Abstract
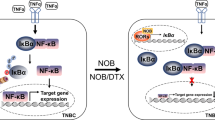
The transcription factor nuclear factor kappa B (NF-κB) is activated in human breast cancer tissues and cell lines. However, it is unclear whether NF-κB activation is a consequence of tumor formation or a contributor to tumor development. We developed a doxycycline (dox)-inducible mouse model, termed DNMP, to inhibit NF-κB activity specifically within the mammary epithelium during tumor development in the polyoma middle T oncogene (PyVT) mouse mammary tumor model. DNMP females and PyVT littermate controls were treated with dox from 4 to 12 weeks of age. We observed an increase in tumor latency and a decrease in final tumor burden in DNMP mice compared with PyVT controls. A similar effect with treatment from 8 to 12 weeks indicates that outcome is independent of effects on postnatal virgin ductal development. In both cases, DNMP mice were less likely to develop lung metastases than controls. Treatment from 8 to 9 weeks was sufficient to impact primary tumor formation. Inhibition of NF-κB increases apoptosis in hyperplastic stages of tumor development and decreases proliferation at least in part by reducing Cyclin D1 expression. To test the therapeutic potential of NF-κB inhibition, we generated palpable tumors by orthotopic injection of PyVT cells and then treated systemically with the NF-κB inhibitor thymoquinone (TQ). TQ treatment resulted in a reduction in tumor volume and weight as compared with vehicle-treated control. These data indicate that epithelial NF-κB is an active contributor to tumor progression and demonstrate that inhibition of NF-κB could have a significant therapeutic impact even at later stages of mammary tumor progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baeuerle PA, Baltimore D . (1988). I kappa B: a specific inhibitor of the NF-kappa B transcription factor. Science 242: 540–546.
Baldwin Jr AS . (1996). The NF-kappa B and I kappa B proteins: new discoveries and insights. Annu Rev Immunol 14: 649–683.
Basseres DS, Ebbs A, Levantini E, Baldwin AS . (2010). Requirement of the NF-kappaB subunit p65/RelA for K-Ras-induced lung tumorigenesis. Cancer Res 70: 3537–3546.
Baud V, Karin M . (2009). Is NF-kappaB a good target for cancer therapy? Hopes and pitfalls. Nat Rev Drug Discov 8: 33–40.
Biswas DK, Iglehart JD . (2006). Linkage between EGFR family receptors and nuclear factor kappaB (NF-kappaB) signaling in breast cancer. J Cell Physiol 209: 645–652.
Biswas DK, Shi Q, Baily S, Strickland I, Ghosh S, Pardee AB et al. (2004). NF-kappa B activation in human breast cancer specimens and its role in cell proliferation and apoptosis. Proc Natl Acad Sci USA 101: 10137–10142.
Bond M, Fabunmi RP, Baker AH, Newby AC . (1998). Synergistic upregulation of metalloproteinase-9 by growth factors and inflammatory cytokines: an absolute requirement for transcription factor NF-kappa B. FEBS Lett 435: 29–34.
Brantley DM, Chen CL, Muraoka RS, Bushdid PB, Bradberry JL, Kittrell F et al. (2001). Nuclear factor-kappaB (NF-kappaB) regulates proliferation and branching in mouse mammary epithelium. Mol Biol Cell 12: 1445–1455.
Cao Y, Karin M . (2003). NF-kappaB in mammary gland development and breast cancer. J Mammary Gland Biol Neoplasia 8: 215–223.
Cao Y, Luo JL, Karin M . (2007). IkappaB kinase alpha kinase activity is required for self-renewal of ErbB2/Her2-transformed mammary tumor-initiating cells. Proc Natl Acad Sci USA 104: 15852–15857.
Cheng DS, Han W, Chen SM, Sherrill TP, Chont M, Park GY et al. (2007). Airway epithelium controls lung inflammation and injury through the NF-kappa B pathway. J Immunol 178: 6504–6513.
Connelly L, Barham W, Pigg R, Saint-Jean L, Sherrill T, Cheng DS et al. (2010). Activation of nuclear factor kappa B in mammary epithelium promotes milk loss during mammary development and infection. J Cell Physiol 222: 73–81.
Connelly L, Robinson-Benion C, Chont M, Saint-Jean L, Li H, Polosukhin VV et al. (2007). A transgenic model reveals important roles for the NF-kappa B alternative pathway (p100/p52) in mammary development and links to tumorigenesis. J Biol Chem 282: 10028–10035.
Cude K, Wang Y, Choi HJ, Hsuan SL, Zhang H, Wang CY et al. (2007). Regulation of the G2-M cell cycle progression by the ERK5-NFkappaB signaling pathway. J Cell Biol 177: 253–264.
Everhart MB, Han W, Sherrill TP, Arutiunov M, Polosukhin VV, Burke JR et al. (2006). Duration and intensity of NF-kappaB activity determine the severity of endotoxin-induced acute lung injury. J Immunol 176: 4995–5005.
Fu M, Wang C, Li Z, Sakamaki T, Pestell RG . (2004). Minireview: cyclin D1: normal and abnormal functions. Endocrinology 145: 5439–5447.
Ghosh S, May MJ, Kopp EB . (1998). NF-kappa B and Rel proteins: evolutionarily conserved mediators of immune responses. Annu Rev Immunol 16: 225–260.
Gunther EJ, Belka GK, Wertheim GB, Wang J, Hartman JL, Boxer RB et al. (2002). A novel doxycycline-inducible system for the transgenic analysis of mammary gland biology. FASEB J 16: 283–292.
Han YP, Tuan TL, Wu H, Hughes M, Garner WL . (2001). TNF-alpha stimulates activation of pro-MMP2 in human skin through NF-(kappa)B mediated induction of MT1-MMP. J Cell Sci 114: 131–139.
Hayden MS, Ghosh S . (2008). Shared principles in NF-kappaB signaling. Cell 132: 344–362.
Hennighausen L, Robinson GW . (2005). Information networks in the mammary gland. Nat Rev Mol Cell Biol 6: 715–725.
Jafri SH, Glass J, Shi R, Zhang S, Prince M, Kleiner-Hancock H . (2010). Thymoquinone and cisplatin as a therapeutic combination in lung cancer: in vitro and in vivo. J Exp Clin Cancer Res 29: 87.
Karin M, Ben-Neriah Y . (2000). Phosphorylation meets ubiquitination: the control of NF-[kappa]B activity. Annu Rev Immunol 18: 621–663.
Lin EY, Jones JG, Li P, Zhu L, Whitney KD, Muller WJ et al. (2003). Progression to malignancy in the polyoma middle T oncoprotein mouse breast cancer model provides a reliable model for human diseases. Am J Pathol 163: 2113–2126.
Liu M, Ju X, Willmarth NE, Casimiro MC, Ojeifo J, Sakamaki T et al. (2009). Nuclear factor-kappaB enhances ErbB2-induced mammary tumorigenesis and neoangiogenesis in vivo. Am J Pathol 174: 1910–1920.
Meylan E, Dooley AL, Feldser DM, Shen L, Turk E, Ouyang C et al. (2009). Requirement for NF-kappaB signalling in a mouse model of lung adenocarcinoma. Nature 462: 104–107.
Mistry P, Deacon K, Mistry S, Blank J, Patel R . (2004). NF-kappaB promotes survival during mitotic cell cycle arrest. J Biol Chem 279: 1482–1490.
Nakshatri H, Bhat-Nakshatri P, Martin DA, Goulet Jr RJ, Sledge Jr GW . (1997). Constitutive activation of NF-kappaB during progression of breast cancer to hormone-independent growth. Mol Cell Biol 17: 3629–3639.
Patel NM, Nozaki S, Shortle NH, Bhat-Nakshatri P, Newton TR, Rice S et al. (2000). Paclitaxel sensitivity of breast cancer cells with constitutively active NF-kappaB is enhanced by IkappaBalpha super-repressor and parthenolide. Oncogene 19: 4159–4169.
Pratt MA, Tibbo E, Robertson SJ, Jansson D, Hurst K, Perez-Iratxeta C et al. (2009). The canonical NF-kappaB pathway is required for formation of luminal mammary neoplasias and is activated in the mammary progenitor population. Oncogene 28: 2710–2722.
Roy PG, Thompson AM . (2006). Cyclin D1 and breast cancer. Breast 15: 718–727.
Sethi G, Ahn KS, Aggarwal BB . (2008). Targeting nuclear factor-kappa B activation pathway by thymoquinone: role in suppression of antiapoptotic gene products and enhancement of apoptosis. Mol Cancer Res 6: 1059–1070.
Singh S, Shi Q, Bailey ST, Palczewski MJ, Pardee AB, Iglehart JD et al. (2007). Nuclear factor-kappaB activation: a molecular therapeutic target for estrogen receptor-negative and epidermal growth factor receptor family receptor-positive human breast cancer. Mol Cancer Ther 6: 1973–1982.
Sovak MA, Bellas RE, Kim DW, Zanieski GJ, Rogers AE, Traish AM et al. (1997). Aberrant nuclear factor-kappaB/Rel expression and the pathogenesis of breast cancer. J Clin Invest 100: 2952–2960.
Stathopoulos GT, Sherrill TP, Cheng DS, Scoggins RM, Han W, Polosukhin VV et al. (2007). Epithelial NF-kappaB activation promotes urethane-induced lung carcinogenesis. Proc Natl Acad Sci USA 104: 18514–18519.
Tapia MA, Gonzalez-Navarrete I, Dalmases A, Bosch M, Rodriguez-Fanjul V, Rolfe M et al. (2007). Inhibition of the canonical IKK/NF kappa B pathway sensitizes human cancer cells to doxorubicin. Cell Cycle 6: 2284–2292.
Vallabhapurapu S, Karin M . (2009). Regulation and function of NF-kappaB transcription factors in the immune system. Annu Rev Immunol 27: 693–733.
Wang TC, Cardiff RD, Zukerberg L, Lees E, Arnold A, Schmidt EV . (1994). Mammary hyperplasia and carcinoma in MMTV-cyclin D1 transgenic mice. Nature 369: 669–671.
Acknowledgements
This work was supported by NIH grant CA113734 awarded to FE Yull and a US Department of Veterans Affairs grant awarded to TS Blackwell.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Connelly, L., Barham, W., Onishko, H. et al. Inhibition of NF-kappa B activity in mammary epithelium increases tumor latency and decreases tumor burden. Oncogene 30, 1402–1412 (2011). https://doi.org/10.1038/onc.2010.521
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.521
Keywords
This article is cited by
-
NEMO- and RelA-dependent NF-κB signaling promotes small cell lung cancer
Cell Death & Differentiation (2023)
-
IKBKE phosphorylates and stabilizes Snail to promote breast cancer invasion and metastasis
Cell Death & Differentiation (2022)
-
Increased canonical NF-kappaB signaling specifically in macrophages is sufficient to limit tumor progression in syngeneic murine models of ovarian cancer
BMC Cancer (2020)
-
Cytotoxic and antiproliferative effects of thymoquinone on rat C6 glioma cells depend on oxidative stress
Molecular and Cellular Biochemistry (2019)
-
Thymoquinone inhibits cell proliferation, migration, and invasion by regulating the elongation factor 2 kinase (eEF-2K) signaling axis in triple-negative breast cancer
Breast Cancer Research and Treatment (2018)