Abstract
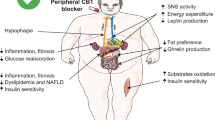
When compared with other modifiable cardiovascular risk factors, such as hypertension, dyslipidemia and smoking, obesity remains a surprisingly puzzling condition to prevent and treat. The history of the development of anti-obesity drugs has known more defeats than even partial victories. With very few drugs on the market, and bad publicity related to adverse events, obesity remains an almost completely unmet challenge for the pharmaceutical industry. In light of past experience with endocannabinoid-system antagonists, such as rimonabant, we propose that a major paradigm shift in clinical practice might be necessary to justify the use of pharmacotherapy for obesity. Furthermore, we suggest that the criteria currently used by regulatory authorities to evaluate and approve anti-obesity drugs should be rigorously re-examined. Finally, we discuss how pharmacological approaches that aim to counteract overactivity of the endocannabinoid system should be revisited in the future to treat visceral (intra-abdominal) obesity and its metabolic consequences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Howlett, A. C. et al. Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology 47 (Suppl. 1), 345–358 (2004).
Devane, W. A. et al. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 258, 1946–1949 (1992).
Di Marzo, V. The endocannabinoid system in obesity and type 2 diabetes. Diabetologia 51, 1356–1367 (2008).
Foltin, R. W., Brady, J. V. & Fischman, M. W. Behavioral analysis of marijuana effects on food intake in humans. Pharmacol. Biochem. Behav. 25, 577–582 (1986).
Van Gaal, L. F. et al. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe study. Lancet 365, 1389–1397 (2005).
Després, J. P. et al. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N. Engl. J. Med. 353, 2121–2134 (2005).
Scheen, A. J. et al. Efficacy and tolerability of rimonabant in overweight or obese patients with type 2 diabetes: a randomised controlled study. Lancet 368, 1660–1672 (2006).
Pi-Sunyer, F. X. et al. Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients: RIO-North America: a randomized controlled trial. JAMA 295, 761–775 (2006).
Cahill, K. & Ussher, M. Cannabinoid type 1 receptor antagonists (rimonabant) for smoking cessation. Cochrane Database of Systematic Reviews, Issue 3. Art. No.: CD005353 10.1002/14651858.CD005353.pub3 (2009).
Prospective Studies Collaboration. Body-mass index and cause-specific mortality in 900,000 adults: collaborative analyzes of 57 prospective studies. Lancet 373, 1083–1096 (2009).
Després, J. P. & Lemieux, I. Abdominal obesity and metabolic syndrome. Nature 444, 881–887 (2006).
Després, J. P. et al. Abdominal obesity and the metabolic syndrome: contribution to global cardiometabolic risk. Arterioscler. Thromb. Vasc. Biol. 28, 1039–1049 (2008).
Després, J. P., Lemieux, I. & Prud'homme, D. Treatment of obesity: need to focus on high risk abdominally obese patients. BMJ 322, 716–720 (2001).
Després, J. P. Drug treatment for obesity. We need more studies in men at higher risk of coronary events. BMJ 322, 1379–1380 (2001).
Rucker, D., Padwal, R., Li, S. K., Curioni, C. & Lau, D. C. Long term pharmacotherapy for obesity and overweight: updated meta-analysis. BMJ 335, 1194–1199 (2007).
Ritchie, S. A. & Connell, J. M. The link between abdominal obesity, metabolic syndrome and cardiovascular disease. Nutr. Metab. Cardiovasc. Dis. 17, 319–326 (2007).
Fabricatore, A. N. et al. Attrition from randomized controlled trials of pharmacological weight loss agents: a systematic review and analysis. Obes. Rev. 10, 333–341 (2009).
Blüher, M. et al. Dysregulation of the peripheral and adipose tissue endocannabinoid system in human abdominal obesity. Diabetes 55, 3053–3060 (2006).
Di Marzo, V. et al. Changes in plasma endocannabinoid levels in viscerally obese men following a 1 year lifestyle modification program and waist circumference reduction: associations with changes in metabolic risk factors. Diabetologia 52, 213–217 (2009).
Nissen, S. E. et al. Effect of rimonabant on progression of atherosclerosis in patients with abdominal obesity and coronary artery disease: the STRADIVARIUS randomized controlled trial. JAMA 299, 1547–1560 (2008).
Carr, R. D. et al. A two-year study to assess the efficacy, safety, and tolerability of taranabant in obese patients: 52 week results. Atherosclerosis 9, 12 (2008).
Pacher, P. & Steffens, S. The emerging role of the endocannabinoid system in cardiovascular disease. Semin. Immunopathol. 31, 63–77 (2009).
Van Gaal, L. F. et al. Long-term effect of CB1 blockade with rimonabant on cardiometabolic risk factors: two year results from the RIO-Europe Study. Eur. Heart J. 29, 1761–1771 (2008).
Osei-Hyiaman, D. et al. Hepatic CB1 receptor is required for development of diet-induced steatosis, dyslipidemia, and insulin and leptin resistance in mice. J. Clin. Invest. 118, 3160–3169 (2008).
Buettner, C. et al. Leptin controls adipose tissue lipogenesis via central, STAT3-independent mechanisms. Nat. Med. 14, 667–675 (2008).
Acknowledgements
The authors thank Isabelle Lemieux (Centre de recherche de l'Institut universitaire de cardiologie et de pneumologie de Québec, Canada) for valuable help during preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Vincenzo Di Marzo has declared associations with the following companies: GW Pharmaceuticals (consultant and research support) and Sanofi-Aventis (speakers bureau and research support).
Jean-Pierre Després has declared associations with the following companies: Abbott Laboratories (speakers bureau), AstraZeneca (speakers bureau), Eli Lilly (research support), GlaxoSmithKline (speakers bureau and research support), INNODIA (consultant), Merck Sharp & Dohme (consultant), Novartis (consultant), Pfizer Canada (speakers bureau), Sanofi-Aventis (consultant, speakers bureau and research support) and Solvay (speakers bureau).
Supplementary information
Rights and permissions
About this article
Cite this article
Di Marzo, V., Després, JP. CB1 antagonists for obesity—what lessons have we learned from rimonabant?. Nat Rev Endocrinol 5, 633–638 (2009). https://doi.org/10.1038/nrendo.2009.197
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2009.197
This article is cited by
-
AM6527, a neutral CB1 receptor antagonist, suppresses opioid taking and seeking, as well as cocaine seeking in rodents without aversive effects
Neuropsychopharmacology (2024)
-
Overweight, Obesity, and CVD Risk: a Focus on Visceral/Ectopic Fat
Current Atherosclerosis Reports (2022)
-
Cerebral μ-opioid and CB1 receptor systems have distinct roles in human feeding behavior
Translational Psychiatry (2021)
-
The cannabinoid ligand LH-21 reduces anxiety and improves glucose handling in diet-induced obese pre-diabetic mice
Scientific Reports (2017)
-
Pro-inflammatory obesity in aged cannabinoid-2 receptor-deficient mice
International Journal of Obesity (2016)