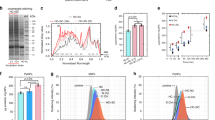
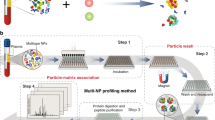
Abstract
Nanoparticle applications in biotechnology and biomedicine are steadily increasing. In biological fluids, proteins bind to nanoparticles that form the protein corona, crucially affecting the nanoparticles' biological identity. As the corona affects in vitro and/or in vivo nanoparticle applications, we developed a method to obtain time-resolved protein corona profiles formed on various nanoparticles. After incubation in plasma or a similar biofluid, or after injection into a mouse, the first analytical step is sedimentation of the nanoparticle-protein complexes through a sucrose cushion, thereby allowing analysis of early corona formation time points. Next, corona profiles are visualized by gel electrophoresis and quantitatively analyzed after tryptic digestion using label-free liquid chromatography–high-resolution mass spectrometry. In contrast to other approaches, our established methodology allows the researcher to obtain qualitative and quantitative high-resolution corona signatures. The protocol can be readily extended to the investigation of protein coronas from various nanomaterials (as an example, we applied this protocol to different silica nanoparticles (SiNPs) and polystyrene nanoparticles (PSNPs)). Depending on the number of samples, the protocol from nanoparticle-protein complex recovery to data evaluation takes ∼8–12 d to complete.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Reese, M. Nanotechnology: using co-regulation to bring regulation of modern technologies into the 21st century. Health Matrix Clevel. 23, 537–572 (2013).
Webster, T.J. Interview: Nanomedicine: past, present and future. Nanomedicine (Lond.) 8, 525–529 (2013).
Nystrom, A.M. & Fadeel, B. Safety assessment of nanomaterials: implications for nanomedicine. J. Control Release 161, 403–408 (2012).
Oberdorster, G. Nanotoxicology: in vitro-in vivo dosimetry. Environ. Health Perspect. 120, A13 (2012).
Rauscher, H., Sokull-Kluttgen, B. & Stamm, H. The European Commission's recommendation on the definition of nanomaterial makes an impact. Nanotoxicology 7, 1195–1197 (2013).
Monopoli, M.P., Aberg, C., Salvati, A. & Dawson, K.A. Biomolecular coronas provide the biological identity of nanosized materials. Nat. Nanotechnol. 7, 779–786 (2012).
Monopoli, M.P., Bombelli, F.B. & Dawson, K.A. Nanobiotechnology: nanoparticle coronas take shape. Nat. Nanotechnol. 6, 11–12 (2011).
Tenzer, S. et al. Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology. Nat. Nanotechnol. 8, 772–781 (2013).
Capriotti, A.L. et al. DNA affects the composition of lipoplex protein corona: a proteomics approach. Proteomics 11, 3349–3358 (2012).
Xia, X.R., Monteiro-Riviere, N.A. & Riviere, J.E. An index for characterization of nanomaterials in biological systems. Nat. Nanotechnol. 5, 671–675 (2010).
Nel, A.E. et al. Understanding biophysicochemical interactions at the nano-bio interface. Nat. Mater. 8, 543–557 (2009).
Tenzer, S. et al. Nanoparticle size is a critical physicochemical determinant of the human blood plasma corona: a comprehensive quantitative proteomic analysis. ACS Nano 5, 7155–7167 (2011).
Zhang, H. et al. Quantitative proteomics analysis of adsorbed plasma proteins classifies nanoparticles with different surface properties and size. Proteomics 11, 4569–4577 (2011).
Salvati, A. et al. Transferrin-functionalized nanoparticles lose their targeting capabilities when a biomolecule corona adsorbs on the surface. Nat. Nanotechnol 8, 137–143 (2013).
Wegner, K.D., Jin, Z., Linden, S., Jennings, T.L. & Hildebrandt, N. Quantum dot–based Forster resonance energy transfer immunoassay for sensitive clinical diagnostics of low-volume serum samples. ACS Nano 7, 7411–7419 (2013).
Barkam, S., Saraf, S. & Seal, S. Fabricated micro-nano devices for in vivo and in vitro biomedical applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 5, 544–568 (2013).
Dobrovolskaia, M.A., Germolec, D.R. & Weaver, J.L. Evaluation of nanoparticle immunotoxicity. Nat. Nanotechnol. 4, 411–414 (2009).
Distler, U. et al. Drift time-specific collision energies enable deep-coverage data-independent acquisition proteomics. Nat. Methods 11, 167–170 (2014).
Meister, S. et al. Nanoparticulate flurbiprofen reduces amyloid-β42 generation in an in vitro blood-brain barrier model. Alzheimer's Res. Ther. 5, 51 (2013).
Evans, C. et al. An insight into iTRAQ: where do we stand now? Anal. Bioanal. Chem. 404, 1011–1027 (2012).
Bantscheff, M., Lemeer, S., Savitski, M.M. & Kuster, B. Quantitative mass spectrometry in proteomics: critical review update from 2007 to the present. Anal. Bioanal. Chem. 404, 939–965 (2012).
Tate, S., Larsen, B., Bonner, R. & Gingras, A.C. Label-free quantitative proteomics trends for protein-protein interactions. J. Proteomics 81, 91–101 (2013).
Patel, V.J. et al. A comparison of labeling and label-free mass spectrometry-based proteomics approaches. J. Proteome Res. 8, 3752–3759 (2009).
Cai, X. et al. Characterization of carbon nanotube protein corona by using quantitative proteomics. Nanomedicine 9, 583–593 (2013).
Farrah, T. et al. A high-confidence human plasma proteome reference set with estimated concentrations in PeptideAtlas. Mol. Cell. Proteomics 10, M110.006353 (2011).
Nahnsen, S., Bielow, C., Reinert, K. & Kohlbacher, O. Tools for label-free peptide quantification. Mol. Cell. Proteomics 12, 549–556 (2013).
Gethings, L.A. & Connolly, J.B. Simplifying the proteome: analytical strategies for improving peak capacity. Adv. Exp. Med. Biol. 806, 59–77 (2014).
Gebauer, J.S. et al. Impact of the nanoparticle-protein corona on colloidal stability and protein structure. Langmuir 28, 9673–9679 (2012).
Walczyk, D., Bombelli, F.B., Monopoli, M.P., Lynch, I. & Dawson, K.A. What the cell 'sees' in bionanoscience. J. Am. Chem. Soc. 132, 5761–5768 (2010).
Casals, E., Pfaller, T., Duschl, A., Oostingh, G.J. & Puntes, V. Time evolution of the nanoparticle protein corona. ACS Nano 4, 3623–3632 (2010).
Dell'Orco, D., Lundqvist, M., Oslakovic, C., Cedervall, T. & Linse, S. Modeling the time evolution of the nanoparticle-protein corona in a body fluid. PLoS ONE 5, e10949 (2010).
Barran-Berdon, A.L. et al. Time evolution of nanoparticle-protein corona in human plasma: relevance for targeted drug delivery. Langmuir 29, 6485–6494 (2013).
Natte, K. et al. Impact of polymer shell on the formation and time evolution of nanoparticle-protein corona. Colloids Surf. B Biointerfaces 104, 213–220 (2013).
Rocker, C., Potzl, M., Zhang, F., Parak, W.J. & Nienhaus, G.U. A quantitative fluorescence study of protein monolayer formation on colloidal nanoparticles. Nat. Nanotechnol. 4, 577–580 (2009).
Lundqvist, M. et al. The evolution of the protein corona around nanoparticles: a test study. ACS Nano 5, 7503–7509 (2011).
Owens, D.E. III & Peppas, N.A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
Mahmoudi, M. et al. Irreversible changes in protein conformation due to interaction with superparamagnetic iron oxide nanoparticles. Nanoscale 3, 1127–1138 (2011).
Ehrenberg, M.S., Friedman, A.E., Finkelstein, J.N., Oberdorster, G. & McGrath, J.L. The influence of protein adsorption on nanoparticle association with cultured endothelial cells. Biomaterials 30, 603–610 (2009).
Gessner, A., Lieske, A., Paulke, B. & Muller, R. Influence of surface charge density on protein adsorption on polymeric nanoparticles: analysis by two-dimensional electrophoresis. Eur. J. Pharm. Biopharm. 54, 165–170 (2002).
Dobrovolskaia, M.A. et al. Interaction of colloidal gold nanoparticles with human blood: effects on particle size and analysis of plasma protein binding profiles. Nanomedicine (Lond.) 5, 106–117 (2009).
Lundqvist, M. et al. Nanoparticle size and surface properties determine the protein corona with possible implications for biological impacts. Proc. Natl. Acad. Sci. USA 105, 14265–14270 (2008).
Chakraborty, S. et al. Contrasting effect of gold nanoparticles and nanorods with different surface modifications on the structure and activity of bovine serum albumin. Langmuir 27, 7722–7731 (2011).
Dutta, D. et al. Adsorbed proteins influence the biological activity and molecular targeting of nanomaterials. Toxicol. Sci. 100, 303–315 (2007).
Cedervall, T. et al. Detailed identification of plasma proteins adsorbed on copolymer nanoparticles. Angew Chem. Int. Ed. Engl. 46, 5754–5756 (2007).
Lacerda, S.H. et al. Interaction of gold nanoparticles with common human blood proteins. ACS Nano 4, 365–379 (2010).
Lindman, S. et al. Systematic investigation of the thermodynamics of HSA adsorption to N-iso-propylacrylamide/N-tert-butylacrylamide copolymer nanoparticles. Effects of particle size and hydrophobicity. Nano Lett. 7, 914–920 (2007).
Mahmoudi, M. et al. Temperature: the 'ignored' factor at the NanoBio interface. ACS Nano 7, 6555–6562 (2013).
Mahmoudi, M., Laurent, S., Shokrgozar, M.A. & Hosseinkhani, M. Toxicity evaluations of superparamagnetic iron oxide nanoparticles: cell 'vision' versus physicochemical properties of nanoparticles. ACS Nano 5, 7263–7276 (2011).
Monopoli, M.P. et al. Physical-chemical aspects of protein corona: relevance to in vitro and in vivo biological impacts of nanoparticles. J. Am. Chem. Soc. 133, 2525–2534 (2011).
Goppert, T.M. & Muller, R.H. Protein adsorption patterns on poloxamer- and poloxamine-stabilized solid lipid nanoparticles (SLN). Eur. J. Pharm. Biopharm. 60, 361–372 (2005).
Labarre, D. et al. Interactions of blood proteins with poly(isobutylcyanoacrylate) nanoparticles decorated with a polysaccharidic brush. Biomaterials 26, 5075–5084 (2005).
Acknowledgements
Grant support for this study: Deutsche Forschungsgemeinschaft (DFG)-SPP1313, DFG-SFB490/Z3; Bundesministerium für Bildung und Forschung (BMBF)-MRCyte/NanoBEL/DENANA; Zeiss-ChemBioMed; University Mainz Forschungszentrum Immunologie; Research Center for Immunology (FZI); and Stiftung Rheinland-Pfalz (NANOSCH, NanoScreen).
Author information
Authors and Affiliations
Contributions
S.T., R.H.S., J.K., U.D. and D.D. developed the protocol; D.D., U.D., J.K., A.H., W.S., D.W., S.K.K., S.T. and R.H.S. conducted the experiments, interpreted the data and drafted the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Settings for ISOQuant post-processing for label-free quantification. (XLSX 11 kb)
Supplementary Table 2
Integrated summary of corona proteins identified on silica and polystyrene nanoparticles8,12, containing averaged (typical) abundance values (expressed in parts per million of total corona protein) for each nanoparticle type, including human plasma as a reference for future studies. The table also contains information regarding functional annotation, molecular weight and isoelectric points for corona-associated proteins. (XLSX 63 kb)
Rights and permissions
About this article
Cite this article
Docter, D., Distler, U., Storck, W. et al. Quantitative profiling of the protein coronas that form around nanoparticles. Nat Protoc 9, 2030–2044 (2014). https://doi.org/10.1038/nprot.2014.139
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.139
This article is cited by
-
Coronas of micro/nano plastics: a key determinant in their risk assessments
Particle and Fibre Toxicology (2022)
-
Detection of ovarian cancer via the spectral fingerprinting of quantum-defect-modified carbon nanotubes in serum by machine learning
Nature Biomedical Engineering (2022)
-
Concentration and composition of the protein corona as a function of incubation time and serum concentration: an automated approach to the protein corona
Analytical and Bioanalytical Chemistry (2022)
-
Comprehensive and systematic characterization of multi-functionalized cisplatin nano-conjugate: from the chemistry and proteomic biocompatibility to the animal model
Journal of Nanobiotechnology (2022)
-
Protein nanoparticles in drug delivery: animal protein, plant proteins and protein cages, albumin nanoparticles
Journal of Nanobiotechnology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.