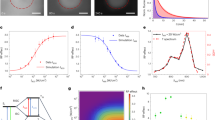
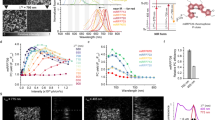
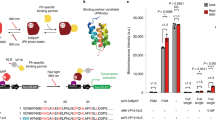
Abstract
The phototoxic red fluorescent GFP-like protein KillerRed has recently been described. The phototoxicity of KillerRed exceeds that of EGFP by at least 1,000-fold, making it the first fully genetically encoded photosensitizer. KillerRed opens up new possibilities for precise light-induced cell killing and target protein inactivation. Because KillerRed is encoded by a gene, it can be expressed in a spatially and temporally regulated manner, under a chosen promoter, and fused with the desired protein of interest or localization signal. Here we provide a protocol for target protein inactivation in cell culture using KillerRed. As KillerRed is a new tool, the protocol focuses on aspects that will allow users to maximize the potential of this protein, guiding the design of chimeric constructs, recommended control experiments and preferred illumination parameters. The protocol, which describes target protein visualization and subsequent inactivation, is a 2- or 3-d procedure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lippincott-Schwartz, J. & Patterson, G.H. Development and use of fluorescent protein markers in living cells. Science 300, 87–91 (2003).
Chudakov, D.M., Lukyanov, S. & Lukyanov, K.A. Fluorescent proteins as a toolkit for in vivo imaging. Trends Biotechnol. 23, 605–613 (2005).
Surrey, T. et al. Chromophore-assisted light inactivation and self-organization of microtubules and motors. Proc. Natl. Acad. Sci. USA 95, 4293–4298 (1998).
Bulina, M.E. et al. A genetically encoded photosensitizer. Nat. Biotechnol. 24, 95–99 (2006).
Eustace, B.K., Buchstaller, A. & Jay, D.G. Adapting chromophore-assisted laser inactivation for high throughput functional proteomics. Brief Funct. Genomic Proteomic 1, 257–265 (2002).
Huang, Z. A review of progress in clinical photodynamic therapy. Technol. Cancer Res. Treat. 4, 283–293 (2005).
Griffin, B.A., Adams, S.R. & Tsien, R.Y. Specific covalent labeling of recombinant protein molecules inside live cells. Science 281, 269–272 (1998).
Adams, S.R. et al. New biarsenical ligands and tetracysteine motifs for protein labeling in vitro and in vivo: synthesis and biological applications. J. Am. Chem. Soc. 124, 6063–6076 (2002).
Grate, D. & Wilson, C. Laser-mediated, site-specific inactivation of RNA transcripts. Proc. Natl. Acad. Sci. USA 96, 6131–6136 (1999).
Tsien, R.Y. Imagining imaging's future. Nat. Rev. Mol. Cell Biol. Suppl: SS16–21 (2003).
Grabenbauer, M. et al. Correlative microscopy and electron tomography of GFP through photooxidation. Nat. Methods 2, 857–862 (2005).
Filippin, L. et al. Improved strategies for the delivery of GFP-based Ca2+ sensors into the mitochondrial matrix. Cell Calcium 37, 129–136 (2005).
Iinuma, S. et al. In vivo fluence rate and fractionation effects on tumor response and photobleaching: photodynamic therapy with two photosensitizers in an orthotopic rat tumor model. Cancer Res. 59, 6164–6170 (1999).
Skene, J.H. & Virag, I. Posttranslational membrane attachment and dynamic fatty acylation of a neuronal growth cone protein, GAP-43. J. Cell Biol. 108, 613–624 (1989).
Shaner, N.C. et al. Improved monomeric red, orange and yellow fluorescent proteins derived from Discosoma sp. red fluorescent protein. Nat. Biotechnol. 22, 1567–1572 (2004).
Fradkov, A.F. et al. Far-red fluorescent tag for protein labelling. Biochem. J. 368, 17–21 (2002).
Acknowledgements
This work was supported by grants from the Howard Hughes Medical Institute (HHMI 55005618), molecular and cell biology program RAS and EC FP-6 integrated project LSHG-CT-2003-503259. D.M.C. is supported by grants of the president of Russian Federation MK-8236.2006.4 and Russian Science Support Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
K.A.L. and D.M.C. have a pending patent application on KillerRed uses.
Rights and permissions
About this article
Cite this article
Bulina, M., Lukyanov, K., Britanova, O. et al. Chromophore-assisted light inactivation (CALI) using the phototoxic fluorescent protein KillerRed. Nat Protoc 1, 947–953 (2006). https://doi.org/10.1038/nprot.2006.89
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.89
This article is cited by
-
All-optical spatiotemporal mapping of ROS dynamics across mitochondrial microdomains in situ
Nature Communications (2023)
-
Tailoring photosensitive ROS for advanced photodynamic therapy
Experimental & Molecular Medicine (2021)
-
A LED light for photo-inducible cell ablation by miniSOG
Optical and Quantum Electronics (2021)
-
The effects of photodynamic therapy on leukemia cells mediated by KillerRed, a genetically encoded fluorescent protein photosensitizer
BMC Cancer (2019)
-
A new bioluminescence-based tool for modulating target proteins in live cells
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.