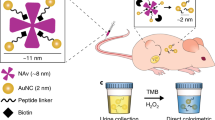
Abstract
Optical nanoparticles are promising diagnostic tools; however, their shallow optical imaging depth and slow clearance from the body have impeded their use for in vivo disease detection. To address these limitations, we develop activatable polyfluorophore nanosensors with biomarker-triggered nanoparticle-to-molecule pharmacokinetic conversion and near-infrared fluorogenic turn-on response. Activatable polyfluorophore nanosensors can accumulate at the disease site and react with disease-associated proteases to undergo in situ enzyme-catalysed depolymerization. This disease-specific interaction liberates renal-clearable fluorogenic fragments from activatable polyfluorophore nanosensors for non-invasive longitudinal urinalysis and outperforms the gold standard blood and urine assays, providing a level of sensitivity and specificity comparable to those of invasive biopsy and flow cytometry analysis. In rodent models, activatable polyfluorophore nanosensors enable ultrasensitive detection of tumours (1.6 mm diameter) and early diagnosis of acute liver allograft rejection. We anticipate that our modular nanosensor platform may be applied for early diagnosis of a range of diseases via a simple urine test.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
Source data are provided with this paper. The authors declare that data generated or analysed during this study are provided as source data or included in the Supplementary Information. Further data are available from the corresponding authors upon request.
References
Park, S. M., Aalipour, A., Vermesh, O., Yu, J. H. & Gambhir, S. S. Towards clinically translatable in vivo nanodiagnostics. Nat. Rev. Mater. 2, 17014 (2017).
Lovell, J. F. et al. Porphysome nanovesicles generated by porphyrin bilayers for use as multimodal biophotonic contrast agents. Nat. Mater. 10, 324–332 (2011).
Miao, Q. et al. Molecular afterglow imaging with bright, biodegradable polymer nanoparticles. Nat. Biotechnol. 35, 1102–1110 (2017).
Zhou, W., Han, Y., Beliveau, B. J. & Gao, X. Combining Qdot nanotechnology and DNA nanotechnology for sensitive single‐cell imaging. Adv. Mater. 32, 1908410 (2020).
Li, J. et al. Dramatic enhancement of the detection limits of bioassays via ultrafast deposition of polydopamine. Nat. Biomed. Eng. 1, 0082 (2017).
Liang, K. et al. Nanoplasmonic quantification of tumour-derived extracellular vesicles in plasma microsamples for diagnosis and treatment monitoring. Nat. Biomed. Eng. 1, 0021 (2017).
The risks of nanomaterial risk assessment. Nat. Nanotechnol. 15, 163–163 (2020).
Lu, Y., Aimetti, A. A., Langer, R. & Gu, Z. Bioresponsive materials. Nat. Rev. Mater. 2, 16075 (2016).
Du, B., Yu, M. & Zheng, J. Transport and interactions of nanoparticles in the kidneys. Nat. Rev. Mater. 3, 358–374 (2018).
Tsoi, K. M. et al. Mechanism of hard-nanomaterial clearance by the liver. Nat. Mater. 15, 1212–1221 (2016).
Poon, W. et al. Elimination pathways of nanoparticles. ACS Nano 13, 5785–5798 (2019).
Yu, M. X. & Zheng, J. Clearance pathways and tumor targeting of imaging nanoparticles. ACS Nano 9, 6655–6674 (2015).
Du, B. et al. Glomerular barrier behaves as an atomically precise bandpass filter in a sub-nanometre regime. Nat. Nanotechnol. 12, 1096–1102 (2017).
Choi, H. S. et al. Renal clearance of nanoparticles. Nat. Biotechnol. 10, 1165–1170 (2007).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010 (2017).
Perrault, S. D. & Chan, W. C. In vivo assembly of nanoparticle components to improve targeted cancer imaging. Proc. Natl Acad. Sci. USA 107, 11194–11199 (2010).
Zhao, T. et al. A transistor-like pH nanoprobe for tumour detection and image-guided surgery. Nat. Biomed. Eng. 1, 0006 (2016).
Yuan, Y. et al. Intracellular self-assembly of cyclic d-luciferin nanoparticles for persistent bioluminescence imaging of fatty acid amide hydrolase. ACS Nano 10, 7147–7153 (2016).
Gao, Y., Shi, J., Yuan, D. & Xu, B. Imaging enzyme-triggered self-assembly of small molecules inside live cells. Nat. Commun. 3, 1033 (2012).
Ye, D. et al. Bioorthogonal cyclization-mediated in situ self-assembly of small-molecule probes for imaging caspase activity in vivo. Nat. Chem. 6, 519 (2014).
Yuan, Y. et al. Furin-mediated intracellular self-assembly of olsalazine nanoparticles for enhanced magnetic resonance imaging and tumour therapy. Nat. Mater. 18, 1376–1383 (2019).
Xie, C., Zhen, X., Lyu, Y. & Pu, K. Nanoparticle regrowth enhances photoacoustic signals of semiconducting macromolecular probe for in vivo imaging. Adv. Mater. 29, 1703693 (2017).
Huynh, E. et al. In situ conversion of porphyrin microbubbles to nanoparticles for multimodality imaging. Nat. Nanotechnol. 10, 325–332 (2015).
Kwon, E. J., Dudani, J. S. & Bhatia, S. N. Ultrasensitive tumour-penetrating nanosensors of protease activity. Nat. Biomed. Eng. 1, 0054 (2017).
Loynachan, C. N. et al. Renal clearable catalytic gold nanoclusters for in vivo disease monitoring. Nat. Nanotechnol. 14, 883–890 (2019).
Jiang, X., Du, B. & Zheng, J. Glutathione-mediated biotransformation in the liver modulates nanoparticle transport. Nat. Nanotechnol. 14, 874–882 (2019).
Verdoes, M. et al. Improved quenched fluorescent probe for imaging of cysteine cathepsin activity. J. Am. Chem. Soc. 135, 14726–14730 (2013).
He, S., Li, J., Lyu, Y., Huang, J. & Pu, K. Near-infrared fluorescent macromolecular reporters for real-time imaging and urinalysis of cancer immunotherapy. J. Am. Chem. Soc. 142, 7075–7082 (2020).
Pashayan, N. & Pharoah, P. D. The challenge of early detection in cancer. Science 368, 589–590 (2020).
Wood, K. J., Bushell, A. & Hester, J. Regulatory immune cells in transplantation. Nat. Rev. Immunol. 12, 417–430 (2012).
Tiegs, G., Hentschel, J. & Wendel, A. A T cell-dependent experimental liver injury in mice inducible by concanavalin A. J. Clin. Invest. 90, 196–203 (1992).
Yamashita, J. et al. Apolipoprotein A-II suppressed concanavalin A-induced hepatitis via the inhibition of CD4 T cell function. J. Immunol. 186, 3410–3420 (2011).
Lechler, R. I., Sykes, M., Thomson, A. W. & Turka, L. A. Organ transplantation—how much of the promise has been realized? Nat. Med. 11, 605–613 (2005).
Jones, K. D. & Ferrell, L. D. Interpretation of biopsy findings in the transplant liver. Semin. Diagn. Pathol. 15, 306–317 (1998).
Portmann, B., Slapak, G., Gane, E. & Williams, R. Pathology and biopsy diagnosis of the transplanted liver. Verh. Dtsch Ges. Pathol. 79, 277–290 (1995).
Welt, S. et al. Antibody targeting in metastatic colon cancer: a phase I study of monoclonal antibody F19 against a cell-surface protein of reactive tumor stromal fibroblasts. J. Clin. Oncol. 12, 1193–1203 (1994).
Erdi, Y. E. Limits of tumor detectability in nuclear medicine and PET. Mol. Imaging Radionucl. Ther. 21, 23–28 (2012).
Serres, S. et al. Molecular MRI enables early and sensitive detection of brain metastases. Proc. Natl Acad. Sci. USA 109, 6674–6679 (2012).
Kwong, G. A. et al. Mass-encoded synthetic biomarkers for multiplexed urinary monitoring of disease. Nat. Biotechnol. 31, 63–70 (2013).
Soleimany, A. P. & Bhatia, S. N. Activity-based diagnostics: an emerging paradigm for disease detection and monitoring. Trends Mol. Med. 26, 450–468 (2020).
Aalipour, A. et al. Engineered immune cells as highly sensitive cancer diagnostics. Nat. Biotechnol. 37, 531–539 (2019).
Kirkpatrick, J. D. et al. Urinary detection of lung cancer in mice via noninvasive pulmonary protease profiling. Sci. Transl. Med. 12, eaaw0262 (2020).
Dravid, V. et al. Arterial abnormalities following orthotopic liver transplantation: arteriographic findings and correlation with Doppler sonographic findings. Am. J. Roentgenol. 163, 585–589 (1994).
Ogura, Y. et al. Radiolabeled annexin V imaging: diagnosis of allograft rejection in an experimental rodent model of liver transplantation. Radiology 214, 795–800 (2000).
Shah, A. N., Dodson, F. & Fung, J. Role of nuclear medicine in liver transplantation. Semin. Nucl. Med. 25, 36–48 (1995).
Huang, J., Li, J., Lyu, Y., Miao, Q. & Pu, K. Molecular optical imaging probes for early diagnosis of drug-induced acute kidney injury. Nat. Mater. 18, 1133–1143 (2019).
Benson, R. C. & Kues, H. A. Absorption and fluorescence properties of cyanine dyes. J. Chem. Eng. Data 22, 379–383 (1977).
Gu, K. et al. Real-time tracking and in vivo visualization of β-galactosidase activity in colorectal tumor with a ratiometric near-infrared fluorescent probe. J. Am. Chem. Soc. 138, 5334–5340 (2016).
Hu, J. J. et al. Fluorescent probe HKSOX-1 for imaging and detection of endogenous superoxide in live cells and in vivo. J. Am. Chem. Soc. 137, 6837–6843 (2015).
Hee Ryu, J. et al. Cathepsin B-sensitive nanoprobe for in vivo tumor diagnosis. J. Mater. Chem. 21, 17631–17634 (2011).
Ding, Y. et al. Tumor microenvironment-responsive multifunctional peptide coated ultrasmall gold nanoparticles and their application in cancer radiotherapy. Theranostics 10, 5195–5208 (2020).
Kamada, N. & Calne, R. Y. Orthotopic liver transplantation in the rat. Technique using cuff for portal vein anastomosis and biliary drainage. Transplantation 28, 47–50 (1979).
Guo, L. et al. Significant enhancement by anti‐ICOS antibody of suboptimal tacrolimus immunosuppression in rat liver transplantation. Liver Transplant. 10, 743–747 (2004).
Medicherla, S. et al. Topical alpha-selective p38 MAP kinase inhibition reduces acute skin inflammation in guinea pig. J. Inflamm. Res. 3, 9–16 (2010).
Wang, W. et al. Prostacyclin in endotoxemia-induced acute kidney injury: cyclooxygenase inhibition and renal prostacyclin synthase transgenic mice. Am. J. Physiol. Ren. Physiol. 293, F1131–F1136 (2007).
Acknowledgements
K.P. thanks the Ministry of Education Singapore, Academic Research Fund Tier 1 (2019-T1-002-045 RG125/19 and RT05/20) and Academic Research Fund Tier 2 (MOE2018-T2-2-042 and MOE-T2EP30220-0010) for financial support. H.W. thanks the Zhejiang Provincial Natural Science Foundation of China (LR19H160002) and the National Natural Science Foundation of China (82073296 and 81773193) for financial support.
Author information
Authors and Affiliations
Contributions
J.H. and K.P. conceived and designed the study. J.H. performed the probe synthesis and in vivo experiments. X.C., Y.J. and J.H. performed the flow cytometry and histology experiments. Y.J., C.Z. and S.H. performed cell imaging experiments. J.H., H.W. and K.P. analysed the data. J.H. and K. P. drafted the manuscript. All authors contributed to the writing of this article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Materials thanks Hak Soo Choi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–38 and Tables 1–3.
Source data
Source Data Fig. 2
Data for spectrum, selectivity studies, HPLC spectrum and so on.
Source Data Fig. 3
Data for NIRF intensity in liver and kidney, residual probes in livers and blood, clearance efficiencies and so on.
Source Data Fig. 4
Data for NIRF intensity in urine, liver and kidneys, data for liver and kidney function tests and so on.
Source Data Fig. 5
Data for NIRF intensity of liver and kidneys, signal enhancement in urine, flow cytometry and cytokines and so on.
Source Data Fig. 6
Data for NIRF intensity in urine, liver and kidneys, data for ROC curves and so on.
Rights and permissions
About this article
Cite this article
Huang, J., Chen, X., Jiang, Y. et al. Renal clearable polyfluorophore nanosensors for early diagnosis of cancer and allograft rejection. Nat. Mater. 21, 598–607 (2022). https://doi.org/10.1038/s41563-022-01224-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41563-022-01224-2
This article is cited by
-
Metabolomic machine learning predictor for diagnosis and prognosis of gastric cancer
Nature Communications (2024)
-
Artificial urinary biomarker probes for diagnosis
Nature Reviews Bioengineering (2024)
-
In situ self-assembly for cancer therapy and imaging
Nature Reviews Materials (2023)
-
Effect of nanoshell geometries, sizes, and quantum emitter parameters on the sensitivity of plasmon-exciton hybrid nanoshells for sensing application
Scientific Reports (2023)
-
Molecular imaging: design mechanism and bioapplications
Science China Chemistry (2023)