Abstract
The nature of autoantigens that trigger autoimmune diseases has been much discussed, but direct biochemical identification is lacking for most. Addressing this question demands unbiased examination of the self-peptides displayed by a defined autoimmune major histocompatibility complex class II (MHC-II) molecule. Here, we examined the immunopeptidome of the pancreatic islets in non-obese diabetic mice, which spontaneously develop autoimmune diabetes based on the I-Ag7 variant of MHC-II. The relevant peptides that induced pathogenic CD4+ T cells at the initiation of diabetes derived from proinsulin. These peptides were also found in the MHC-II peptidome of the pancreatic lymph nodes and spleen. The proinsulin-derived peptides followed a trajectory from their generation and exocytosis in β cells to uptake and presentation in islets and peripheral sites. Such a pathway generated conventional epitopes but also resulted in the presentation of post-translationally modified peptides, including deamidated sequences. These analyses reveal the key features of a restricted component in the self-MHC-II peptidome that caused autoreactivity.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
01 April 2020
A Correction to this paper has been published: https://doi.org/10.1038/s41590-020-0670-0
References
Hattori, M. et al. The NOD mouse: recessive diabetogenic gene in the major histocompatibility complex. Science 231, 733–735 (1986).
Todd, J. A., Bell, J. I. & McDevitt, H. O. HLA-DQβ gene contributes to susceptibility and resistance to insulin-dependent diabetes mellitus. Nature 329, 599–604 (1987).
Acha-Orbea, H. & McDevitt, H. O. The first external domain of the nonobese diabetic mouse class II I-A β chain is unique. Proc. Natl Acad. Sci. USA 84, 2435–2439 (1987).
Miyazaki, T. et al. Direct evidence for the contribution of the unique I-ANOD to the development of insulitis in non-obese diabetic mice. Nature 345, 722–724 (1990).
Corper, A. L. et al. A structural framework for deciphering the link between I-Ag7 and autoimmune diabetes. Science 288, 505–511 (2000).
Latek, R. R. et al. Structural basis of peptide binding and presentation by the type I diabetes-associated MHC class II molecule of NOD mice. Immunity 12, 699–710 (2000).
Lee, K. H., Wucherpfennig, K. W. & Wiley, D. C. Structure of a human insulin peptide–HLA-DQ8 complex and susceptibility to type 1 diabetes. Nat. Immunol. 2, 501–507 (2001).
Suri, A., Walters, J. J., Gross, M. L. & Unanue, E. R. Natural peptides selected by diabetogenic DQ8 and murine I-Ag7 molecules show common sequence specificity. J. Clin. Invest. 115, 2268–2276 (2005).
Lund, T. et al. Prevention of insulin-dependent diabetes mellitus in non-obese diabetic mice by transgenes encoding modified I-A β-chain or normal I-E α-chain. Nature 345, 727–729 (1990).
Singer, S. M. et al. Prevention of diabetes in NOD mice by a mutated I-Ab transgene. Diabetes 47, 1570–1577 (1998).
Gioia, L. et al. Position β57 of I-Ag7 controls early anti-insulin responses in NOD mice, linking an MHC susceptibility allele to type 1 diabetes onset. Sci. Immunol. 4, eaaw6329 (2019).
Nakayama, M. et al. Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature 435, 220–223 (2005).
Daniel, D., Gill, R. G., Schloot, N. & Wegmann, D. Epitope specificity, cytokine production profile and diabetogenic activity of insulin-specific T cell clones isolated from NOD mice. Eur. J. Immunol. 25, 1056–1062 (1995).
Wegmann, D. R., Norbury-Glaser, M. & Daniel, D. Insulin-specific T cells are a predominant component of islet infiltrates in pre-diabetic NOD mice. Eur. J. Immunol. 24, 1853–1857 (1994).
Halbout, P., Briand, J.-P., Bécourt, C., Muller, S. & Boitard, C. T cell response to preproinsulin I and II in the nonobese diabetic mouse. J. Immunol. 169, 2436–2443 (2002).
Levisetti, M. G., Lewis, D. M., Suri, A. & Unanue, E. R. Weak proinsulin peptide–major histocompatibility complexes are targeted in autoimmune diabetes in mice. Diabetes 57, 1852–1860 (2008).
Mohan, J. F. et al. Unique autoreactive T cells recognize insulin peptides generated within the islets of Langerhans in autoimmune diabetes. Nat. Immunol. 11, 350–354 (2010).
Ziegler, A. G. et al. Seroconversion to multiple islet autoantibodies and risk of progression to diabetes in children. J. Am. Med. Assoc. 309, 2473–2479 (2013).
Babon, J. A. B. et al. Analysis of self-antigen specificity of islet-infiltrating T cells from human donors with type 1 diabetes. Nat. Med. 22, 1482–1487 (2016).
Michels, A. W. et al. Islet-derived CD4 T cells targeting proinsulin in human autoimmune diabetes. Diabetes 66, 722–734 (2017).
Wan, X. et al. Pancreatic islets communicate with lymphoid tissues via exocytosis of insulin peptides. Nature 560, 107–111 (2018).
Unanue, E. R. Antigen presentation in the autoimmune diabetes of the NOD mouse. Annu. Rev. Immunol. 32, 579–608 (2014).
Unanue, E. R., Ferris, S. T. & Carrero, J. A. The role of islet antigen presenting cells and the presentation of insulin in the initiation of autoimmune diabetes in the NOD mouse. Immunol. Rev. 272, 183–201 (2016).
Andreatta, M., Alvarez, B. & Nielsen, M. GibbsCluster: unsupervised clustering and alignment of peptide sequences. Nucleic Acids Res. 45, W458–W463 (2017).
Mohan, J. F., Petzold, S. J. & Unanue, E. R. Register shifting of an insulin peptide–MHC complex allows diabetogenic T cells to escape thymic deletion. J. Exp. Med. 208, 2375–2383 (2011).
Wiles, T. A. et al. An insulin-IAPP hybrid peptide is an endogenous antigen for CD4 T cells in the non-obese diabetic mouse. J. Autoimmun. 78, 11–18 (2017).
Delong, T. et al. Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science 351, 711–714 (2016).
Gagnerault, M.-C., Luan, J. J., Lotton, C. & Lepault, F. Pancreatic lymph nodes are required for priming of β cell reactive T cells in NOD mice. J. Exp. Med. 196, 369–377 (2002).
Levisetti, M. G., Suri, A., Frederick, K. & Unanue, E. R. Absence of lymph nodes in NOD mice treated with lymphotoxin-β receptor immunoglobulin protects from diabetes. Diabetes 53, 3115–3119 (2004).
Faridi, P., Purcell, A. W. & Croft, N. P. In immunopeptidomics we need a sniper instead of a shotgun. Proteomics. 18, e1700464 (2018).
Babad, J., Geliebter, A. & DiLorenzo, T. P. T-cell autoantigens in the non-obese diabetic mouse model of autoimmune diabetes. Immunology 131, 459–465 (2010).
Mohan, J. F., Calderon, B., Anderson, M. S. & Unanue, E. R. Pathogenic CD4+ T cells recognizing an unstable peptide of insulin are directly recruited into islets bypassing local lymph nodes. J. Exp. Med. 210, 2403–2414 (2013).
Chang, K. Y. & Unanue, E. R. Prediction of HLA-DQ8 β cell peptidome using a computational program and its relationship to autoreactive T cells. Int. Immunol 21, 705–713 (2009).
Aaron Wiles, T. et al. Identification of hybrid insulin peptides (HIPs) in mouse and human islets by mass spectrometry. J. Proteome Res. 18, 814–825 (2019).
Han, X., He, L., Xin, L., Shan, B. & Ma, B. PeaksPTM: mass spectrometry-based identification of peptides with unspecified modifications. J. Proteome Res. 10, 2930–2936 (2011).
Han, Y., Ma, B. & Zhang, K. SPIDER: software for protein identification from sequence tags with de novo sequencing error. J. Bioinform. Comput. Biol. 3, 697–716 (2005).
Berkers, C. R., de Jong, A., Ovaa, H. & Rodenko, B. Transpeptidation and reverse proteolysis and their consequences for immunity. Int. J. Biochem. Cell Biol. 41, 66–71 (2009).
Ebstein, F. et al. Proteasomes generate spliced epitopes by two different mechanisms and as efficiently as non-spliced epitopes. Sci. Rep. 6, 24032 (2016).
Mishto, M. & Liepe, J. Post-translational peptide splicing and T cell responses. Trends Immunol. 38, 904–915 (2017).
Mohan, J. F. & Unanue, E. R. Unconventional recognition of peptides by T cells and the implications for autoimmunity. Nat. Rev. Immunol. 12, 721–728 (2012).
Van Lummel, M. et al. Posttranslational modification of HLA-DQ binding islet autoantigens in type 1 diabetes. Diabetes 63, 237–247 (2014).
Marre, M. L. et al. Modifying enzymes are elicited by ER stress, generating epitopes that are selectively recognized by CD4+ T cells in patients with type 1 diabetes. Diabetes 67, 1356–1368 (2018).
Wright, H. T. Nonenzymatic deamidation of asparaginyl and glutaminyl residues in proteins. Crit. Rev. Biochem. Mol. Biol. 26, 1–52 (1991).
Robinson, N. E. et al. Structure-dependent nonenzymatic deamidation of glutaminyl and asparaginyl pentapeptides. J. Pept. Res. 63, 426–436 (2004).
Vader, L. W. et al. Specificity of tissue transglutaminase explains cereal toxicity in celiac disease. J. Exp. Med. 195, 643–649 (2002).
Tisch, R. et al. Immune response to glutamic acid decarboxylase correlates with insulitis in non-obese diabetic mice. Nature 366, 72–75 (1993).
Kelemen, K., Wegmann, D. R. & Hutton, J. C. T-cell epitope analysis on the autoantigen phogrin (IA-2β) in the nonobese diabetic mouse. Diabetes 50, 1729–1734 (2001).
Tai, N. et al. Microbial antigen mimics activate diabetogenic CD8 T cells in NOD mice. J. Exp. Med. 213, 2129–2146 (2016).
Hebbandi Nanjundappa, R. et al. A gut microbial mimic that hijacks diabetogenic autoreactivity to suppress colitis. Cell. 171, 655–667.e17 (2017).
So, M. et al. Proinsulin C-peptide is an autoantigen in people with type 1 diabetes. Proc. Natl Acad. Sci. USA 115, 10732–10737 (2018).
Sofron, A., Ritz, D., Neri, D. & Fugmann, T. High-resolution analysis of the murine MHC class II immunopeptidome. Eur. J. Immunol. 46, 319–328 (2016).
Acknowledgements
Members of the Unanue laboratory provided advice on many aspects of this project. We thank K. Frederick for maintaining the animal colony, P. Zakharov for assistance in prediction of the MHC-II epitopes and M. Gross for advice on mass spectrometry. This study is supported by grants from the National Institutes of Health (DK120340, DK058177 and AI114551), Juvenile Diabetes Research Foundation and Kilo Diabetes and Vascular Research Foundation.
Author information
Authors and Affiliations
Contributions
E.R.U., C.F.L., X.W. and A.N.V. planned the experiments, interpreted the results and evaluated the data. X.W. and O.J.P. prepared the biological samples. A.N.V. isolated the MHC-II molecules. C.F.L. performed the mass spectrometry experiments and analyzed the data. A.N.V. and X.W. carried out the immunological experiments. A.V.C. contributed the germ-free mice used in the experiments and reviewed and examined the paper. X.W., C.F.L. and E.R.U. wrote the paper with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Ioana Visan was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
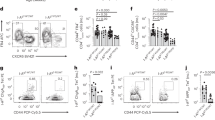
Extended Data Fig. 1 Assessing the MHCII peptidomes of the pancreatic islets, pancreatic lymph nodes and spleens from NOD mice.
a, Representative FACS plots showing the APC populations in the islets from 8-10-week old female NOD mice. The major APCs in the islet were the islet macrophages (CD45+CD11c+F4/80+) and dendritic cells (CD45+CD11c+F4/80–), along with a minor population of B cells (CD45+CD11c–F4/80–B220+). Data are representative of n = 4 independent experiment including n = 6 mice per experiment. b, Workflow for isolating the MHCII peptidome followed by mass spectrometry and immunological analyses. c, The lengths of all the peptides identified in the MHCII peptidomes from the indicated sites. Most of the peptides were 14-17 residues long, and a small number was longer than 25 residues. Data (mean) are from the mass spectrometry analysis of the MHCII peptidomes depicted in Supplementary Tables 1–3. d, Antigen presentation assay showing responses of an Ins1C-specific CD4+ T cell hybridoma to C3g7 APCs treated with or without chloroquine and pulsed with the full-length 29-residue Ins1C:33-61. The assay measures IL-2 production by the CD4+ T cell hybridoma during stimulation with the cognate antigen, assessed by the proliferation (3H incorporation) of the IL-2-depedent cell line CTLL-2 (see Methods). The Data (mean ± s.e.m.) are representative of n = 2 independent experiments with similar results.
Extended Data Fig. 2 Evaluation of T cell reactivity and relative MHCII binding affinity in peptides selected from the MHCII peptidome.
a, Pooled islet and pLN cells from 8-10-week old female NOD mice were challenged with each indicated peptide for two cycles, and ELISPOT assays were conducted to read IFN-γ production in the cells upon recalling with the same peptide (see Methods). The results show positive T cell responses to two immunogenic β-cell-derived peptides, Ins1C:33-61 and InsB:9-23, indicating the presence of the effector T cells to these peptides. No IFN-γ responses were observed to the HEL protein or the I-Ag7-binding HEL:11-25 peptide, demonstrating that the culture assay did not generate de novo T cells. Data (mean ± s.e.m.) are representative of n = 3 experiments using n = 5 mice per experiment. b, Different numbers of pre-activated HEL-reactive T cells were spiked into the normal two-cycle culture, followed by recall with the HEL peptide and ELISPOT assay. The data depict a ~1:1 recovery ratio of the spiked HEL-reactive T cells with the IL-2 spots. The assay detected as few as ~20 pre-activated HEL-reactive T cells, indicating a high sensitivity. Data (mean ± s.e.m.) are representative of n = 2 experiments using n = 8 mice per experiment. c, An example of the competitive binding assay and the presentation of the data. A standard APC line (C3g7) was cultured with 1 µM HEL:11-25 peptide together with serial dilutions of a competitor peptide: the response of a specific T cell hybridoma to HEL:11-25 was probed by standard antigen presentation assay (see Methods). In every experiment, each competitor peptides were compared to a reference peptide, g7-MIME, with known strong binding to I-Ag7. The amounts of the peptide required to compete half-maximal T cell response to HEL:11-25 (IC50) was estimated and compared to the reference peptide. A higher amount indicated a weaker binding affinity. The table (right) depicts the IC50 calculated from several representative peptides. The results are presented as the percent of reference after normalization to the reference g7-MIME peptide. Data are presentative of n = 4 independent experiments with similar results.
Extended Data Fig. 3 CD4+ T cell recognition of the InsB:15-23 register is influenced by the nature of the flanking residues.
a, Predicted I-Ag7-binding registers included in the InsB:11-25 peptide. A preferred binding register is indicated by a log of odds (LOD) score (see Methods). b, c, An InsB:11-25-specific T cell clone (clone 58) was examined for its responses to C3g7 APCs pulsed with peptides containing the InsB:15-23 binding core with varied flanking residues (grey shaded). b, Clone 58 also reacted with the InsB:12-26 peptide, an MHCII-bound sequence identified in the spleen peptidome. Mutation of the G23 into R23, an inhibitory residue, nullified the response, suggesting that the G23 was the P9 anchoring residue. Clone 58 is unreactive to InsB:12-20 or InsB:13-21. c, Comparison of T cell (clone 58) recognition between InsB:11-25 and peptides with varied residues flanking the InsB:15-23 register. The data showed the importance of the flanking residues in T cell recognition. The InsB:15-23 segment without any flanking residues did not induce any responses. Reducing, removing, or mutating the flanking residues at either the amino or carboxy end compromised the recognition to different extents. We note the role of having hydrophobic residues an the carboxy flank: changing the native FF residues into hydrophobic WW preserved the responses. Data (b,c) are representative of n = 2 independent experiments with similar results.
Extended Data Fig. 4 Analysis of the immunogenic segment in InsC peptides.
a, ELISPOT assay showing IFN-γ and IL-2 production by CD4+ T cells from 8-week old male NOD mice immunized with two Ins1C peptides lacking the complete C-terminus upon recalling with indicated relevant peptides. Responses to either Ins1C:41-55 (left) or Ins1C:37-56 (right) were indistinguishable from background (no antigen); the positive control (ConA) generated strong responses. Data (mean ± s.e.m.) summarize results from n = 3 independent experiments from n = 6 mice. b, The mass spectrometry spectrum of the synthetic citrullinated Ins1C:53-61 peptide (TLALEVArQ; the lowercase r indicates the presence of citrulline). This spectrum was distinct from the deamidated Ins1C:53-61E peptide found in the islet MHCII peptidome, confirming that the biologically induced PTM was deamidation but not citrullination.
Extended Data Fig. 5 An example of a false positive MHCII-bound HIP.
Mass spectrum of a putative Ins1C-ChgA (PQVEQLEL-WSRMDQLAK) peptide in the islet MHCII peptidome (upper) that failed to match the synthetic standard peptide (middle). Further analysis suggested a probable correct match of the putative Ins1C-ChgA HIP to an Ins1C peptide fragment with sodium adduct (lower).
Supplementary information
Supplementary Tables
Supplementary Tables 1–5.
Rights and permissions
About this article
Cite this article
Wan, X., Vomund, A.N., Peterson, O.J. et al. The MHC-II peptidome of pancreatic islets identifies key features of autoimmune peptides. Nat Immunol 21, 455–463 (2020). https://doi.org/10.1038/s41590-020-0623-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-020-0623-7
This article is cited by
-
De novo identification of CD4+ T cell epitopes
Nature Methods (2024)
-
I-Ag7 β56/57 polymorphisms regulate non-cognate negative selection to CD4+ T cell orchestrators of type 1 diabetes
Nature Immunology (2023)
-
Live enteroviruses, but not other viruses, detected in human pancreas at the onset of type 1 diabetes in the DiViD study
Diabetologia (2022)
-
T cell receptor recognition of hybrid insulin peptides bound to HLA-DQ8
Nature Communications (2021)
-
Nine residues in HLA-DQ molecules determine with susceptibility and resistance to type 1 diabetes among young children in Sweden
Scientific Reports (2021)