Abstract
Stability and current–voltage hysteresis stand as major obstacles to the commercialization of metal halide perovskites. Both phenomena have been associated with ion migration, with anecdotal evidence that stable devices yield low hysteresis. However, the underlying mechanisms of the complex stability–hysteresis link remain elusive. Here we present a multiscale diffusion framework that describes vacancy-mediated halide diffusion in polycrystalline metal halide perovskites, differentiating fast grain boundary diffusivity from volume diffusivity that is two to four orders of magnitude slower. Our results reveal an inverse relationship between the activation energies of grain boundary and volume diffusions, such that stable metal halide perovskites exhibiting smaller volume diffusivities are associated with larger grain boundary diffusivities and reduced hysteresis. The elucidation of multiscale halide diffusion in metal halide perovskites reveals complex inner couplings between ion migration in the volume of grains versus grain boundaries, which in turn can predict the stability and hysteresis of metal halide perovskites, providing a clearer path to addressing the outstanding challenges of the field.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. All other datasets generated and/or analysed during the current study are available from the corresponding authors upon request.
Code availability
The code used for the conversion of a 2D SIMS image to a matrix and subsequent integration for 2D-to-1D conversion is available from the corresponding authors upon request.
References
Tan, Z.-K. et al. Bright light-emitting diodes based on organometal halide perovskite. Nat. Nanotechnol. 9, 687–692 (2014).
Fang, Y. et al. Highly narrowband perovskite single-crystal photodetectors enabled by surface-charge recombination. Nat. Photon. 9, 679–686 (2015).
Burschka, J. et al. Sequential deposition as a route to high-performance perovskite-sensitized solar cells. Nature 499, 316–319 (2013).
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131, 6050–6051 (2009).
Lee, M. M. et al. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338, 643–647 (2012).
Li, Z. et al. Extrinsic ion migration in perovskite solar cells. Energy Environ. Sci. 10, 1234–1242 (2017).
Hassan, Y. et al. Ligand-engineered bandgap stability in mixed-halide perovskite LEDs. Nature 591, 72–77 (2021).
Mao, W. et al. Light-induced reversal of ion segregation in mixed-halide perovskites. Nat. Mater. 20, 55–61 (2021).
Saidaminov, M. I. et al. Suppression of atomic vacancies via incorporation of isovalent small ions to increase the stability of halide perovskite solar cells in ambient air. Nat. Energy 3, 648–654 (2018).
Maiti, A., Chatterjee, S., Peedikakkandy, L. & Pal, A. J. Defects and their passivation in hybrid halide perovskites toward solar cell applications. Sol. RRL 4, 2000505 (2020).
Richardson, G. et al. Can slow-moving ions explain hysteresis in the current–voltage curves of perovskite solar cells? Energy Environ. Sci. 9, 1476–1485 (2016).
Bai, S. et al. Planar perovskite solar cells with long-term stability using ionic liquid additives. Nature 571, 245–250 (2019).
Zhao, Y. et al. Suppressing ion migration in metal halide perovskite via interstitial doping with a trace amount of multivalent cations. Nat. Mater. 21, 1396–1402 (2022).
Turren-Cruz, S.-H., Hagfeldt, A. & Saliba, M. Methylammonium-free, high-performance, and stable perovskite solar cells on a planar architecture. Science 362, 449–453 (2018).
Bertoluzzi, L. et al. In situ measurement of electric-field screening in hysteresis-free PTAA/FA0.83Cs0.17Pb(I0.83Br0.17)3/C60 perovskite solar cells gives an ion mobility of ∼3 × 10–7 cm2/(V s), 2 orders of magnitude faster than reported for metal-oxide-contacted perovskite cells with hysteresis. J. Am. Chem. Soc. 140, 12775–12784 (2018).
Zhang, C. et al. Temperature-dependent electric field poling effects in CH3NH3PbI3 optoelectronic devices. J. Phys. Chem. Lett. 8, 1429–1435 (2017).
Elmelund, T., Scheidt, R. A., Seger, B. & Kamat, P. V. Bidirectional halide ion exchange in paired lead halide perovskite films with thermal activation. ACS Energy Lett. 4, 1961–1969 (2019).
Eames, C. et al. Ionic transport in hybrid lead iodide perovskite solar cells. Nat. Commun. 6, 7497 (2015).
Sajedi Alvar, M., Blom, P. W. M. & Wetzelaer, G.-J. A. H. Space-charge-limited electron and hole currents in hybrid organic-inorganic perovskites. Nat. Commun. 11, 4023 (2020).
Mehrer, H. Diffusion in Solids: Fundamentals, Methods, Materials, Diffusion-Controlled Processes Vol. 155 (Springer, 2007).
Suzuoka, T. Lattice and grain boundary diffusion in polycrystals. Trans. Jpn Inst. Met. 2, 25–32 (1961).
Mishin, Y. M. in Defect and Diffusion Forum Vol. 194 1113–1126 (Trans Tech Publications, 2001).
Joesten, R. in Diffusion, Atomic Ordering, and Mass Transport: Selected Topics in Geochemistry (ed. Ganguly, J.) 345–395 (Springer, 1991).
Laemmle, A. et al. Investigation of the diffusion behavior of sodium in Cu(In,Ga)Se2 layers. J. Appl. Phys. 115, 154501 (2014).
Phung, N. et al. The role of grain boundaries on ionic defect migration in metal halide perovskites. Adv. Energy Mater. 10, 1903735 (2020).
Park, J.-S. et al. Accumulation of deep traps at grain boundaries in halide perovskites. ACS Energy Lett. 4, 1321–1327 (2019).
Aristidou, N. et al. Fast oxygen diffusion and iodide defects mediate oxygen-induced degradation of perovskite solar cells. Nat. Commun. 8, 15218 (2017).
Shao, Y. et al. Origin and elimination of photocurrent hysteresis by fullerene passivation in CH3NH3PbI3 planar heterojunction solar cells. Nat. Commun. 5, 5784 (2014).
Kowalski, K., Bernasik, A. & Sadowski, A. Bulk and grain boundary diffusion of titanium in yttria-stabilized zirconia. J. Eur. Ceram. Soc. 20, 951–958 (2000).
Harrison, L. Influence of dislocations on diffusion kinetics in solids with particular reference to the alkali halides. J. Chem. Soc. Faraday Trans. 57, 1191–1199 (1961).
Pan, D. et al. Visualization and studies of ion-diffusion kinetics in cesium lead bromide perovskite nanowires. Nano Lett. 18, 1807–1813 (2018).
Harvey, S. P. et al. Mitigating measurement artifacts in TOF-SIMS analysis of perovskite solar cells. ACS Appl. Mater. Interfaces 11, 30911–30918 (2019).
Vaidya, M. et al. Ni tracer diffusion in CoCrFeNi and CoCrFeMnNi high entropy alloys. J. Alloy. Compd. 688, 994–1001 (2016).
Shao, Y. et al. Grain boundary dominated ion migration in polycrystalline organic–inorganic halide perovskite films. Energy Environ. Sci. 9, 1752–1759 (2016).
Azpiroz, J. M., Mosconi, E., Bisquert, J. & De Angelis, F. Defect migration in methylammonium lead iodide and its role in perovskite solar cell operation. Energy Environ. Sci. 8, 2118–2127 (2015).
Delugas, P., Caddeo, C., Filippetti, A. & Mattoni, A. Thermally activated point defect diffusion in methylammonium lead trihalide: anisotropic and ultrahigh mobility of iodine. J. Phys. Chem. Lett. 7, 2356–2361 (2016).
Oranskaia, A. et al. Halogen migration in hybrid perovskites: the organic cation matters. J. Phys. Chem. Lett. 9, 5474–5480 (2018).
Walsh, A. et al. Self-regulation mechanism for charged point defects in hybrid halide perovskites. Angew. Chem. Int. Ed. 54, 1791–1794 (2015).
Alarousu, E. et al. Ultralong radiative states in hybrid perovskite crystals: compositions for submillimeter diffusion lengths. J. Phys. Chem. Lett. 8, 4386–4390 (2017).
Sajedi Alvar, M., Blom, P. W. M. & Wetzelaer, G.-J. A. H. Device model for methylammonium lead iodide perovskite with experimentally validated ion dynamics. Adv. Electron. Mater. 6, 1900935 (2020).
Lee, J.-W. et al. The role of grain boundaries in perovskite solar cells. Mater. Today Energy 7, 149–160 (2018).
Cao, Q. et al. Efficient and stable inverted perovskite solar cells with very high fill factors via incorporation of star-shaped polymer. Sci. Adv. 7, eabg0633 (2021).
Tennyson, E. M., Doherty, T. A. S. & Stranks, S. D. Heterogeneity at multiple length scales in halide perovskite semiconductors. Nat. Rev. Mater. 4, 573–587 (2019).
Jariwala, S. et al. Local crystal misorientation influences non-radiative recombination in halide perovskites. Joule 3, 3048–3060 (2019).
Li, W. et al. The critical role of composition-dependent intragrain planar defects in the performance of MA1–xFAxPbI3 perovskite solar cells. Nat. Energy 6, 624–632 (2021).
Lee, D. S. et al. Passivation of grain boundaries by phenethylammonium in formamidinium-methylammonium lead halide perovskite solar cells. ACS Energy Lett. 3, 647–654 (2018).
Wang, H. et al. Interfacial residual stress relaxation in perovskite solar cells with improved stability. Adv. Mater. 31, 1904408 (2019).
Niu, T. et al. Stable high-performance perovskite solar cells via grain boundary passivation. Adv. Mater. 30, 1706576 (2018).
Akriti et al. Layer-by-layer anionic diffusion in two-dimensional halide perovskite vertical heterostructures. Nat. Nanotechnol. 16, 584–591 (2021).
Yun, J. S. et al. Critical role of grain boundaries for ion migration in formamidinium and methylammonium lead halide perovskite solar cells. Adv. Energy Mater. 6, 1600330 (2016).
Gao, X.-X. et al. Stable and high-efficiency methylammonium-free perovskite solar cells. Adv. Mater. 32, 1905502 (2020).
Kuno, M. & Brennan, M. C. What exactly causes light-induced halide segregation in mixed-halide perovskites? Matter 2, 21–23 (2020).
Hoke, E. T. et al. Reversible photo-induced trap formation in mixed-halide hybrid perovskites for photovoltaics. Chem. Sci. 6, 613–617 (2015).
McGovern, L. et al. Grain size influences activation energy and migration pathways in MAPbBr3 perovskite solar cells. J. Phys. Chem. Lett. 12, 2423–2428 (2021).
Acknowledgements
M.G. and A.A. acknowledge helpful discussions with D. Irving at North Carolina State University (NCSU) in relation to the GB strength model. M.G., B.G. and A.A. acknowledge support from Office of Naval Research grant N00014-20-1-2573. C.-W.H. and J.M.A. acknowledge support from the National Science Foundation Chemical Measurement and Imaging programme under grant no. CHE-1848278. A.A., L.T., G.B., K.D. and M.G. also acknowledge the support of NCSU and the Carbon Electronics cluster for start-up funding (to A.A.). K.W. acknowledges support from the US Department of Energy’s Office of Energy Efficiency and Renewable Energy under the Solar Energy Technologies Office, award no. DE-EE0009364. S.P. acknowledges support through the US Department of Energy’s Small Business Technology Transfer programme (Prime – NanoSonic Inc.), no. DE-SC0019844. NanoSonic Inc. is lead on a Small Business Innovation Research project (“prime” is commonly used for indicating “lead”). Penn State has received a subcontract on this project. SIMS measurements were performed at the Analytical Instrumentation Facility at NCSU, which is partially supported by the State of North Carolina and the National Science Foundation, and the Materials Characterization Lab at Pennsylvania State University. We acknowledge C. Zhou for providing support for SIMS measurements. M.G. and E.D.G. acknowledge financial support from the Penn State Institutes of Energy and the Environment and Office of Naval Research grant no. N00014-19-1-2453 for X-ray photoemission spectroscopy and SIMS experiments. We acknowledge the support of B. Hengstebeck for SIMS and X-ray photoemission spectroscopy measurements at Pennsylvania State University and S. Koohfar for supporting the analysis of X-ray photoemission spectroscopy results. We also acknowledge F. Castellano for providing a PL facility for superoxide measurements. This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or responsibility for the accuracy, completeness, or usefulness of any information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. Reference herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recommendation, or favoring by the United States Government or any agency thereof. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof. Abridged legal disclaimer: The views expressed herein do not necessarily represent the views of the US Department of Energy or the United States Government.
Author information
Authors and Affiliations
Contributions
A.A. and M.G. conceived the scientific framework and designed all experiments. M.G. designed and executed experimental protocols, coordinated the experimental work, performed SIMS and halide exchange measurements and analysed SIMS results. M.G., B.G., G.B., B.M.L. and L.T. prepared the MHP samples used for different measurements. B.M.L. performed additional SIMS measurements. B.G. and K.D. performed the UV–visible absorbance measurements and degradation studies. T.W. prepared the perovskite single crystals and carried out the single-crystal mechanical polishing. M.C., B.G. and K.D. performed the superoxide measurements. K.D. performed the X-ray diffraction measurements. K.W. prepared PV devices and PV stability tests, and performed time-resolved PL measurements with the supervision of S.P.; M.G. prepared the X-ray photoemission spectroscopy samples and the samples used for SIMS using the PHI nanoTOF instrument, with the supervision of E.D.G.; and C.-W.H. and J.M.A. performed the µPL measurements and analysed the data.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Juan-Pablo Correa-Baena and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–32, Equations 1–4 and Tables 1–4.
Source data
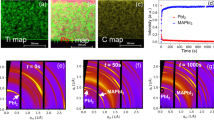
Source Data Fig. 1
The 2D SIMS map matrix and 1D SIMS profile.
Source Data Fig. 2
The 2D SIMS map matrix and 1D SIMS profile; µPL plots; temperature-dependent 1D diffusion profiles; and volume diffusion coefficients of halide in different MHP compositions.
Source Data Fig. 3
The 1D SIMS profiles and temperature-dependent GB diffusion coefficients of halide in different MHP systems, as well as extrapolated values of DGB and DV at room temperature and EV and Eg.
Source Data Fig. 4
Absorbance values for MAPbI3 and FACsRbI as a function of time at 750 nm.
Source Data Fig. 5
J–V data for MAPbI3 and FACsI PVs in forward and reverse bias, and the bandgap changes in MAPbI3/MAPbBr3 and FACsRbI/FAPbBr3 heterostructures as a function of annealing time.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghasemi, M., Guo, B., Darabi, K. et al. A multiscale ion diffusion framework sheds light on the diffusion–stability–hysteresis nexus in metal halide perovskites. Nat. Mater. 22, 329–337 (2023). https://doi.org/10.1038/s41563-023-01488-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-023-01488-2