Abstract
The disialoganglioside GD2 is overexpressed on several solid tumors, and monoclonal antibodies targeting GD2 have substantially improved outcomes for children with high-risk neuroblastoma. However, approximately 40% of patients with neuroblastoma still relapse, and anti-GD2 has not mediated significant clinical activity in any other GD2+ malignancy. Macrophages are important mediators of anti-tumor immunity, but tumors resist macrophage phagocytosis through expression of the checkpoint molecule CD47, a so-called ‘Don’t eat me’ signal. In this study, we establish potent synergy for the combination of anti-GD2 and anti-CD47 in syngeneic and xenograft mouse models of neuroblastoma, where the combination eradicates tumors, as well as osteosarcoma and small-cell lung cancer, where the combination significantly reduces tumor burden and extends survival. This synergy is driven by two GD2-specific factors that reorient the balance of macrophage activity. Ligation of GD2 on tumor cells (a) causes upregulation of surface calreticulin, a pro-phagocytic ‘Eat me’ signal that primes cells for removal and (b) interrupts the interaction of GD2 with its newly identified ligand, the inhibitory immunoreceptor Siglec-7. This work credentials the combination of anti-GD2 and anti-CD47 for clinical translation and suggests that CD47 blockade will be most efficacious in combination with monoclonal antibodies that alter additional pro- and anti-phagocytic signals within the tumor microenvironment.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All statistics for the main and extended data figures are provided in the Supplementary Information. Source data for Figs. 1–3 and 5 and 6 and Extended Data Figs. 1–2 and 4–9 are supplied. The unprocessed gel blot image for Extended Data Fig. 3 is also provided. Source data are provided with this paper.
References
Schulz, G. et al. Detection of ganglioside GD2 in tumor tissues and sera of neuroblastoma patients. Cancer Res. 44, 5914–5920 (1984).
Long, A. H. et al. Reduction of MDSCs with all-trans retinoic acid improves CAR therapy efficacy for sarcomas. Cancer Immunol. Res. 4, 869–880 (2016).
Dobrenkov, K., Ostrovnaya, I., Gu, J., Cheung, I. Y. & Cheung, N. K. Oncotargets GD2 and GD3 are highly expressed in sarcomas of children, adolescents, and young adults. Pediatr. Blood Cancer 63, 1780–1785 (2016).
Mount, C. W. et al. Potent antitumor efficacy of anti-GD2 CAR T cells in H3-K27M+ diffuse midline gliomas. Nat. Med. 24, 572–579 (2018).
Cheresh, D. A., Rosenberg, J., Mujoo, K., Hirschowitz, L. & Reisfeld, R. A. Biosynthesis and expression of the disialoganglioside GD2, a relevant target antigen on small cell lung carcinoma for monoclonal antibody-mediated cytolysis. Cancer Res. 46, 5112–5118 (1986).
Battula, V. L. et al. Ganglioside GD2 identifies breast cancer stem cells and promotes tumorigenesis. J. Clin. Invest. 122, 2066–2078 (2012).
Yu, A. L. et al. Anti-GD2 antibody with GM-CSF, interleukin-2, and isotretinoin for neuroblastoma. N. Engl. J. Med. 363, 1324–1334 (2010).
Ladenstein, R. et al. Interleukin 2 with anti-GD2 antibody ch14.18/CHO (dinutuximab beta) in patients with high-risk neuroblastoma (HR-NBL1/SIOPEN): a multicentre, randomised, phase 3 trial. Lancet Oncol. 19, 1617–1629 (2018).
Cheung, N. K. et al. Murine anti-GD2 monoclonal antibody 3F8 combined with granulocyte-macrophage colony-stimulating factor and 13-cis-retinoic acid in high-risk patients with stage 4 neuroblastoma in first remission. J. Clin. Oncol. 30, 3264–3270 (2012).
Hobbie, W. L. et al. Late effects in survivors of tandem peripheral blood stem cell transplant for high-risk neuroblastoma. Pediatr. Blood Cancer 51, 679–683 (2008).
Suh, E. et al. Late mortality and chronic health conditions in long-term survivors of early-adolescent and young adult cancers: a retrospective cohort analysis from the Childhood Cancer Survivor Study. Lancet Oncol. 21, 421–435 (2020).
Moreno, L. et al. Outcome of children with relapsed or refractory neuroblastoma: a meta-analysis of ITCC/SIOPEN European phase II clinical trials. Pediatr. Blood Cancer 64, 25–31 (2017).
Hingorani, P. et al. Phase II study of antidisialoganglioside antibody, dinutuximab, in combination with GM-CSF in patients with recurrent osteosarcoma (AOST1421): a report from the Children’s Oncology Group. J. Clin. Oncol. 38, 10508–10508 (2020).
Edelman, M. et al. The anti-disialoganglioside (GD2) antibody dinutuximab (D) for second-line treatment (2LT) of patients (pts) with relapsed/refractory small cell lung cancer (RR SCLC): results from part II of the open-label, randomized, phase II/III distinct study. J. Clin. Oncol. 38, 9017–9017 (2020).
Grant, S. C. et al. Targeting of small-cell lung cancer using the anti-GD2 ganglioside monoclonal antibody 3F8: a pilot trial. Eur. J. Nucl. Med. 23, 145–149 (1996).
Chao, M. P. et al. Calreticulin is the dominant pro-phagocytic signal on multiple human cancers and is counterbalanced by CD47. Sci. Transl. Med. 2, 63ra94 (2010).
Jaiswal, S. et al. CD47 is upregulated on circulating hematopoietic stem cells and leukemia cells to avoid phagocytosis. Cell 138, 271–285 (2009).
Sikic, B. I. et al. First-in-human, first-in-class phase I trial of the anti-CD47 antibody Hu5F9-G4 in patients with advanced cancers. J. Clin. Oncol. 37, 946–953 (2019).
Advani, R. et al. CD47 blockade by Hu5F9-G4 and rituximab in non-Hodgkin’s lymphoma. N. Engl. J. Med. 379, 1711–1721 (2018).
Chao, M. P. et al. Anti-CD47 antibody synergizes with rituximab to promote phagocytosis and eradicate non-Hodgkin lymphoma. Cell 142, 699–713 (2010).
Desai, A. V. et al. Pharmacokinetics of the chimeric anti-GD2 antibody, ch14.18, in children with high-risk neuroblastoma. Cancer Chemother. Pharmacol. 74, 1047–1055 (2014).
Weiss, W. A., Aldape, K., Mohapatra, G., Feuerstein, B. G. & Bishop, J. M. Targeted expression of MYCN causes neuroblastoma in transgenic mice. EMBO J. 16, 2985–2995 (1997).
Sockolosky, J. T. et al. Durable antitumor responses to CD47 blockade require adaptive immune stimulation. Proc. Natl Acad. Sci. USA 113, E2646–E2654 (2016).
Willingham, S. B. et al. The CD47-signal regulatory protein alpha (SIRPa) interaction is a therapeutic target for human solid tumors. Proc. Natl Acad. Sci. USA 109, 6662–6667 (2012).
Lammie, G., Cheung, N., Gerald, W., Rosenblum, M. & Cordoncardo, C. Ganglioside gd(2) expression in the human nervous-system and in neuroblastomas - an immunohistochemical study. Int. J. Oncol. 3, 909–915 (1993).
Svennerholm, L. et al. Gangliosides and allied glycosphingolipids in human peripheral nerve and spinal cord. Biochim. Biophys. Acta 1214, 115–123 (1994).
Zhang, S. et al. Selection of tumor antigens as targets for immune attack using immunohistochemistry: I. Focus on gangliosides. Int J. Cancer 73, 42–49 (1997).
Ngamukote, S., Yanagisawa, M., Ariga, T., Ando, S. & Yu, R. K. Developmental changes of glycosphingolipids and expression of glycogenes in mouse brains. J. Neurochem. 103, 2327–2341 (2007).
Xiao, W. H., Yu, A. L. & Sorkin, L. S. Electrophysiological characteristics of primary afferent fibers after systemic administration of anti-GD2 ganglioside antibody. Pain 69, 145–151 (1997).
Sorkin, L. S. et al. Anti-GD2 with an FC point mutation reduces complement fixation and decreases antibody-induced allodynia. Pain 149, 135–142 (2010).
Hamers, F. P., Lankhorst, A. J., van Laar, T. J., Veldhuis, W. B. & Gispen, W. H. Automated quantitative gait analysis during overground locomotion in the rat: its application to spinal cord contusion and transection injuries. J. Neurotrauma 18, 187–201 (2001).
Loo, D. et al. Development of an Fc-enhanced anti-B7-H3 monoclonal antibody with potent antitumor activity. Clin. Cancer Res. 18, 3834–3845 (2012).
Crocker, P. R., Paulson, J. C. & Varki, A. Siglecs and their roles in the immune system. Nat. Rev. Immunol. 7, 255–266 (2007).
Wei, J. S. et al. Clinically relevant cytotoxic immune cell signatures and clonal expansion of T-cell receptors in high-risk MYCN-not-amplified human neuroblastoma. Clin. Cancer Res. 24, 5673–5684 (2018).
Asgharzadeh, S. et al. Clinical significance of tumor-associated inflammatory cells in metastatic neuroblastoma. J. Clin. Oncol. 30, 3525–3532 (2012).
Avril, T., Floyd, H., Lopez, F., Vivier, E. & Crocker, P. R. The membrane-proximal immunoreceptor tyrosine-based inhibitory motif is critical for the inhibitory signaling mediated by Siglecs-7 and -9, CD33-related Siglecs expressed on human monocytes and NK cells. J. Immunol. 173, 6841–6849 (2004).
Yamaji, T., Mitsuki, M., Teranishi, T. & Hashimoto, Y. Characterization of inhibitory signaling motifs of the natural killer cell receptor Siglec-7: attenuated recruitment of phosphatases by the receptor is attributed to two amino acids in the motifs. Glycobiology 15, 667–676 (2005).
Tsao, C. Y. et al. Anti-proliferative and pro-apoptotic activity of GD2 ganglioside-specific monoclonal antibody 3F8 in human melanoma cells. Oncoimmunology 4, e1023975 (2015).
Doronin, I. I. et al. Ganglioside GD2 in reception and transduction of cell death signal in tumor cells. BMC Cancer 14, 295 (2014).
Gardai, S. J. et al. Cell-surface calreticulin initiates clearance of viable or apoptotic cells through trans-activation of LRP on the phagocyte. Cell 123, 321–334 (2005).
Lo, M. et al. Effector-attenuating substitutions that maintain antibody stability and reduce toxicity in mice. J. Biol. Chem. 292, 3900–3908 (2017).
Roth, M. et al. Ganglioside GD2 as a therapeutic target for antibody-mediated therapy in patients with osteosarcoma. Cancer 120, 548–554 (2014).
Gibson, T. M. et al. Temporal patterns in the risk of chronic health conditions in survivors of childhood cancer diagnosed 1970–99: a report from the Childhood Cancer Survivor Study cohort. Lancet Oncol. 19, 1590–1601 (2018).
Jaffe, N., Puri, A. & Gelderblom, H. Osteosarcoma: evolution of treatment paradigms. Sarcoma 2013, 203531 (2013).
Majzner, R. G. et al. Assessment of programmed death-ligand 1 expression and tumor-associated immune cells in pediatric. Cancer Tissues Cancer 123, 3807–3815 (2017).
Yoshida, S. et al. Ganglioside GD2 in small cell lung cancer cell lines: enhancement of cell proliferation and mediation of apoptosis. Cancer Res. 61, 4244–4252 (2001).
Rudin, C. M. et al. Pembrolizumab or placebo plus etoposide and platinum as first-line therapy for extensive-stage small-cell lung cancer: randomized, double-blind, phase III KEYNOTE-604 study. J. Clin. Oncol. 38, 2369–2379 (2020).
Aghighi, M. et al. Magnetic resonance imaging of tumor-associated macrophages: clinical translation. Clin. Cancer Res. 24, 4110–4118 (2018).
Wiebel, M. et al. Surface expression of the immunotherapeutic target GD2 in osteosarcoma depends on cell confluency. Cancer Rep. (Hoboken) 4, e1394 (2021).
Slart, R., Yu, A. L., Yaksh, T. L. & Sorkin, L. S. An animal model of pain produced by systemic administration of an immunotherapeutic anti-ganglioside antibody. Pain 69, 119–125 (1997).
Song, L. et al. Vα24-invariant NKT cells mediate antitumor activity via killing of tumor-associated macrophages. J. Clin. Invest. 119, 1524–1536 (2009).
Chen, Y., Zhao, B. & Wang, X. Tumor infiltrating immune cells (TIICs) as a biomarker for prognosis benefits in patients with osteosarcoma. BMC Cancer 20, 1022 (2020).
Best, S. A. et al. Harnessing natural killer immunity in metastatic SCLC. J. Thorac. Oncol. 15, 1507–1521 (2020).
White, P., Liebhaber, S. A. & Cooke, N. E. 129×1/SvJ mouse strain has a novel defect in inflammatory cell recruitment. J. Immunol. 168, 869–874 (2002).
Shan, D., Ledbetter, J. A. & Press, O. W. Signaling events involved in anti-CD20-induced apoptosis of malignant human B cells. Cancer Immunol. Immunother. 48, 673–683 (2000).
Mohanty, S., Aghighi, M., Yerneni, K., Theruvath, J. L. & Daldrup-Link, H. E. Improving the efficacy of osteosarcoma therapy: combining drugs that turn cancer cell ‘don’t eat me’ signals off and ‘eat me’ signals on. Mol. Oncol. 13, 2049–2061 (2019).
Barkal, A. A. et al. CD24 signalling through macrophage Siglec-10 is a target for cancer immunotherapy. Nature 572, 392–396 (2019).
Wang, J. et al. Siglec-15 as an immune suppressor and potential target for normalization cancer immunotherapy. Nat. Med. 25, 656–666 (2019).
Siglec-15: an attractive immunotherapy target. Cancer Discov. 10, 7–8 (2020).
Fisher, G. A. et al. A phase Ib/II study of the anti-CD47 antibody magrolimab with cetuximab in solid tumor and colorectal cancer patients. J. Clin. Oncol. 38, 114–114 (2020).
Majzner, R. G. et al. Tuning the antigen density requirement for CAR T-cell activity. Cancer Discov. 10, 702–723 (2020).
Lynn, R. C. et al. c-Jun overexpression in CAR T cells induces exhaustion resistance. Nature 576, 293–300 (2019).
Weiskopf, K. et al. Engineered SIRPα variants as immunotherapeutic adjuvants to anticancer antibodies. Science 341, 88–91 (2013).
Gholamin, S. et al. Disrupting the CD47-SIRPα anti-phagocytic axis by a humanized anti-CD47 antibody is an efficacious treatment for malignant pediatric brain tumors. Sci. Transl. Med. 9, eaaf2968 (2017).
Gray, M. A. et al. Targeted glycan degradation potentiates the anticancer immune response in vivo. Nat. Chem. Biol. 16, 1376–1384 (2020).
Wu, H. W. et al. Anti-CD105 antibody eliminates tumor microenvironment cells and enhances anti-GD2 antibody immunotherapy of neuroblastoma with activated natural killer cells. Clin. Cancer Res. 25, 4761–4774 (2019).
George, J. et al. Comprehensive genomic profiles of small cell lung cancer. Nature 524, 47–53 (2015).
Acknowledgements
This work was supported by an Alex’s Lemonade Stand ‘A’ Award (R.G.M.) and National Institutes of Health P01 CA217959 (C.L.M., W.A.W. and R.G.M.). R.G.M. is the Taube Distinguished Scholar for Pediatric Immunotherapy at Stanford University School of Medicine. J.T. is supported by German Cancer Aid (Deutsche Krebshilfe) grant number P-91650709. This work was supported by the National Cancer Institute (R01-CA227942 to C.R.B., F30-CA232541 to B.A.H.S., U01-CA217864 to W.A.W. and U01-CA213273 and R35-CA231997 to J.S.), the American Cancer Society (postdoctoral fellowship to G.L.C.) and the Virginia and D. K. Ludwig Fund for Cancer Research (I.W., and M. Monje). B.A.H.S. is supported by the Stanford School of Medicine Medical Scientist Training Program (T32-GM007365). M.H.L. is supported by a Blavatnik Family Fellowship. P.L.L. is supported by the Stanford Bio-X Bowes Fellowship and the Stanford Medical Scientist Training Program. A.C.G. is supported by the Waxman Family Research Fund. We thank the Stanford Neuropathology Department for their help with immunohistochemistry. We thank K. Stegmaier and N. Mabe for providing the SY5Y-GD2-high cell line as well as expertise and advice. All cartoons were created with BioRender.com.
Author information
Authors and Affiliations
Contributions
J.T., M. Menard, B.A.H.S., M.H.L., G.L.C., G.N.D., W.W., L.K., A.D., E.S., J.L.S., A.C.G., A.B., M.T.R., S.D., S.H., A.T., J.L., P.X., J.H., N.N., A.H., A.B., R.B., K.D.M., J.G.V.-M., P.L.M., R.F. and R.G.M. designed experiments, performed experiments and/or performed data analysis. M. Monje, J.R.C., P.H.S., H.E.D.-L., I.L.W., J.S., R.M., C.R.B., W.A.W., C.L.M. and R.G.M. designed experiments and/or oversaw the research. T.C.K., E.R.B.S. and J.P. generated and provided ALX301. J.T., E.S. and R.G.M. wrote the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.G.M. and C.L.M. are founders of, hold equity in and receive consulting fees from Syncopation Life Sciences. C.L.M. is a founder of, holds equity in and receives consulting fees from Lyell Immunopharma. R.G.M. and E.S. are consultants for Lyell Immunopharma. R.G.M. is a consultant for NKarta, Illumina Radiopharmaceuticals, GammaDelta Therapeutics, Aptorum Group and Zai Labs. J.T. is a consultant for Dorian Therapeutics. C.L.M. has also received consulting fees from NeoImmune Tech, Nektar Therapeutics and Apricity Health and royalties from Juno Therapeutics for CD22-CAR. W.A.W. is a founder of, holds equity in and receives consulting fees from StemSynergy Therapeutics. R.M. is on the Board of Directors of BeyondSpring and the Scientific Advisory Boards of Coherus BioSciences, Kodikaz Therapeutic Solutions and Zenshine Pharmaceuticals. R.M. and I.W. are inventors on several patents related to CD47 cancer immunotherapy that are licensed to Gilead Sciences. J.P. is an employee and shareholder of ALX Oncology, and T.C.K. and E.R.B.S. are shareholders of ALX Oncology. J.S. receives research funding from Stemcentrx/Abbvie and Pfizer and licensed a patent to Forty Seven/Gilead on the use of CD47 blocking strategies in SCLC (with I.W.). C.R.B. is a co-founder of Redwood Biosciences (a subsidiary of Catalent), Enable Biosciences, Palleon Pharmaceuticals, InterVenn Bio, Lycia Therapeutics and OliLux Biosciences and is a member of the Board of Directors of Eli Lilly. J.R.C. is a co-founder and equity holder of xCella Biosciences, Combangio and Trapeze Therapeutics. All other authors declare no competing interests.
Peer review information
Nature Medicine thanks Leonid Metelitsa, John Anderson and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Editor recognition statement Saheli Sadanand was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 anti-GD2 and anti-CD47 synergize to promote tumor cell phagocytosis.
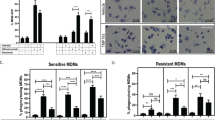
a-b, Representative Images of Incucyte based live cell phagocytosis assay. a, pHrodo red labeled KCNR (top) or CHLA255 (bottom) neuroblastoma cells were co-cultured with human blood derived macrophages in the presence of anti-GD2 mAb, anti-CD47 mAb or dual treatment. Red indicative of tumor cell phagocytosis. Images were obtained after 24 hours of co-culture. b, Magnified image of cells shown in a. c-d, Quantification of phagocytosis normalized to the phagocytosis in the untreated control for each cell line, c, KCNR (Untreated vs. GD2+CD47 p = 2.9E-14, GD2 vs. GD2+CD47 p = 5.4681E-10, CD47 vs. GD2+CD47 p = 1.13E-12) d, CHLA255 (Untreated vs. GD2+CD47 p = 3.3E-14, GD2 vs. GD2+CD47 p = 2.128E-12, CD47 vs. GD2+CD47 p = 3.3E-14). Data are mean values +/− s.e.m. of five experimental replicates. Experiment was performed one time. e, Flow-based phagocytosis assay of KCNR neuroblastoma cells co-cultured with human blood derived macrophages in the presence of different concentrations of anti-GD2 mAb, anti-CD47 mAb or dual treatment. % phagocytosing macrophages are reported. Data are mean values +/− s.d. of three experimental replicates. Experiment was performed one time. Statistical comparisons performed with one-way ANOVA with multiple comparisons correction, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns P>0.05.
Extended Data Fig. 2 Anti-CD47 and anti-GD2 synergize to mediate significant anti-tumor activity in orthotopic models of neuroblastoma.
One million KCNR neuroblastoma cells expressing GFP-luciferase were implanted into the renal capsule and treated four days later with IgG control, anti-G2 mAb, anti-CD47 (magrolimab, Hu5F9-G4) mAb or dual anti-GD2/anti-CD47 (Hu5F9-G4) every other day for three doses as in Fig. 1c. a, Quantification of tumor progression for each individual mouse as measured by flux values acquired via bioluminescence (BLI) photometry. b, BLI images of representative mice from each treatment group at different time points. Experiment was performed one time. N = 4 mice for IgG, and N = 5 for anti-GD2 per group and those groups are the same as in Fig. 1c–f. N=3 for anti-CD47 and anti-CD47 + anti-GD2 c, One million CHLA255 neuroblastoma cells expressing GFP-luciferase cells were implanted into the renal capsule in NSG mice. Four days later, mice were injected with IgG control, anti-GD2 mAb, anti-CD47 mAb or dual anti-GD2/anti-CD47 treatment every other day for three doses. d, Quantification of tumor progression for each individual mouse as measured by flux values acquired via BLI photometry. e, BLI images of representative mice from each treatment group shown in d at different time points. Red cross indicates deceased mouse. f, Survival curves for mice bearing tumors shown in d. d-f, Representative data from three independent experiments with N = 5 mice per group. All survival curves were compared using the Log-rank test (two-tailed).
Extended Data Fig. 3 Expression and affinity of ALX301, an engineered molecule capable of blocking murine CD47.
a, Schematic of ALX301: Murine IgG1 containing a N297A mutation was fused to a mutated SIRPα capable of binding murine CD47 with enhanced affinity. b, Purified ALX301 was detected on 4-20% Tris-glycine gel in non-reducing (NR) and reducing (R) buffer. ALX301 runs slightly larger than the expected 76.16 kDa (NR) and 38.038 kDa (R). c, ALX301 was immobilized on GLC sensor chip (Bio-rad). Recombinant mouse CD47 protein was injected as analyte over the chip at 5 concentrations, 3-fold dilution (100nM, 33.3nM, 11.1nM, 3.7nM, 1.2nM). Using Langmuir binding model for curve fitting, the binding of ALX301 to mouse SIRPα was determined to be 3.41nM. Association rate (ka), dissociation rate (kd), affinity (KD). b-c were performed one time.
Extended Data Fig. 4 Absence of toxicity of anti-GD2 and anti-CD47 treated mice in a model of metastatic neuroblastoma.
a, One million CHLA255 neuroblastoma cell were injected in the tail vein of the mice. After 4 days mice were treated intraperitoneally with PBS (control), anti-GD2, anti-CD47 or the combination of anti-GD2 and anti-CD47 3x/week for a total of 7 doses. b, Weight was serially recorded. c-e, CatWalk gait analysis was performed at indicated time points and c, swing speed, d, stride length and e, regularity index are reported from mice treated as in a. Data shown are mean values +/− s.e.m. n = 5 mice per group. Experiment was performed one time. f, Hematoxylin and eosin (H&E) stained sections of all organs examined were within normal limits as assessed by brightfield microscopy. Shown are sections of cerebrum, cerebellum, peripheral nerve, heart, liver, kidney, spleen, and lung of animals from mice treated as in a. Magnification: 40x, Scale bars: 20𝝁m.
Extended Data Fig. 5 Lack of synergy of anti-B7-H3 with anti-CD47 in xenograft models of neuroblastoma or osteosarcoma.
a, Graphs show flow cytometry-based quantification of phagocytosis of neuroblastoma cell lines co-cultured with human blood derived macrophages in the presence of anti-CD47 mAb, anti-B7-H3 mAb, or dual treatment, compared with untreated control; results normalized to the phagocytosis in the untreated control for each cell line and blood donor. Shown are mean values +/− s.d. of three experimental replicates. Statistical comparisons performed with one-way ANOVA with multiple comparisons correction (KCNR: control vs. anti-B7-H3 p = 0.9643, anti-CD47 vs. anti-B7-H3 and anti-CD47 p = 0.9976; CHLA255: control vs. anti-B7-H3 p>0.9999, anti-CD47 vs. anti-B7-H3 and anti-CD47 p = 0.8519). Representative data from at least three experiments with three different blood donors. Inset: B7-H3 expression as assessed by flow cytometry on respective cell lines. b, One million CHLA255 neuroblastoma cells were engrafted into NSG mice by tail-vein injection. On D+4, mice were treated with IgG control, anti-CD47 mAb, anti-B7-H3 mAb, or anti-B7-H3+anti-CD47 every other day for three doses. Quantification of tumor progression for each individual mouse as measured by flux values acquired via BLI photometry. c, BLI images of representative mice from each treatment group shown in b at one time point. d, Survival curves for mice bearing tumors shown in b. N = 5 mice per group, performed one time. Survival curves were compared using the Log-rank test (two-tailed). e, Graph shows flow cytometry-based quantification of phagocytosis of osteosarcoma cell lines co-cultured with human blood derived macrophages; results normalized to the phagocytosis in the untreated control for each cell line and blood donor. Shown are mean values +/− s.d. of three experimental replicates. Statistical comparisons performed as in a. Representative data from at least three experiments with three different blood donors. Inset: B7-H3 expression as assessed by flow cytometry on respective cell lines. f, Survival curves for mice that received hind leg injection of 1e6 MG63.3 cells and treated on D+7 with indicated antibodies 3x/week for four weeks. N = 5 mice per group, performed one time. Survival curves were compared using the Log-rank test (two-tailed). *P < 0.05, ns P>0.05.
Extended Data Fig. 6 Upregulation of ‘Eat me’ signal calreticulin and induction of cell death by ligation with anti-GD2 mAb on neuroblastoma cell lines.
a, Flow cytometric analysis of the levels of expression of GD2 on the surface of neuroblastoma cell lines CHLA255 and KCNR and their GD2-KO (B4GALNT1 KO) versions. b, Flow-based quantification of cell viability. Neuroblastoma cell lines and their GD2-KO versions were incubated with anti-GD2 mAb for the indicated times at 37 degrees and stained with DAPI. Percent of DAPI- populations were normalized to the untreated control for each cell line. Shown are mean values +/− s.d. of 6 experimental replicates. (CHLA255: 3hr p = 4.17E-08, 6hr p = 6.5044E-09, 12hr p = 1.2983E-11, 24hr p = 1.9094E-07, 48hr p = 7E-15, 72hr p = 1.2397E-08; CHLA255 GD2 KO: 3hr p = 0.3013, 6hr p = 0.506, 12hr p = 0.4125, 24hr p = 0.983, 48hr p = 0.4263, 72hr p = 0.8453; KCNR: 3hr p = 3.75E-10, 6hr p = 7.0881E-07, 12hr p = 1.1406E-07, 24hr p = 4.2986E-08, 48hr p = 2.6777E-06, 72hr p = 2.9544E-06; KCNR GD2 KO: 3hr p = 0.0059, 6hr p = 0.0083, 12hr p = 0.3223, 24hr p = 0.8197, 48hr p = 0.1443, 72hr p = 0.1002). c, Graph shows flow cytometric quantification of the expression of calreticulin (∆MFI) on the surface of live neuroblastoma cell lines treated as in b. Shown are mean values +/− s.d. of 3 experimental replicates. CHLA255: 3hr p = 6.57E-05, 6hr p = 0.0004, 12hr p = 0.0002, 24hr p = 9.2150E-06, 48hr p = 0.0167, 72hr p = 0.027; CHLA255 GD2 KO: 3hr p = 0.4701, 6hr p = 0.9563, 12hr p = 0.9024, 24hr p = 0.7235, 48hr p = 0.6415, 72hr p = 0.4069; KCNR: 3hr p = 7.41E-07, 6hr p = 7.4552E-07, 12hr p = 0.0038, 24hr p = 0.0024, 48hr p = 0.0019, 72hr p = 0.0005; KCNR GD2 KO: 3hr p = 0.671, 6hr p = 0.9445, 12hr p = 0.023, 24hr p = 0.2359, 48hr p = 0.0426, 72hr p = 0.8656. ∆MFI was calculated as the difference between the MFI in the PE channel of the calreticulin stained sample and the MFI in the PE channel of an isotype stained sample from the same experimental condition. The full time-course experiment was performed twice and the twelve-hour timepoint shown is identical to Fig. 4h. b-c, Statistical comparisons performed with the unpaired t-test, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns P>0.05.
Extended Data Fig. 7 Anti-CD47 and anti-GD2 synergize to mediate significant anti-tumor activity in xenograft model of small cell lung cancer (SCLC).
a, NCI-H69 SCLC were engrafted on both flanks of NSG mice. Quantification of tumor growth for each individual tumor was assessed by caliper measurement. b, Survival curves for mice bearing tumors shown in a. Survival curves were compared using the Log-rank test (two-tailed). Representative of two independent experiments with n = 5 mice per group.
Extended Data Fig. 8 Anti-GD2 and anti-CD47 treatment leads to an increase in macrophage infiltration and reduction of M2 macrophages.
a, Flow cytometric analysis of M2 macrophages (defined as CD163+ and CD206+, gated on CD45+, CD11b+, F4/80 + macrophages) in osteosarcoma tumors treated as described in Fig. 6, pooled results from two independent experiments. Shown are mean values +/− s.em. of 10 experimental replicates. Control vs. GD2+CD47 p = 0.0007, CD47 vs. GD2+CD47 p = 0.0738, GD2 vs. GD2+CD47 p = 0.8869. b-f, CHLA255 neuroblastoma tumor cells were engrafted in the flank and allowed to grow for 19 days before initiation of treatment with IgG control, anti-GD2 mAb, anti-CD47 mAb or dual anti-GD2/anti-CD47. After one week of treatment, tumors were harvested for immunohistochemistry (IHC) and flow cytometric analysis. b, Representative IHC images showing detection of macrophages via staining with anti-F4/80 on tumors harvested from mice treated with indicated mAbs. c, Quantification of percent of positive F4/80 staining obtained from IHC analysis. Shown are mean values +/− s.e.m. of 4-5 biologic replicates. Control vs. GD2+CD47 p = 0.0281, CD47 vs. GD2+CD47 p = 0.0415, GD2 vs. GD2+CD47 p = 0.2804. d, Quantification of percent of positive iNOS staining obtained from IHC analysis. Shown are mean values +/− s.e.m. of 4-5 biologic replicates. Control vs. GD2+CD47 p = 0.2401, CD47 vs. GD2+CD47 p = 0.413, GD2 vs. GD2+CD47 p = 0.7487. e-f, Flow cytometric analysis of M2 macrophages (defined as CD163+ and CD206+, gated on CD45+, CD11b+, F4/80 + macrophages) in CHLA255 tumors. e, Representative flow plots and f, quantification of M2 macrophages. Shown are mean values +/− s.e.m. of 4-5 biologic replicates. Control vs. GD2+CD47 p = 0.0218, CD47 vs. GD2+CD47 p = 0.9999, GD2 vs. GD2+CD47 p = 0.7102. b-f, Experiment was performed one time. All statistical comparisons were made between groups with the one-way ANOVA with correction for multiple comparisons correction, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns P>0.05.
Extended Data Fig. 9 GD2 antigen density may determine response to anti-GD2/anti-CD47.
a, Expression levels of CD47 mRNA in samples from three different datasets of primary tumors from high-risk, stage 4 neuroblastoma stratified by MYCN status (amplified vs non-amplified), age at diagnosis, and ploidy (where available). b, Expression levels of CD163, CD86, and SIGLEC7, together with CD47 and MYCN in samples from the same databases as in a grouped based on MYCN amplification status. c-d, Expression levels of CD47 and MYC family genes in samples from osteosarcoma c or SCLC d primary tumors. Samples are stratified based on high or low expression of any of the three MYC family genes: MYC, MYCL or MYCN. Dot plots represent log2 (a-c) or TPM (d) expression values. Error bars represent median ± s.d. Differences in gene expression levels between groups were calculated using the Mann-Whitney U test (unpaired, two-sided). e, Flow cytometric analysis of the expression of GD2 (top) and CD47 (bottom) on tumor cell lines used in in vivo models. f, Flow cytometric analysis of the expression of GD2 (top) and CD47 (bottom) on isogenic SH-SY5Y neuroblastoma GD2-low and GD2-high cell lines. g, Graphs show flow cytometry-based quantification of phagocytosis of SY5Y-GD2-low and SY5Y-GD2-high neuroblastoma cell lines co-cultured with human blood derived macrophages in the presence of anti-GD2 mAb, anti-CD47 mAb or dual treatment, compared with untreated control; results normalized to the phagocytosis in the untreated (UT) control for each cell line and blood donor. Shown are mean values +/− s.d. of three experimental replicates. SY5Y-GD2-high-UT vs. SY5Y-GD2-low-UT p>0.9999, SY5Y-GD2-high-GD2-treated vs. SY5Y-GD2-low-GD2-treated p = 5.1941E-07, SY5Y-GD2-high-CD47-treated vs. SY5Y-GD2-low-CD47-treated p = 0.5091, SY5Y-GD2-high-GD2+CD47-treated vs. SY5Y-GD2-low-GD2+CD47-treated p = 9.8567E-07. Representative data from two experiments performed with two different blood donors. Statistical comparisons performed with one-way ANOVA with multiple comparisons correction, *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns P>0.05.
Supplementary information
41591_2021_1625_MOESM1_ESM.pdf
Supplementary Information Representative gating strategies for flow cytometry. a, Representative gating strategy for macrophage phagocytosis assay. b, Representative gating strategy for Ly6G depletion experiments. c, Representative gating strategy for M2 macrophage phenotyping experiments.
Supplementary Data
Statistical data for main and extended data figures
Supplementary Data
Supplementary Table 1
Source data
Source Data Fig. 1
Fig. 1_Source Data
Source Data Fig. 2
Fig. 2_Source Data
Source Data Fig. 3
Fig. 3_Source Data
Source Data Fig. 5
Fig. 5_Source Data
Source Data Fig. 6
Fig. 6_Source Data
Source Data Extended Data Fig. 1
ED Fig. 1 Source Data
Source Data Extended Data Fig. 2
ED Fig. 2 Source Data
Source Data Extended Data Fig. 4
ED Fig. 4 Source Data
Source Data Extended Data Fig. 5
ED Fig. 5 Source Data
Source Data Extended Data Fig. 6
ED Fig. 6 Source Data
Source Data Extended Data Fig. 7
ED Figure 7 Source Data
Source Data Extended Data Fig. 8
ED Figure 8 Source Data
Source Data Extended Data Fig. 9
ED Figure 9 Source Data
Source Data for ED Figure3
Source Data for ED Figure3_Uncropped Gel
Rights and permissions
About this article
Cite this article
Theruvath, J., Menard, M., Smith, B.A.H. et al. Anti-GD2 synergizes with CD47 blockade to mediate tumor eradication. Nat Med 28, 333–344 (2022). https://doi.org/10.1038/s41591-021-01625-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41591-021-01625-x
This article is cited by
-
Nanoparticles in tumor microenvironment remodeling and cancer immunotherapy
Journal of Hematology & Oncology (2024)
-
Sirpα on tumor-associated myeloid cells restrains antitumor immunity in colorectal cancer independent of its interaction with CD47
Nature Cancer (2024)
-
RNAi-based drug design: considerations and future directions
Nature Reviews Drug Discovery (2024)
-
Co-targeting CD47 and VEGF elicited potent anti-tumor effects in gastric cancer
Cancer Immunology, Immunotherapy (2024)
-
Safety and antitumor activity of GD2-Specific 4SCAR-T cells in patients with glioblastoma
Molecular Cancer (2023)