Abstract
Objectives
To evaluate the changes of retinal nerve fibre layer (RNFL) and ganglion cell layer/inner plexiform layer (GCL/IPL) with the severity of thyroid eye disease (TED).
Methods
One hundred and forty-five eyes of 75 patients with TED and 70 eyes of 35 healthy controls were included. The eyes with TED were divided into mild group (35 eyes), moderate-to-severe group (42 eyes) and DON group (68 eyes). The thickness of RNFL and GCL/IPL were measured by optic coherence tomography (OCT). Clinical activity score (CAS), best corrected visual acuity (BCVA), intraocular pressure (IOP), proptosis and mean deviation (MD) by Humphrey perimetry were assessed.
Results
The CAS had significant difference between the three groups (p < 0.001). The proptosis and IOP were significantly higher in DON group and moderate-to-severe group than mild group (p < 0.05). The MD and BCVA were significantly worse in DON group compared with mild group and moderate-to-severe group (p < 0.001). The mean GCL/IPL thickness was thinnest in DON group (p < 0.001). The mean RNFL thickness had significant difference between moderate-to-severe group and DON group (p = 0.036). The mean GCL/IPL thickness had a significant correlation with MD (r = 0.449, p < 0.001) and VA (r = −0.388, p < 0.001), whereas the mean RNFL thickness had no significant correlation with MD (p = 0.082) or VA (p = 0.226).
Conclusions
Subclinical optic neuropathy might progress in the patients with moderate-to-severe TED. OCT measurements of GCL/IPL and RNFL are useful to detect the early changes of optic nerve. The thinning of GCL/IPL might be a strong suggestion for closer vision follow-up and earlier decompression surgery.
Similar content being viewed by others
Introduction
Thyroid eye disease (TED) is the most common orbital disease, affecting 25–50% of patients with Grave’s disease [1]. The natural history of TED is characterised by an active phase and followed by an inactive phase. TED demonstrates a variable clinical presentation. While the majority of patients exhibit mild-to-moderate symptoms, about 3–7% develop vision threatening complications such as dysthyroid optic neuropathy (DON) [2].
TED is characterised by an enlargement of orbital soft tissue including extraocular muscles and orbital fat, which could lead to orbital apex compression, ischemia and optic nerve damage [2]. DON was always evaluated clinically in terms of visual acuity, colour vision, pupillary reaction and visual field (VF).
Recent years, optic coherence tomography (OCT) has emerged as a more reliable and reproducible tool for assessing the anatomy of the optic nerve. Retinal nerve fibre layer (RNFL) thickness had been proved to decrease in TED patients who had no clinical sign of DON, which indicated structural damage might occur earlier than functional impairment [3, 4]. However, the changes of RNFL thickness were not entirely consistent in previous studies because optic disc could appear as swelling, pallor or normal in different stages and severities of TED [5, 6]. Retinal ganglion cells (RGCs) measurement has shown to be a useful marker in the diagnosis and monitoring of multiple optic neuropathies, and it is thought to be more sensitive than RNFL [7]. The thickness of macular retinal layer had shown to be thinner in TED patients, which might be caused by the thinning of RGCs [5, 8]. However, the changes of RGCs thickness in different stages and severities were rarely studied.
In this study, we evaluated the thickness of ganglion cell layer/inner plexiform layer (GCL/IPL) and RNFL in TED patients and healthy controls, and analysed the changes in different disease severities.
Patients and methods
Seventy-five TED patients and 35 healthy controls were recruited in Eye & ENT Hospital, Fudan University from January 2018 to July 2019. All the eyes diagnosed with TED were divided into three groups based on their severity: (1) mild group, (2) moderate-to-severe group, (3) DON group. Mild and moderate-to-severe group were defined using the European Group on Graves’ Orbitopathy (EUGOGO) criteria [9]. DON was diagnosed based on the followings: decreased visual acuity, relative afferent pupillary defect, abnormal colour vision (Ishihara plates), compatible VF defect, disc swelling or atrophy as well as orbital apex crowding.
The inclusion criteria for healthy controls included: best corrected visual acuity (BCVA) better than 20/30, no ophthalmopathy except mild cataract, intraocular pressure (IOP) < 21 mmHg.
The common exclusion criteria included: age younger than 18 years, the history of ocular surgery or ocular trauma, evident refractive error (spherical: >−3.0 D or >+2.0 D, cylindrical: >±1.5 D), evident exposure keratopathy which could affect vision, other ophthalmopathies that could affect OCT measurements (e.g. glaucoma, optic nerve diseases, retinal diseases, obvious media opacities, obvious strabismus).
The following clinical data were obtained in TED patients: gender, age, disease duration, corticosteroid treatment, clinical activity score (CAS), BCVA (logMAR), IOP (Goldmann applanation tonometry), slit-lamp and fundus examinations, Hertel exophthalmometry, Humphrey perimetry and OCT measurements.
The study adhered to the tenets of the Declaration of Helsinki and was approved by the Ethics Committee of Fudan University. Informed consent was obtained from all patients in the study.
Visual field examination
Humphrey VF analyser was performed using the 30–2 SITA-standard protocol (Humphrey Field Analyzer 750i, Carl Zeiss Meditec Inc. Dublin). Only reliable VFs were considered (≤20% false positives or false negatives; fixation losses < 20%), and mean deviation (MD) was recorded.
OCT measurements
The TED patients and healthy controls underwent OCT scanning with a Cirrus HD-OCT (Carl Zeiss Meditec Inc. Germany). Peripapillary RNFL thickness was obtained using the optic disc cube 200 × 200 protocol and GCL/IPL thickness was obtained using the Macular Cube 512 × 128 protocol. The included scans must have a signal strength ≥6, no movement artifacts nor segmentation errors, and good centring on the fovea.
Statistical analysis
The mean values are presented with standard deviations. Student’s t test was used to compare ages between TED patients and healthy controls. Fisher’s exact was used to compare genders between patients and controls. One-way ANOVA was used to analyse the measurements among different groups. Spearman correlation test was used to analyse the correlation between measurement parameters. All differences with a value of p < 0.05 were considered statistically significant. The analyses were performed using SPSS 19.
Results
Clinical characteristics
Seventy-five TED patients (50 females, 25 males), mean age 46.4 ± 13.1 years (range 18–72 years) were included. The disease duration was 18 ± 21 months (range 3–180 months). The mean CAS was 1.7 ± 1.4 (range 0–5). 28 (37.3%) patients received steroids therapy prior to evaluation. No patient underwent radiation therapy before. 145 eyes with TED (74 right eyes and 71 left eyes) were included in the study. There were 35 eyes (17 right eyes and 18 left eyes) in mild group, 42 eyes (22 right eyes and 20 left eyes) in moderate-to-severe group and 68 eyes (35 right eyes and 33 left eyes) in DON group.
The control group included 70 eyes of 35 cases (22 females, 13 males), the mean age was 53.3 ± 11.7 years (range 26–70 years). No significant difference was found between TED patients and controls in age (p = 0.06) and gender (p = 0.7).
The clinical examinations of the TED eyes are presented in Table 1. The mean CAS had significant differences among the three groups (p < 0.001). The mean proptosis and IOP were significantly higher in DON and moderate-to-severe groups than those in mild group (p < 0.05). The MD and BCVA were significantly worse in DON group than those in mild and moderate-to-severe groups (p < 0.001). In addition, the mean IOP of healthy controls was 15.1 ± 2.1 mmHg, and it was lower than moderate-to-severe and DON groups (p < 0.05).
OCT measurements analysis
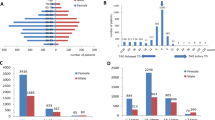
The thickness of RNFL and GCL/IPL in different groups are presented in Table 2. The mean RNFL thickness was thinnest in moderate-to-severe group and thickest in DON group, though the difference did not reach statistical significance between TED groups and control group. The mean RNFL thickness had significant difference between moderate-to-severe group and DON group (p = 0.036). The mean GCL/IPL thickness was thinnest in DON group compared with mild, moderate-to-severe and control groups (all p < 0.001). In addition, the mean GCL/IPL thickness in moderate-to-severe group was significantly thinner than that in control group (p < 0.001).
For TED eyes, the mean GCL/IPL thickness had a significant correlation with MD (r = 0.449, p < 0.001) and had a significant correlation with logMAR VA (r = −0.388, p < 0.001) (Fig. 1). However, no significant correlation was found between the mean RNFL thickness and MD (p = 0.082) or logMAR VA (p = 0.226).
Discussions
Due to the chronic damage of neural structure and the compensation of neural function, the measurement parameters of optic nerve (e.g. GCC, RNFL) and visual functions (e.g. VA, VF) are not always consistent with each other in compressive optic neuropathies (CON). In the patients with chiasmal compression, the thickness of GCC and RNFL may become thinning while the VF remained normal [10, 11]. In addition, it has been widely proved that structural damage occurred much earlier than functional impairment in glaucoma patients [12].
The inconsistency between structure and function of optic nerve also exists in TED. Wu et al. had reported that the inner intra-retinal layers including NFL, GCL/ IPL and GCC significantly decreased in TED patients without DON [8]. In our study, the TED eyes without DON were further divided into mild and moderate-to-severe subgroups based on EUGOGO criteria. The GCL/IPL thickness decreased in DON and moderate-to-severe groups compared with healthy controls, whereas the GCL/IPL thickness had no obvious decrease in mild group. The results indicated that the thinning of RGCs probably began from the moderate-to-severe stage and was earlier than the appearance of visual abnormality. The RNFL thickness had a thinning trend in moderate-to-severe group although it showed no significant difference, it indicated a subclinical atrophy of optic nerve in this stage. The average RNFL thickness increased in DON group mainly because of the oedematous optic disc.
The pathogenesis and mechanism of DON appears to be multifactorial, which might involve the optic nerve compression, elevated retrobulbar pressure, active intraorbital inflammation and vascular insufficiency. Hypoxia and ischemia may be the main reasons that retinal changes precede optic nerve changes [5, 13]. Proptosis is caused by the enlargement and congestion of orbital soft tissue in TED, and it may indicate more pressure on optic nerve and eyeball [2]. Increased IOP had been proved to play a crucial role in the thinning of RNFL and macula thickness in TED patients [5, 14]. In addition, active inflammation was considered as an important factor for optic neuropathy [15], and previous studies showed that early damage of optic nerve and macular might exist in the active TED patients [16]. In our study, the proptosis, IOP and CAS were significant higher in moderate-to-severe and DON groups compared with mild group, these might be the main causes of GCL/IPL thinning in moderate-to-severe and DON groups.
However, the eyes with moderate-to-severe TED had a relatively normal vision compared to the eyes with DON, though their RNFL and GCL/IPL thickness decreased. Consistent with previous studies, the GCL/IPL thickness were thinner in DON group compared to moderate-to-severe group in our study [17]. In addition, DON group tended to have a higher CAS than moderate-to-severe group, but there was no significant difference in proptosis or IOP between the two groups. We speculated that, in DON group, marked inflammation could damage the neural axoplasm flow and orbital blood supply during a short time, and the vision function could not compensate these damages effectively. What’s more, we speculated that slight vision damage might have happened in some eyes with moderate-to-severe TED and have improved following thyroid function recovery and/or steroids treatments, which could not be detected at this visit.
In our patients, there was a significant correlation between visual functions and GCL/IPL thickness rather than RNFL thickness. The results were similar to previous reports on chiasmal compression optic neuropathy [18]. The optic disc could be oedematous with a normal visual function at the early stage of disease, oppositely, it could present as swelling, pallor or normal with damaged visual functions later [15]. The oedema of optic disc and increase of RNFL thickness could obscure the loss in retinal nerve fibre, whereas RGCs changes could detect optic nerve injury when optic disc oedema is present and is more sensitive in detecting early optic nerve damage.
Previous studies have shown that more preserved GCC and/or RNFL thickness indicated better visual recovery. The patients with early DON could have excellent visual recovery after treatments even though the thinning RNFL and GCC had no significant improvement [19, 20]. The thinning trend of GCL/IPL might be a strong suggestion for closer vision follow-up and earlier decompression surgery in TED patients.
There were some limitations in our study. Firstly, a long-term follow-up was lack to assess the changes of GCL/IPL and RNFL thickness. Secondly, although the highest CAS score was recorded, part of the patients had received steroids therapy before the examinations and measurement parameters might be affected [2].
In conclusion, subclinical optic neuropathy may progress in the patients with moderate-to-severe TED although they have no obvious visual abnormality. The combination of structural and functional measurements at this stage could provide more information about the status of optic nerve. The thickness of GCL/IPL and RNFL is useful to monitor the potential damage of optic nerve, and the thinning of GCL/IPL might be a strong suggestion for closer vision follow-up and earlier decompression surgery.
Summary
What was known before
-
Retinal nerve fibre layer (RNFL) and RGCs could decrease in TED patients who had no clinical sign of optic neuropathy.
What this study adds
-
The changes of RNFL and RGCs thickness in different stages and severities. subclinical optic neuropathy may progress in the patients with moderate-to-severe TED.
References
Lazarus JH. Epidemiology of Graves’ orbitopathy (GO) and relationship with thyroid disease. Best Pr Res Clin Endocrinol Metab. 2012;26:273–9.
Blandford AD, Zhang D, Chundury RV, Perry JD. Dysthyroid optic neuropathy: update on pathogenesis, diagnosis, and management. Expert Rev Ophthalmol. 2017;12:111–21.
Sen E, Berker D, Elgin U, Tutuncu Y, Ozturk F, Guler S. Comparison of Optic Disc Topography in the Cases With Graves Disease and Healthy Controls. J Glaucoma. 2012;21:586–9.
Mugdha K, Kaur A, Sinha N, Saxena S. Evaluation of retinal nerve fiber layer thickness profile in thyroid ophthalmopathy without optic nerve dysfunction. Int J Ophthalmol. 2016;9:1634–7.
SayJn O, Yeter V, ArJtürk N. Optic Disc, Macula, and Retinal Nerve Fiber Layer Measurements Obtained by OCT in Thyroid-Associated Ophthalmopathy. J Neuroophthalmol. 2016;2016:9452687. https://doi.org/10.1155/2016/9452687.
Meirovitch SB, Leibovitch I, Kesler A, Varssano D, Rosenblatt A, Neuderfer M. Retina and nerve fiber layer thickness in eyes with thyroid-associated ophthalmopathy. Isr Med Assoc J. 2017;19:277–81.
Lam BL. Retinal Ganglion Cell Thickness to Assess the Optic Nerve. J Neuroophthalmol. 2015;35:107–8.
Wu Y, Tu Y, Wu C, Bao L, Wang J, Lu F, et al. Reduced macular inner retinal thickness and microvascular density in the early stage of patients with dysthyroid optic neuropathy. Eye Vis. 2020;7:1–12.
Bartalena L, Baldeschi L, Boboridis K, Eckstein A, Kahaly GJ, Marcocci C, et al. The 2016 European Thyroid Association/European Group on Graves’ Orbitopathy Guidelines for the Management of Graves’ Orbitopathy. Eur Thyroid J. 2016;1:9–26.
Yum HR, Park SH, Park HL, Shin SY. Macular Ganglion Cell Analysis Determined by Cirrus HD Optical Coherence Tomography for Early Detecting Chiasmal Compression. PLoS One. 2016;11:1–14.
Danesh-Meyer HelenV, Papchenko Taras, Savino PeterJ, Law Andrew, Evans James, Gamble GD. In Vivo Retinal Nerve Fiber Layer Thickness Measured by Optical Coherence Tomography Predicts Visual Recovery after Surgery for Parachiasmal Tumors. Invest Ophthalmol Vis Sci. 2008;49:1879–85.
Begum VU, Addepalli UK, Yadav RK, Shankar K, Senthil S, Garudadri CS, et al. Ganglion Cell-Inner Plexiform Layer Thickness of High Definition Optical Coherence Tomography in Perimetricand Preperimetric Glaucoma. Investig Ophthalmol Vis Sci. 2014;55:4768–75.
Saeed P, Rad ST, Bisschop PHLT. Dysthyroid Optic Neuropathy. Ophthalmic Plast Reconstr Surg. 2018;34:60–67.
Forte R, Bonavolontà P, Vassallo P. Evaluation of Retinal Nerve Fiber Layer with Optic Nerve Tracking Optical Coherence Tomography in Thyroid-Associated Orbitopathy. Ophthalmologica. 2010;224:116–21.
McKeag D, Lane C, Lazarus JH, Baldeschi L, Boboridis K, Dickinson AJ, et al. Clinical features of dysthyroid optic neuropathy: a European Group on Graves’ Orbitopathy (EUGOGO) survey. Br J Ophthalmol. 2007;91:455–8.
Tehrani MJ, Mahdizad Z, Kasaei A, Fard MA. Early macular and peripapillary vasculature dropout in active thyroid eye disease. Graefe’s Arch Clin Exp Ophthalmol. 2019;257:2533–40.
Zhang T, Xiao W, Ye H, Chen R, Mao Y, Yang H. Peripapillary and Macular Vessel Density in Dysthyroid Optic Neuropathy: An Optical Coherence Tomography Angiography Study. Invest Ophthalmol Vis Sci. 2019;60:1863–9.
Moura FC, Costa-Cunha LVF, Malta RFS. Relationship between visual field sensitivity loss and quadrantic macular thickness measured with Stratus-Optical coherence tomography in patients with chiasmal syndrome. Arq Bras Oftalmol. 2010;73:409–13.
Rajabi MT, Ojani M, Esfahani HR, Tabatabaei SZ, Rajabi MB, Hosseini SS. Correlation of peripapillary nerve fiber layer thickness with visual outcomes after decompression surgery in subclinical and clinical thyroid-related compressive optic neuropathy. J Curr Ophthalmol. 2019;31:86–91.
Park K, Kim Y, Woo KI. Changes in optical coherence tomography measurements after orbital wall decompression in dysthyroid optic neuropathy. Eye. 2018;32:1123–9.
Funding
National Natural Science Foundation of China [Grant number 82000940, 81800867 and 81970835]. Foundation of Shanghai Municipal Commission of Health and Family Planning [Grant No. 20164Y0144].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Guo, J., Li, X., Ma, R. et al. The changes of retinal nerve fibre layer and ganglion cell layer with different severity of thyroid eye disease. Eye 36, 129–134 (2022). https://doi.org/10.1038/s41433-021-01453-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-021-01453-w
This article is cited by
-
Dysthyroid optic neuropathy: a case series at a tertiary ophthalmic referral centre
Eye (2023)
-
Changes in retinal nerve fibre layer, ganglion cell layer and visual function in eyes with thyroid eye disease of different severities with and without orbital decompression
Eye (2023)
-
Using 24-h intraocular pressure-related patterns to identify open-angle glaucoma in thyroid eye disease
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)
-
An evaluation of corneal endothelial cell morphology in patients with thyroid-associated ophthalmopathy
International Ophthalmology (2023)
-
A diagnostic model based on color vision examination for dysthyroid optic neuropathy using Hardy-Rand-Rittler color plates
Graefe's Archive for Clinical and Experimental Ophthalmology (2023)