 1–10.
Source data
Source Data Fig. 1
Numerical source data.
Source Data Fig. 2
Numerical source data.
Source Data Fig. 3
Numerical source data.
Source Data Fig. 4
Numerical source data.
Source Data Fig. 5
Numerical source data.
Source Data Fig. 6
Numerical source data.
Source Data Extended Data Fig. 1
Numerical source data.
Source Data Extended Data Fig. 2
Numerical source data.
Source Data Extended Data Fig. 3
Numerical source data.
Source Data Extended Data Fig. 4
Numerical source data.
Source Data Extended Data Fig. 5
Numerical source data.
Source Data Extended Data Fig. 6
Numerical source data.
Source Data Extended Data Fig. 7
Numerical source data.
Source Data Extended Data Fig. 8
Numerical source data.
Source Data Extended Data Fig. 9
Numerical source data.
Source Data Extended Data Fig. 10
Numerical source data.
Rights and permissions
About this article
Cite this article
Wang, S., Falcone, R., Richmond, B. et al. Attractor dynamics reflect decision confidence in macaque prefrontal cortex. Nat Neurosci 26, 1970–1980 (2023). https://doi.org/10.1038/s41593-023-01445-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-023-01445-x