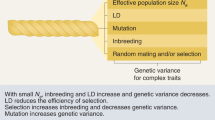
Key Points
-
Parent-of-origin effects probably contribute to the genetic architecture of complex traits, but they are rarely included in studies of genetic architecture.
-
It is crucial to distinguish between reciprocal heterozygotes when identifying parent-of-origin effects, but several phenomena besides genomic imprinting can potentially produce phenotypic differences between reciprocal heterozygotes.
-
In human studies, large-scale samples that incorporate pedigree information will be important for developing models and tools that can accommodate parent-of-origin effects into analyses.
-
Animal studies will be essential for developing a framework of DNA sequence–imprint–function relationships, particularly because parent-of-origin effects can be context dependent.
-
Research that integrates complex trait mapping results with next-generation sequencing data to identify patterns that have predictive power will be essential to advance the field.
Abstract
Parent-of-origin effects occur when the phenotypic effect of an allele depends on whether it is inherited from the mother or the father. Several phenomena can cause parent-of-origin effects, but the best characterized is parent-of-origin-dependent gene expression associated with genomic imprinting. The development of new mapping approaches applied to the growing abundance of genomic data has demonstrated that imprinted genes can be important contributors to complex trait variation. Therefore, to understand the genetic architecture and evolution of complex traits, including complex diseases and traits of agricultural importance, it is crucial to account for these parent-of-origin effects. Here, we discuss patterns of phenotypic variation associated with imprinting, evidence supporting its role in complex trait variation and approaches for identifying its molecular signatures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Reik, W. & Walter, J. Genomic imprinting: parental influence on the genome. Nature Rev. Genet. 2, 21–32 (2001).
Ferguson-Smith, A. Genomic imprinting: the emergence of an epigenetic paradigm. Nature Rev. Genet. 12, 565–575 (2011).
Tycko, B. & Morison, I. M. Physiological functions of imprinted genes. J. Cell. Physiol. 192, 245–258 (2002).
Swaney, W. T., Curley, J. P., Champagne, F. A. & Keverne, E. B. The paternally expressed gene Peg3 regulates sexual experience-dependent preferences for estrous odors. Behav. Neurosci. 122, 963–973 (2008).
Curley, J. P. Is there a genomically imprinted social brain? Bioessays 33, 662–668 (2011).
Wolf, J. B., Hager, R. & Cheverud, J. M. Genomic imprinting effects on complex traits: a phenotype-based perspective. Epigenetics 3, 295–299 (2008).
de Koning, D.-J. et al. Genome-wide scan for body composition in pigs reveals important role of imprinting. Proc. Natl Acad. Sci. USA 97, 7947–7950 (2000).
Wolf, J. B., Cheverud, J. M., Roseman, C. & Hager, R. Genome-wide analysis reveals a complex pattern of genomic imprinting in mice. PLoS Genet. 4, e1000091 (2008). This study developed the framework (outlined in box 1 ) to describe the complex patterns of imprinting that have been identified, including the first description of the patterns of polar underdominance and bipolar dominance imprinting.
Cheverud, J. M. et al. Genomic imprinting effects on adult body composition in mice. Proc. Natl Acad. Sci. USA 105, 4253–4258 (2008).
Lawson, H. A. et al. The importance of context to the genetic architecture of diabetes-related traits is revealed in a genome-wide scan of a LG/J × SM/J murine model. Mamm. Genome 22, 197–208 (2011).
Morison, I. M., Ramsay, J. P. & Spencer, H. G. A census of mammalian imprinting. Trends Genet. 21, 457–465 (2005).
Raissig, M. T., Baroux, C. & Grossniklaus, U. Regulation and flexibility of genomic imprinting during seed development. Plant Cell 23, 16–26 (2011).
Pearson, C. E. Slipping while sleeping? Trinucleotide repeat expansions in germ cells. Trends Mol. Med. 9, 490–495 (2003).
Tome, S. et al. Maternal germline-specific effect of DNA ligase I on CTG/CAG instability. Hum. Mol. Genet. 20, 2131–2143 (2011).
Haley, C. S., Knott, S. A. & Elsen, J.-M. Mapping quantitative trait loci in crosses between outbred lines using least squares. Genetics 136, 1195–1207 (1994).
de Koning, D.-J., Bovenhuis, H. & Van Arendonk, J. A. M. On the detection of imprinted quantitative trait loci in experimental crosses of outbred species. Genetics 161, 931–938 (2002).
Sandor, C. & Georges, M. On the detection of imprinted quantitative trait loci in line crosses: effect of linkage disequilibrium. Genetics 180, 1167–1175 (2008).
Hager, R., Cheverud, J. M., Roseman, C. & Wolf, J. B. Maternal effects as the cause of parent-of-origin effects that mimic genomic imprinting. Genetics 178, 1755–1762 (2008).
Wolf, J. B. & Wade, M. J. What are maternal effects (and what are they not)? Phil. Trans. R. Soc. B 364, 1107–1115 (2009).
Gleason, G. et al. The serotonin1A receptor gene as a genetic and prenatal maternal environmental factor in anxiety. Proc. Natl Acad. Sci. USA 107, 7592–7597 (2010).
Knott, S. A. et al. Multiple marker mapping of quantitative trait loci in a cross between outbred wild boar and large white pigs. Genetics 149, 1069–1080 (1998). This study developed the first model for identifying imprinted loci in QTL analyses. It was also the first application of the line-cross design to identify parent-of-origin effects.
Mantey, C., Brockmann, G. A., Kalm, E. & Reinsch, N. Mapping and exclusion mapping of genomic imprinting effects in mouse F2 families. J. Hered. 96, 329–338 (2005). This paper developed a formal framework for decomposing and characterizing the effects of imprinted loci using a linear model (as outlined in box 2).
Cui, Y., Cheverud, J. M. & Wu, R. A statistical model for dissecting genomic imprinting through genetic mapping. Genetica 130, 227–239 (2007).
Cui, Y., Lu, Q., Cheverud, J. M., Littell, R. C. & Wu, R. Model for mapping imprinted quantitative trait loci in an inbred F2 design. Genomics 87, 543–551 (2006).
Martin, E. R. & Rampersaud, E. Family-based genetic association tests. Cold Spring Harb. Protoc. http://dx.doi.org/10.1101/pdb.top96 (2011).
Wallace, C. et al. The imprinted DLK1-MEG3 gene region on chromosome 14q32.2 alters susceptibility to type 1 diabetes. Nature Genet. 42, 68–71 (2009).
Belonogova, N. M., Axenovich, T. I. & Aulchenko, Y. S. A powerful genome-wide feasible approach to detect parent-of-origin effects in studies of quantitative traits. Eur. J. Hum. Genet. 18, 379–384 (2010).
Li, J. & Jiang, T. Efficient inference of haplotypes from genotypes on a pedigree. J. Bioinformat. Computat. Biol. 1, 41–69 (2003).
Kong, A. et al. Parental origin of sequence variants associated with complex diseases. Nature 462, 868–874 (2009).
Luedi, P. P., Hartemink, A. J. & Jirtle, R. L. Genome-wide prediction of imprinted murine genes. Genome Res. 15, 875–884 (2005). This work developed and implemented a bioinformatic approach to identify imprinted genes in mice using sequence characteristics to train a statistical model based on a machine-learning algorithm.
Imumorin, I. G. et al. Genome scan for parent-of-origin QTL effects on bovine growth and carcass traits. Front. Genet. 2, 44 (2011).
Cheverud, J. M. et al. Diet-dependent genetic and genomic imprinting effects on obesity in mice. Obesity 19, 160–170 (2010).
Lawson, H. A. et al. Genetic, epigenetic, and gene-by-diet interaction effects underlie variation in serum lipids in a LG/JxSM/J murine model. J. Lipid Res. 51, 2976–2984 (2010).
Magee, D. A. et al. DNA sequence polymorphisms in a panel of eight candidate bovine imprinted genes and their association with performance traits in Irish Holstein-Friesian cattle. BMC Genet. 11, 93 (2010).
Nezer, C. et al. Haplotype sharing refines the location of an imprinted quantitative trait locus with major effect on muscle mass to a 250-kb chromosome segment containing the porcine IGF2 gene. Genetics 165, 277–285 (2003).
Nezer, C. et al. An imprinted QTL with major effect on muscle mass and fat deposition maps to the IGF2 locus in pigs. Nature Genet. 21, 155–156 (1999).
Jeon, J.-T. et al. A paternally expressed QTL affecting skeletal and cardiac muscle mass in pigs maps to the IGF2 locus. Nature Genet. 21, 157–158 (1999).
Van Laere, A.-S. et al. A regulatory mutation in IGF2 causes a major QTL effect on muscle growth in the pig. Nature 425, 832–836 (2003). This investigation demonstrated that a QTL identified in a mapping study is caused by a particular nucleotide substitution in an intron of the imprinted gene IGF2.
Markljung, E. et al. ZBED6, a novel transcription factor derived from a domesticated DNA transposon regulates IGF2 expression and muscle growth. PLoS Biol. 7, e1000256 (2009).
Berkowicz, E. W. et al. Single nucleotide polymorphisms at the imprinted bovine insulin-like growth factor 2 (IGF2) locus are associated with dairy performance in Irish Holstein-Friesian cattle. J. Dairy Res. 78, 1–8 (2011).
Bassett, S. S., Avramopoulos, D. & Fallin, D. Evidence for parent of origin effect in late-onset Alzheimer disease. Am. J. Med. Genet. 114, 679–686 (2002).
Lamb, J. A. et al. Analysis of IMGSAC autism susceptibility loci: evidence for sex limited and parent of origin specific effects. J. Med. Genet. 42, 132–137 (2005).
Francks, C. et al. Parent-of-origin effects on handedness and schizophrenia susceptibility on chromosome 2p12-q11. Hum. Mol. Genet. 12, 3225–3230 (2003).
Cichon, S. et al. A genome screen for genes predisposing to bipolar affective disorder detects a new susceptibility locus on 8q. Hum. Mol. Genet. 10, 2933–2944 (2001).
Lindsay, R. S., Kobes, S., Knowler, W. C., Bennett, P. H. & Hanson, R. L. Genome-wide linkage analysis assessing parent-of-origin effects in the inheritance of type 2 diabetes and BMI in Pima Indians. Diabetes 50, 2850–2857 (2001).
Small, K. S. et al. Identification of an imprinted master trans regulator at the KLF14 locus related to multiple metabolic phenotypes. Nature Genet. 43, 561–564 (2011).
Sha, K. A mechanistic view of genomic imprinting. Annu. Rev. Genom. Hum. Genet. 9, 197–216 (2008).
Cockett, N. E. et al. Polar overdominance at the ovine callipyge locus. Science 273, 236–238 (1996). This report was the first to identify an imprinted locus affecting a quantitative trait and was also the first to identify a locus showing a pattern of dominance imprinting.
Georges, M., Charlier, C. & Cockett, N. The callipyge locus: evidence for the trans interaction of reciprocally imprinted genes. Trends Genet. 19, 248–252 (2003).
Wermter, A. K. et al. Preferential reciprocal transfer of paternal/maternal DLK1 alleles to obese children: first evidence of polar overdominance in humans. Eur. J. Hum. Genet. 16, 1126–1134 (2008).
Sapienza, C., Paquette, J., Pannunzio, P., Albrechtson, S. & Morgan, K. The polar-lethal Ovum mutant gene maps to the distal portion of mouse chromosome 11. Genetics 132, 241–246 (1992).
Takeda, H. et al. The callipyge mutation enhances bidirectional long-range DLK1-GTL2 intergenic transcription in cis. Proc. Natl Acad. Sci. USA 103, 8119–8124 (2006).
Davis, E. et al. RNAi-mediated allelic trans-interaction at the imprinted Rtl1/Peg11 locus. Curr. Biol. 15, 743–749 (2005).
Bidwell, C. A. et al. in Livestock Epigenetics (ed. Khatib, H.) 73–88 (Wiley-Blackwell, 2012).
Lawson, H. A. et al. Genetic effects at pleiotropic loci are context-dependent with consequences for the maintenance of genetic variation in populations. PLoS Genet. 7, e1002256 (2011).
Wagschal, A. & Feil, R. Genomic imprinting in the placenta. Cytogenet. Genome Res. 113, 90–98 (2006).
Mabb, A. M., Judson, M. C., Zylka, M. J. & Philpot, B. D. Angelman syndrome: insights into genomic imprinting and neurodevelopmental phenotypes. Trends Neurosci. 34, 293–303 (2011).
Marcos, L. et al. Genome-wide assessment of imprinted expression in human cells. Genome Biol. 12, R25 (2011).
Luedi, P. P. et al. Computational and experimental identification of novel human imprinted genes. Genome Res. 17, 1723–1730 (2007).
Steinhoff, C., Paulsen, M., Kielbasa, S., Walter, J. & Vingron, M. Expression profile and transcription factor binding site exploration of imprinted genes in human and mouse. BMC Genomics 10, 144 (2009).
Monk, D. et al. Limited evolutionary conservation of imprinting in the human placenta. Proc. Natl Acad. Sci. USA 103, 6623–6628 (2006).
Frost, J. M. & Moore, G. E. The importance of imprinting in the human placenta. PLoS Genet. 6, e1001015 (2010).
Brideau, C. M., Eilertson, K. E., Hagarman, J. A., Bustamante, C. D. & Soloway, P. D. Successful computational prediction of novel imprinted genes from epigenomic features. Mol. Cell. Biol. 30, 3357–3370 (2010).
Mancini-Dinardo, D., Steele, S. J., Levorse, J. M., Ingram, R. S. & Tilghman, S. M. Elongation of the Kcnq1ot1 transcript is required for genomic imprinting of neighboring genes. Genes Dev. 20, 1268–1282 (2006).
Waters, A. J. et al. Parent-of-origin effects on gene expression and DNA methylation in the maize endosperm. Plant Cell 23, 4221–4233 (2011).
Gregg, C. et al. High-resolution analysis of parent-of-origin allelic expression in the mouse brain. Science 329, 643–648 (2010).
Wang, X. et al. Transcriptome-wide identification of novel imprinted genes in neonatal mouse brain. PLoS ONE 3, e3839 (2008).
DeVeale, B., van der Kooy, D. & Babak, T. Critical evaluation of imprinted gene expression by RNA-Seq: a new perspective. PLoS Genet. 8, e1002600 (2012).
Bell, A. C. & Felsenfeld, G. Methylation of a CTCF-dependent boundary controls imprinted expression of the Igf2 gene. Nature 405, 482–485 (2000).
Hark, A. T. et al. CTCF mediates methylation-sensitive enhancer-blocking activity at the H19/Igf2 locus. Nature 405, 486–489 (2000).
Lister, R. et al. Human DNA methylomes at base resolution show widespread epigenomic differences. Nature 462, 315–322 (2009).
Harris, R. A. et al. Comparison of sequencing-based methods to profile DNA methylation and identification of monoallelic epigenetic modifications. Nature Biotechnol. 28, 1097–1105 (2010).
Li, M. et al. An atlas of DNA methylomes in porcine adipose and muscle tissues. Nature Commun. 3, 850 (2012).
Hager, R., Cheverud, J. M. & Wolf, J. B. Relative contribution of additive, dominance and imprinting effects to phenotypic variation in body size and growth between divergent selection lines of mice. Evolution 63, 1118–1128 (2009).
Falconer, D. S. & Mackay, T. F. C. Introduction to Quantitative Genetics 4th edn (Longman, 1996).
Varrault, A. et al. Zac1 regulates an imprinted gene network critically involved in the control of embryonic growth. Dev. Cell 11, 711–722 (2006).
Wolf, J. B. & Cheverud, J. M. A framework for detecting and characterizing genetic backgroun-dependent imprinting effects. Mamm. Genome 20, 681–698 (2009).
Li, S. et al. Bayesian mapping of genome-wide epistatic imprinted loci for quantitative traits. Theor. Appl. Genet. 124, 1561–1571 (2012).
Reilly, K. M. et al. An imprinted locus epistatically influences Nstr1 and Nstr2 to control resistance to nerve sheath tumors in a neuorofibromatosis type 1 mouse model. Cancer Res. 66, 62–68 (2006).
Cattanach, B. M., Beechey, C. V. & Peters, J. Interactions between imprinting effects: summary and review. Cytogenet. Genome Res. 113, 17–23 (2006).
Acknowledgements
H.A.L. is supported by the US National Institute of Diabetes and Digestive and Kidney Diseases of the US National Institutes of Health (awards K01DK095003 to H.A.L. and P30DK056341 to the Washington University School of Medicine Nutrition and Obesity Research Center). J.B.W. is supported by grant support from the UK Biotechnology and Biological Sciences Research Council (BBSRC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Glossary
- Epigenetic
-
Pertaining to a difference in phenotype resulting from variations in DNA chemistry rather than DNA sequence. Epigenetic changes can be cell specific, can be modified by environmental factors, can affect gene expression and may underlie some parent-of-origin effects on complex traits.
- Genomic imprinting
-
An epigenetic phenomenon in which the expression of a gene occurs in a parent-of-origin-dependent manner.
- Complex traits
-
Quantitative traits that are influenced by many genetic, epigenetic and environmental factors and their interactions.
- Line-cross design
-
An approach to quantitative trait locus mapping in which two non-inbred lines are crossed to produce a mapping population. The approach assumes that the two lines are fixed for different quantitative trait locus alleles, but there is variation at marker loci segregating within the lines.
- Quantitative trait locus
-
(QTL). A region of the genome in which genetic variation at a marker locus is significantly correlated with phenotypic variation for a complex trait.
- Parental genetic effects
-
Effects that occur when genes expressed in the mother or father have a causal influence on the phenotype of the offspring.
- Parental imprinting
-
A phenomenon that occurs when either only the maternally or only the paternally inherited allele affects a phenotype. In a two-allele system, genotypes will group into two phenotypic classes based on the maternally or paternally expressed allele.
- Advanced intercross
-
The result of continued random mating of a population derived from a cross between inbred lines. Advanced intercrosses provide higher resolution for quantitative trait loci than traditional (for example, F2) mapping approaches because of the accumulation of recombination through each generation of random mating.
- Dominance imprinting
-
A complex imprinting pattern in which the parent of origin of alleles affects dominance at a locus. For example, bipolar dominance imprinting occurs when one heterozygote shows overdominance and the reciprocal heterozygote shows underdominance.
- Allele-specific biases
-
Biases that occur when the two alleles in a heterozygote are not functionally equivalent. This situation can arise from an expression bias wherein one allele is expressed at a higher level than the other (the null expectation being that both alleles will be expressed at approximately the same level). There can also be methylation biases, wherein one allele is preferentially methylated (or unmethylated); this can underlie allele-specific expression biases.
- Differentially methylated regions
-
(DMRs). Genomic regions in which the pattern of methylation (the ratio of methylated to unmethylated sequence) is different between two alleles at the same locus.
Rights and permissions
About this article
Cite this article
Lawson, H., Cheverud, J. & Wolf, J. Genomic imprinting and parent-of-origin effects on complex traits. Nat Rev Genet 14, 609–617 (2013). https://doi.org/10.1038/nrg3543
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrg3543
This article is cited by
-
Phenome-wide analyses identify an association between the parent-of-origin effects dependent methylome and the rate of aging in humans
Genome Biology (2023)
-
Rhesus macaque social functioning is paternally, but not maternally, inherited by sons: potential implications for autism
Molecular Autism (2023)
-
Epigenetic and metabolic reprogramming in inflammatory bowel diseases: diagnostic and prognostic biomarkers in colorectal cancer
Cancer Cell International (2023)
-
Mir125b-2 imprinted in human but not mouse brain regulates hippocampal function and circuit in mice
Communications Biology (2023)
-
Noncanonical regulation of imprinted gene Igf2 by amyloid-beta 1–42 in Alzheimer’s disease
Scientific Reports (2023)